| Research Article | ||
Open Vet. J.. 2025; 15(2): 1032-1042 Open Veterinary Journal, (2025), Vol. 15(2): 1032-1042 Research Article Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopyAmanda Dominica Theqla1, Srikanth Karnati2, and Dwi Liliek Kusindarta1*1Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 2Institute of Anatomy and Cell Biology, Julius Maximillian University, Wuerzburg, Germany *Corresponding Author: Dwi Liliek Kusindarta. Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia. Email: indarta [at] ugm.ac.id Submitted: 12/08/2024 Accepted: 19/12/2024 Published: 28/02/2025 © 2025 Open Veterinary Journal
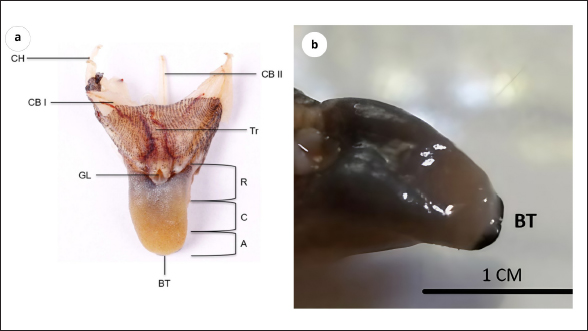
AbstractBackground: The forest dragon (Gonocephalus chamaeleontinus) is a reptile of the Agamidae family, and its distribution includes Indonesia and Malaysia. The forest dragon uses its tongue to catch insects and invertebrates. In terms of morphology, the tongue of the Agamidae family is different from other reptiles. The study of morphology in the tongues of Agamidae is crucial for understanding their feeding behavior, prey capture mechanisms, and evolutionary relationships. Aim: This research attempts to analyze the morphology of the dorsal surface of the tongue of G. chamaeleontinus by using the scanning electron microscope (SEM) and its histological structure by using hematoxylin-eosin (HE) staining. Methods: This study achieves the SEM and light microscope images using hematoxylin eosin stains and employs six samples of G. chamaeleontinus. Results: The tongue of G. chamaeleontinus separated into three distinct parts: the apex (A), corpus (C), and radix ®. The structure’s A contains dome-shaped papillae (DP). The C section contains DP and circumvallate papillae. The R contains scale-like papillae. Additionally, histological analysis using HE stains revealed the taste buds on the DP and circumvallate papillae and the presence of lingual salivary glands (SG) on the lamina propria mucosa. Conclusion: The tongue’s papillae of G. chamaeleontinus comprise sensory and mechanic papillae, which are also completed by lingual SG. Keywords: Gonocephalus chamaeleontinus, Tongue, Light microscopy, Scanning electron microscopy. IntroductionThe forest dragon, Gonocephalus chamaeleontinus (G. chamaeleontinus), is a species of reptile from the Agamidae family that is related to the Iguanidae family. This reptile is found in humid rainforests at low altitudes up to about 500 m above sea level with a distribution on the West Coast of Peninsular Malaysia (Tioman and Pahang Islands), Sumatra, Mentawai and Natuna Islands, and Java (Harlow et al., 2010). Recently, the population of G. chamaeleontinus in the wild has been declining due to the increasing public interest in these reptiles as exotic pets (Howard, 2018). Gonocephalus chamaeleontinus is a diurnal and arboreal animal found in bushes and trees. Gonocephalus chamaeleontinus plays a vital role in nature as a pest controller due to its diet consisting of insects and small invertebrates. This natural pest control behavior helps regulate insect populations in their habitats, contributes to ecosystem balance, and reduces potential agricultural pest damage (Belagalla, 2024; Tiwari, 2024). Gonocephalus chamaeleontinus uses their tongues to lick, capture, and swallow their prey (Herrel et al., 2005). In general, the tongue structure is morphologically divided into three parts: apex (A), corpus (C), and radix (R), in which each of it has rough-textured papillae (Anjani et al., 2023; Selan et al., 2023). The tongue of Agamidae is short and thick and is classified as a “fleshy and sticky tongue” that can extend and stick to capture prey (Schwenk, 2000). The tongues can produce thick, sticky mucus at the tip of the tongue (Ferando et al., 2010). They catch insects by opening their mouths, rapidly advancing their bodies slightly, and then sticking out their sticky, saliva-coated tongues in the direction of the target. The insect will stick to the tongue and then be pulled into its mouth (Meyers et al., 2002). In terms of morphology, the tongue of the Agamidae family is different from the other reptile species (Schwenk, 2000). The study of morphology in the tongues of Agamidae, a family of lizards that includes various species such as the bearded dragon, is crucial for understanding their feeding behavior, prey capture mechanisms, and evolutionary relationships. Research by Delheusy et al. (1994) highlights the importance of comparing tongue morphology in different lizard species to understand the evolutionary relationships within the group. The study emphasizes that phylogeny plays a significant role in determining tongue morphology, supporting the classification of lizards into distinct groups based on their evolutionary history. Zghikh et al. (2014) emphasize the need for detailed knowledge of the distribution of muscle fiber types in the tongues of iguanian lizards similar to Agamidae to enhance our understanding of the functional aspects of their tongues. This highlights the importance of studying the microscopic structure of the tongue muscles to gain insights into how these lizards use their tongues for feeding and other activities. Moreover, the adaptations observed in the tongues of other lizards, such as the blue-tongued skink (Tiliqua scincoides), indicate that the morphology of the lingual papillae is influenced by both the living environment and dietary preferences (Abbate et al., 2009). The presence of distinct morphological areas on the dorsal surface of the tongue, as noted in various studies, highlights the evolutionary significance of these adaptations in response to ecological pressures (Abbate et al., 2009). For G. chamaeleontinus, the scanning electron microscope (SEM) analysis may reveal similar adaptations that enhance its feeding efficiency, particularly in capturing and manipulating prey. In addition, the evolutionary implications of tongue morphology are underscored by the relationship between tongue structure and feeding strategies. For example, the bifurcated tongue observed in some lizard species, which aids in prey capture and manipulation, may also be present in G. chamaeleontinus, suggesting a convergent evolution of feeding adaptations among squamates (Yang and Wang, 2016). The morphological characteristics of the tongue, including the arrangement and type of lingual papillae, play a crucial role in the mechanics of feeding, as they facilitate the transport of food within the oral cavity (Abbate et al., 2008). Furthermore, the SEM studies of other reptiles have demonstrated that the structural adaptations of the tongue can enhance sensory functions, such as chemoreception, which is vital for locating prey (El-Mansi et al., 2020). This aspect is particularly relevant for lizards in common and especially for G. chamaeleontinus, as its feeding behavior may rely heavily on both visual and chemical cues to effectively hunt in its natural habitat. Until now, there has been no research on the morphology and histology of the tongue of G. chamaeleontinus. To determine the morphology of the tongue of the G. chamaeleontinus, it is necessary to study the structure of the tongue using an SEM and light microscopy (LM). It is hoped that this study of the tongue of G. chamaeleontinus provides critical insights into the biological and ecological adaptations of this species, particularly in relation to its feeding behavior and evolutionary strategies. The morphological characteristics of the tongue, as revealed by the SEM, can elucidate how this reptile has adapted to its ecological niche, enhancing its foraging efficiency and overall survival. Materials and MethodsSpecimens and ethical clearanceThe specimens included in this study consisted of six adult G. chamaeleontinus without distinction of sex. The requirements for G. chamaeleontinus to be categorized as an adult animal are that it has a body length, including the tail, reaching 40 cm; an adult forest dragon has an elongated head and prominent dorsal crest and often displays vibrant green or brown hues. The calculation of the sample size was based on GPower software (HHU, Duseldorf, Germany), with full consideration of the ethics committee. The species was extracted from Kulon Progo, Special Region of Yogyakarta, Indonesia. The capture procedure received approval from the Ethics Commission of the Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia (EC number: 134/EC-FKH/Int./2023). The morphological traits were determined at the Animal Systematics Laboratory in the Faculty of Biology at Universitas Gadjah Mada. Conservation statusGonocephalus chamaeleontinus is registered as least concern in the International Union for Conservation of Nature. Gonocephalus chamaeleontinus is not a protected species in Indonesia (Regulation of the Minister of Environment and Forestry Republic of Indonesia No P.20/MENLHK/SETJEN/KUM.1/6/2018). Animal preparatory proceduresSix animals were euthanized by using a high dosage of 50 mg/kg B.W. of ketamine (Kepro, Maagdenburgstraat, Holand) and 1.25 mg/kg B.W. of xylazine (Interchemie, Metaalweg, Holand). The cavum oris was gently opened to separate the cranium and mandible, and the mandible was fixed on a wooden board to separate the basal part of the tongue from the mandible. The samples were cleaned and rinsed using working phosphate buffer saline (PBS) (1x PBS after dilution) (PBS 10x, pH 7.4, Nacalai Tesque, Kyoto, Japan). Three samples were kept in 4% paraformaldehyde (Nacalai Tesque, Kyoto, Japan) for hematoxylin-eosin (HE) staining, while the other three were kept in the SEM fixative solution (glutaraldehyde 0.5% (Chemcruz, Dallas, Texas), paraformaldehyde 1.5% (Nacalai Tesque Kyoto, Japan), Hepes (Chemcruz, Dallas, Texas), and PBS working (Nacalai Tesque, Kyoto, Japan) for at least 6–8 hours. Scanning electron microscopyThree tongues were extracted from the SEM fixative solution (glutaraldehyde 0.5% (Chemcruz, Dallas, Texas), paraformaldehyde 1.5% (Nacalai Tesque, Kyoto, Japan), Hepes (Chemcruz, Dallas, Texas), and PBS working (Nacalai Tesque, Kyoto, Japan). The samples were dehydrated with various ethanol solutions (KgaA, Darmstadt, Germany). The tongues were placed on a plate made of metal. The Buehler 1,000 Vacuum System from Stuttgart, Germany, was used to vacuum dry the samples at 25°C and 4 Pa of pressure. The JEOL JEC-3000FC from Tokyo, Japan, was then used to coat them with platinum. The SEM equipped with a Tokyo, Japan-made JEOL-JSM6510LA type was used to examine the specimens. The accelerating current used by the SEM was 15 kV. HE stainingThe samples were placed in a tissue cassette and subjected to the washing process. The samples were then immersed and embedded in paraffin (Leica Biosystems, Wetzlar, Germany) after being dehydrated and cleaned using xylene (KgaA, Darmstadt, Germany). Ultimately, they were incubated for 24 hours at a temperature between 58°C and 60°C. A rotary microtome (Yamato RV 240, Asaka, Japan) was used to segment samples with a thickness of 8 µm. The samples were subsequently placed into gelatine-coated slides (KgaA, Darmstadt, Germany). HE (Bio-Optica, Milan, Spain) was then used as part of a conventional histology process to stain the samples. An LM (Olympus BX51, Tokyo, Japan) was used for the inspections. Optilab software was used to take photographic micrographs of the materials (Optilab, Yogyakarta, Indonesia). ResultsMacroscopic analysisGonocephalus chamaeleontinus has a fleshy, triangle-shaped tongue, that is short, widening, and thickening posteriorly. It is yellowish white at the A, while blue-black at the C to the R. The length of the tongue reaches 1.5 ± 0.5 cm and the width of the R is 1 ± 0.3 cm. The tongue is macroscopically divided into three regions from anterior to posterior, namely A, C, and R. The A region of the tongue of G. chamaeleontinus starts from the most anterior part toward the posterior through a cushion-like formation to the indentation that separates the A and C regions. The tip of the tongue appears rounded; a bifurcated tongue is found with a very small size. The sample has a slightly bent tongue tip toward the ventral so that the bifurcation is difficult to see. The C region is after the curve of the A and C region boundaries posteriorly to the highest visible formation on the tongue. The downward curve from the C to the posterior part is the R region. Between the two branches of the R region is the glottis. Macroscopic observation of the tongue can also identify the trachea, as well as the hyobranchial apparatus consisting of ceratohyal, ceratobranchial I, and ceratobranchial II, each of which has a pair of ceratobranchs (Fig. 1). Scanning electron microscopyApex The observations of the dorsal surface of G. chamaeleontinus tongue in the A region by using an SEM with a magnification of 30 times showed bifurcatio tongue (BT) and papillae lingualis (Fig. 2a). Bifurcatio tongue (BT) at the anterior A on observation with 100 times magnification has a small size and does not divide the A of the tongue deeply (Fig. 2b). In addition to the bifurcate tongue (BT), it is also seen that the lingual papillae on the anterior A are elongated so that between the papillae it forms a gap called the interpapillary groove. At the posterior A with a magnification of 100 times, the dome-shaped papillae (DP) are crowded together (Fig. 2c), more clearly visible along with the taste pores (TP) in the observation with a magnification of 400 times (Fig. 2d). DP have a polygonal shape with each papillae having a different size (Fig. 2c). Fig. 1. Macroscopic view of G. chamaeleontinus tongue (a) in dorsal section (A=apex region, C=corpus region, R=radix region, BT=bifurcatio tongue, GL=glottis, Tr=trachea, CB I=ceratobranchial I, CB II=ceratobranchial II, CH=ceratohyal); (b) in lateral section (BT=bifurcatio tongue).
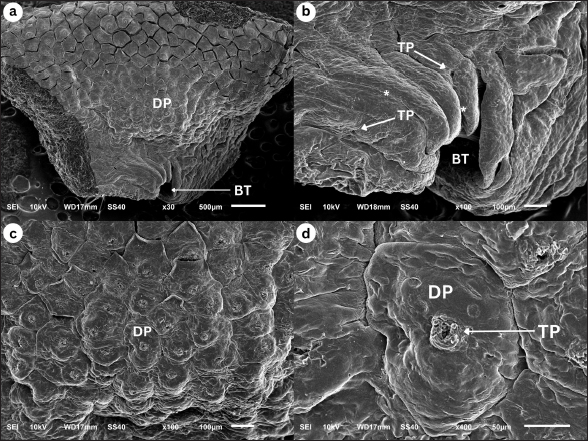
Fig. 2. SEM image of G. chamaeleontinus tongue at the A. (a) whole A with 30 times magnification; (b) the anterior tip of the tongue shows bifurcatio tongue (BT), TP, and interpapillary groove (*) with 100 times magnification; (c) DP are polygonal with 100 times magnification; (d) at the superficial papillae there are TP with 400 times magnification.
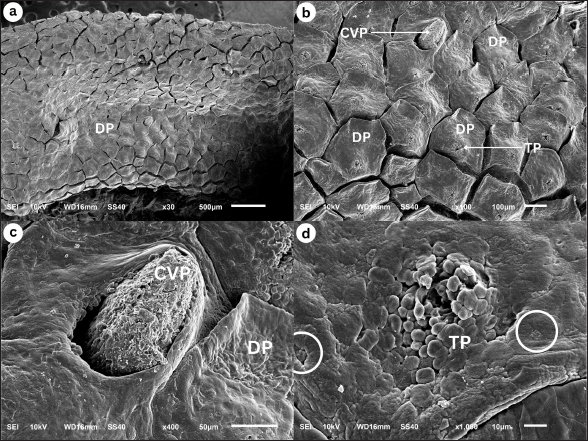
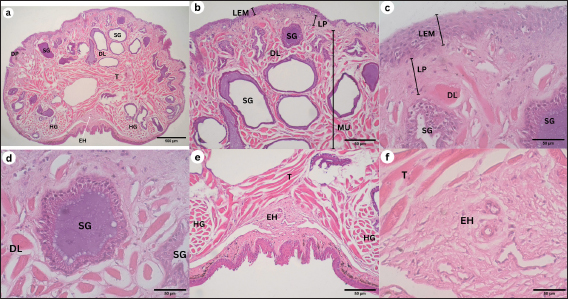
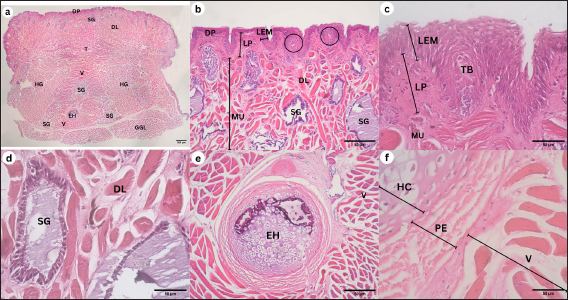
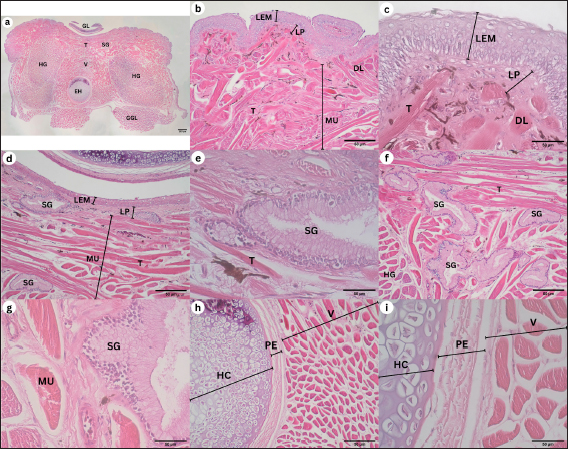
Corpus The observation of the dorsal surface of G. chamaeleontinus tongue in the C region using an SEM with a magnification of 30 times shows the distribution of lingual papillae (Fig. 3a). The observations with 100 times magnification show that the tongue is composed of DP lingual and TP which are scattered on each papilla (Fig. 3b). Papillae can be seen more clearly on observation with a magnification of 1000 times (Fig. 3d). There are papillae that are thought to be circumvallate papillae (CVP) at 400 times magnification (Fig. 3c). Radix The observation of the dorsal surface of G. chamaeleontinus tongue in the R region using an SEM with a magnification of 30 times shows the presence of lingual papillae that have scale-like papillae shape (SP) (Fig. 4a) and CVP. On observation with a magnification of 100 times on the R, not all papillae have TP as at the A and C (Fig. 4b). Microanatomy analysisApex The observation of the histological structure of G. chamaeleontinus tongue with HE staining at 4x magnification, scale bar 500 μm, shows the tongue at the A of the lingual papillae with a DP that has a short and wide stalk (Fig. 5a). Papillae at the A are crowded together so that the surface of the papillae looks slightly convex. The tongue A is composed of two tunicae, namely tunica mucosa and tunica muscularis (MU), clearly visible at 10x magnification, scale bar 50 μm (Fig. 5b). Tunica mucosa composed of lamina epithelialis mucosa (LEM) and lamina propria mucosa (LP) can be distinguished at 40x magnification, scale bar 50 μm (Fig. 5c). The LEM is a thick stratified squamous epithelium while the LP mucosa is composed of loose connective tissue layer situated beneath the epithelium of the tongue. The LP is richly vascularized, containing numerous blood vessels that supply nutrients and oxygen to the epithelial cells. The tunica MU consists of both intrinsic and extrinsic muscles, in the most dorsal part of the tunica MU, presence the m. dorsal longitudinalis (DL), following m. transversalis (T), and m. hyoglosus (HG) in the ventral part (Fig. 5a). There are lingual salivary glands (SG) distributed almost throughout the mucosal LP to the tunica MU A (Fig. 5a–d). Lingual SG have a stratified cuboidal epithelium seen at 40x magnification, scale bar 50 μm (Fig. 5d). On the ventral part of the tongue, there is a very small entoglossal process of hyoid (EH) seen at 10x magnification, scale bar 50 μm (Fig. 5e) and more clearly at 40x magnification, scale bar 50 μm (Fig. 5f). Fig. 3. SEM image of G. chamaeleontinus tongue at the C. (a) whole C with 30 times magnification; (b) polygonal DP with 100 times magnification; (c) on the superficial papillae there are CVP with 400 times magnification; and (d) and (TP and white circles) with 1000 times magnification.
Corpus The observations of the histological structure of G. chamaeleontinus tongue with HE staining showed that the tongue in the C section with a magnification of 4x, scale bar 500 μm has lingual papillae with a dome shape (Fig. 6a). The tongue consists of tunica mucosa and tunica MU. Tunica mucosa is composed of LEM and LP mucosa. The lamina epithelialis is a thick stratified squamous epithelium and taste buds (TB) were found in the lamina epithelialis at 10x magnification, scale bar 50 μm (Fig. 6b), more clearly seen at 40x magnification, scale bar 50 μm (Fig. 6c). The LP of the mucosa is composed of connective tissue. The tunica MU is composed of m. DL at the most dorsal part of the tunica MU, m. T, m. verticalis (V), m. HG, and m. genioglosus lateralis (GGL) on the ventral to lateral part of the tongue (Fig. 6a). Lingual SG were found scattered dorsally to laterally on the LP mucosa C and m. V (Fig. 6a and b), more clearly visible at 40x magnification, scale bar 50 μm (Fig. 6d). On the ventral part of the tongue, the EH is larger than the EH at the A at 10x magnification, scale bar 50 μm (Fig. 6e). EH at 40x magnification, scale bar 50 μm consists of hyaline cartilage (HC) enveloped by perichondrium (PE) and is in the ventral part of m. V, between m. HG (Fig. 6f). Radix The results of observations of the histological structure of G. chamaeleontinus tongue with HE staining showed that the tongue in the R section of 4x magnification, scale bar 500 μm was thick and had lingual papillae with a scale-like shape (Fig. 7a). The tongue consists of two tunicae, namely tunica mucosa and tunica MU. The tunica mucosa is composed of LEM and LP mucosa with magnification 10x, scale bar 50 μm (Fig. 7b and d). The LEM is a thick stratified squamous epithelium with 40x magnification, scale bar 50 μm (Fig. 7c), while the LP mucosa is composed of connective tissue. Tunica MU is composed of a few m. DL on the dorsal part, m. T, m. V, m. HG, and m. GGL on the ventral to the lateral part of the tongue (Fig. 7a). Lingual SG with 10x magnification, scale bar 50 μm were slightly present dorsally (Fig. 7d and f) and visible stratified cuboidal epithelium with 40x magnification, scale bar 50 μm (Fig. 7e and g). Between the m. HG, there is m. V which ventrally contains the EH (Fig. 7a) which is larger than the EH at the A and C (Fig. 7h). The EH at 10x magnification, scale bar 50 μm consists of HC enveloped by PE (Fig. 7h); the arrangement is more clearly visible at 40x magnification, scale bar 50 μm (Fig. 7i). Fig. 4. SEM image of G. chamaeleontinus tongue at the R. (a) whole R with 30 times magnification; (b) SP with 100 times magnification, not all papillae on the R found TP (arrow).
Fig. 5. Histological structure of the tongue of G. chamaeleontinus with HE staining of the A. (a) whole A visible DP; (b) distribution of lingual SG in the mucosal LP to the tunica MU; (c) tunica mucosa composed of LEM and mucosal LP; (d) lingual SG between the m. DL; (e) the ventral A shows m. T, two bundles of m. HG; and (f) entoglossal process of hyoid (EH).
DiscussionFig. 6. Histological structure of the tongue of a G. chamaeleontinus with HE staining of the C. (a) whole C visible tunica MU consists of m. DL, m. T, m. verticalis (V), two bundles of m. HG, and m. GGL ventrally; (b) C dorsally showing DP, lingual SG distribution, LEM, mucosal LP, and tunica MU; (c) TB on LEM; (d) lingual SG between the m. DL; (e) entoglossal process of hyoid (EH) composed of (f) HC enveloped by PE.
Fig. 7. Histological structure of the tongue of G. chamaeleontinus with HE staining of the R. (a) whole R visible tunica MU consisting of m. T, m. verticalis (V), two bundles of m. HG, and m. GGL in the ventral; (b) R dorsolateral part showed LEM, lamina LP, and tunica MU; (c) little m. DL; (d) R dorsal midline found (e) lingual SG; (f,g) lingual SG R located between m. T; (h) entoglossal process of hyoid (EH) composed of (i) HC enveloped by PE.
Macroscopic observation of the tongue of G. chamaeleontinus showed a triangular tongue with a rounded anterior end, short, thick, and widened posteriorly, similar to other iguanian lizards and gekkotan such as Gonocephalus sp. (Schwenk, 2000), Eublepharis macularius (Jamniczky et al., 2009), Pogona vitticeps (Zghikh et al., 2014), Tarentola annularis, and Uromastyx aegypticus (Bayoumi et al., 2011), which are also triangular and widen posteriorly. The tongue is divided into three parts: A, C, and R. The bifurcatio tongue is a special formation of the reptile tongue at the A with a cleavage at the anterior end of the tongue. In G. chamaeleontinus, the tongue has a bifurcation that is not as deep as that found in reptiles such as the snake Elaphe climacophora (Iwasaki et al., 1996) and the monitor lizard Varanus salvator (Briggs et al., 2022), which have a deep bifurcation that splits the A of the tongue. The small tongue bifurcation of G. chamaeleontinus is similar to that of other iguanian lizards and gekkotan such as Gonocephalus sp. (Schwenk, 2000), E. macularius (Jamniczky et al., 2009), T. annularis, and U. aegypticus (Bayoumi et al., 2011), which have small bifurcation. The deep bifurcation on the tongues of snakes and monitor lizards is a form of adaptation to their ability to detect signals from each side of the body (Baeckens et al., 2017), while the non-deep bifurcation, as in iguana, functions in the feeding process to help swallow prey without any process in the oral cavity (Bayoumi et al., 2011). In addition to the anterior part of the tongue, bifurcation is also seen in the posterior part of the tongue, namely in the R. The tongue on the R divides into two parts on the left and right sides of the larynx, which then the posterior limb (Schwenk, 2000). Tongue morphology is more clearly visible on observation using an SEM (Sheren et al., 2018). On the tongue, lingual papillae were found to scatter on the dorsal surface of the tongue; the distribution of lingual papillae was similar to P. vitticeps (Zghikh et al., 2014). Observation with high magnification shows the rough dorsal surface of the tongue with the A of the papillae pointing posteriorly. At the A and C, found DP. These papillae are wide, densely arranged, and short polygonal in shape. DP are also found on the tongue of T. annularis (Bayoumi et al., 2011). In addition, the R showed a SP shape as found in the tongue of Gekko japonicus (Iwasaki, 1990). TP were found on the tongue papillae of G. chamaeleontinus on the superficial part of the papillae and have a unique shape and appear similar to those found in Crocodylus niloticus (El-Sayyad et al., 2011). In the C, TP were also found similar to those found in Phrynosoma platyrhinos (Schwenk, 1985). The discovery of TB on the tongue A, C, and R was also found in other agamid reptiles such as Agama sp., Uromastyx hardwickii, and Gonocephalus grandis (Schwenk, 1985). TB have function of receiving chemical and mechanical information on food (Taha, 2013), which means they help in sensing food and assist transport in the process of swallowing prey. The results of histological observations of the tongue of G. chamaeleontinus starting from the A, C to the R have almost the same structure consisting of tunica mucosa and tunica MU. Tunica mucosa is composed of LEM and LP mucosa. The outermost layer of the tongue is the LEM, which is composed of stratified squamous epithelium. LP mucosa is a layer under the LEM, which is composed of connective tissue. Tunica MU is a layer of several types of muscles from the ventral part. The overall histological structure of the G. chamaeleontinus tongue is similar to that of T. annularis (Bayoumi et al., 2011). The LEM of the tongue from the A to the R is composed of thick stratified squamous epithelium similar to the tongue of Liolaemus monticola (Schwenk, 1985), which has a thick LEM. The mucosal LP consists of loose connective tissue. Lingual SG are found from the A, C, and R scattered on the LP mucosa to the tunica MU. SG are most commonly found at the A. The tunica MU at the A of the tongue of G. chamaeleontinus consists of m. DL at the most dorsal part of the tunica MU, m. T, and two bundles of m. HG on the left and right ventral parts, while the tunica MU at the C and R is composed of m. DL in the most dorsal part of the tunica MU, m. T, m. V in the center of the tongue, m. HG between m. V, and m. GGL in the ventral to lateral part of the tongue. Musculus DL increasingly posterior will be found less and less. The muscle arrangement found in the tongue of G. chamaeleontinus is similar to that found in Phrynosoma mcallii (Schwenk, 2021). Observation on the ventral part of the tongue is the EH or lingual processus in the middle of m. V, between m. HG. EH consists of HC enveloped by PE. The EH at the R is larger than the EH at the A and C as found in P. mcallii (Schwenk, 2021). The m. DL at the most dorsal part of the tunica MU and m. T work together to alter the shape and position of the tongue and responsible for the fine movements of the tongue, allowing for precise control during feeding (Herrel et al., 2001). Meanwhile, the genioglossus and hyoglossus, anchor the tongue to the hyoid apparatus and allow for broader movements, such as protrusion and retraction (Herrel et al., 2001). Papillae lingualis at the A and C are DP that histologically have short and wide stalks. TBs were found histologically on the dorsal and lateral surfaces of the C papillae wall, such as T. annularis (Bayoumi et al., 2011), which has DP and found TB on the dorsal and lateral surfaces of DP the papillae wall. The role of TB on the tongue is to test the palatability of prey when it comes into contact with the tongue (Bayoumi et al., 2011). When capturing prey, the tongue and larynx will move forward along with the head which also advances rapidly. The tongue can move forward because of the hyobranchial protraction that supports it. Hyobranchial protraction with the pull of the laryngohyoid ligament. Tongue protraction is accompanied by protrusion of the tongue whose anterior end will be curved ventrally so that the dorsal surface and papillae of the tongue can lead to prey (Schwenk, 2000). Lingual SG found in histological observations of the A, C, and R of the tongue of G. chamaeleontinus have an important role in feeding patterns. The sticky mucus produced by lingual SG will help in capturing prey so that prey can stick to the tongue (Meyers et al., 2002), as well as helping in the process of swallowing and digesting prey. The tongue of G. chamaeleontinus known “fleshy” because it is short, thick, wide, with rounded edges. The tongue is also known “sticky” because it is rich in sticky saliva, especially at the A where prey sticks to the dorsal surface of the tongue. The tongue is not elongated or ejected because it is not equipped with an accelerator m. that envelops the entoglossal processus of the hyoid, but the hyobranchial apparatus helps the tongue to extend and catch prey. The tongue of G. chamaeleontinus has a unique shape with some formations that are slightly different from other reptiles. However, with this uniqueness, its morphology and structure have important functions for its survival. The application of SEM and histological techniques to study the tongue of G. chamaeleontinus significantly enhances our understanding of the species’ physiology and behavior. These methodologies provide detailed insights into the structural adaptations of the tongue, which are closely linked to feeding strategies, sensory functions, and ecological interactions. The SEM allows for the visualization of the surface morphology of the tongue at a microscopic level, revealing the arrangement and types of papillae present. In reptiles, including G. chamaeleontinus, the tongue’s surface is typically covered with various types of lingual papillae, which serve multiple functions such as prey capture, manipulation, and sensory perception Crole and Soley (2010), Beisser et al., (2004). For instance, the presence of conical or filiform papillae, as observed in other lizard species, can enhance the mechanical grip on prey, facilitating effective feeding (Martins et al., 2014). This morphological adaptation is crucial for an insectivorous diet, as it allows the lizard to capture and hold onto fast-moving insects efficiently. Histological analysis complements SEM findings by providing insights into the tissue organization and cellular composition of the tongue. The histological structure of the tongue can reveal the presence of specialized glands, such as mucous and serous glands, which play a vital role in lubrication and digestion. For G. chamaeleontinus, the distribution and density of these glands can indicate how the species has adapted to its dietary needs and feeding behaviors. For example, a higher density of mucous glands may correlate with a diet that requires significant moisture, while serous glands may be more prevalent in species that consume drier food items (Igado et al., 2021). Furthermore, the histological examination can shed light on the sensory capabilities of the tongue. The presence of TB and their distribution across the tongue surface can provide insights into the species’ foraging behavior and dietary preferences (Martins et al., 2014). In reptiles, the arrangement of TB is often correlated with feeding habits, suggesting that G. chamaeleontinus may have evolved specific adaptations to enhance its ability to detect chemical cues in its environment, which is essential for locating prey (Singh et al., 2000; Martins et al., 2014). ConclusionThis study presents the initial comprehensive data on the papillae of G. chamaeleontinus tongue from Yogyakarta, Indonesia with SEM and LM. The use of SEM and histological techniques to study the tongue of G. chamaeleontinus provides a comprehensive understanding of its physiological and behavioral adaptations. These methods reveal critical information about the structural features of the tongue that facilitate feeding of the animal. Overall, the tongue papillae of G. chamaeleontinus consist of sensory and mechanical papillae accompanied by the presence of mucosal and seromucosal SG. AcknowledgmentsThe authors would like to thank to the Integrated Laboratory for Research and Testing, Universitas Gadjah Mada, for providing the facilities for this study. This study was supported by Thesis Recognition Grant 2024 (Rekognisi Tugas Akhir-RTA 2024) with grant number 5286/UN1.P1/PT.01.03/2024. Conflict of interestThe authors declare that there is no conflict of interest. Authors’ contributionsConceptualization, D.L.K.; Methodology, D.L.K. and S.K.; Software, A.D.T; Validation, D.L.K. and A.D.T.; Formal analysis, D.L.K., A.D.T. and S.K.; Investigation, A.D.T.; Resources, D.L.K.; Data Curation, ADT; Writing—Original Draft Preparation, A.D.T, S.K., and D.L.K.; Writing—Review and Editing, D.L.K, S.K., and A.D.T; Visualization, A.D.T; Supervision, D.L.K.; Funding Acquisition, D.L.K. All authors have read and agreed to the published version of the manuscript. Data availabilityAll data supporting the findings of this study are available within the manuscript. ReferencesAbbate, F., Latella, G., Montalbano, G., Guerrera, M.C., Germanà, G. and Levanti, M. 2009. The lingual dorsal surface of the blue-tongue skink (Tiliqua scincoides). Anat. Histol. Embryol. 38(5), 348–350. Abbate, F., Latella, G., Montalbano, G., Guerrera, M.C., Levanti, M. and Ciriaco, E. 2008. Scanning electron microscopical study of the lingual epithelium of green iguana (Iguana iguana). Anat. Histol. Embryol. 37(4), 314–316. Anjani, A., Saragih, G., Wihadmadyatami, H. and Kusindarta, D. 2023. Lingual morphology of domesticated asian small-clawed otters in Yogyakarta, Indonesia. Vet. Med. 68(3), 91–105. Baeckens, S., Herrel, A., Broeckhoven, C., Vasilopoulou-Kampitsi, M., Huyghe, K., Goyens, J. and van Damme, R. 2017. Evolutionary morphology of the lizard chemosensory system. Sci. Rep. 7(1), 1–13. Bayoumi, S.S., Abd-Elhameed, A.E. and Mohamed, E.S.M. 2011. Comparative studies on the dorsal lingual surface of two Egyptian squamate reptile with two different feeding habits. Egypt. J. Exp. Biol. (Zool.). 7(2), 203–211. Beisser, C.J., Lemell, P. and Weisgram, J. 2004. The dorsal lingual epithelium of Rhinoclemmys pulcherrima incisa (chelonia, cryptodira). Anat. Rec. A. Discov. Mol. Cell. Evol. Biol. 277A(1), 227–235. Belagalla, N. 2024. Eco-friendly and targeted through next-generation approaches to insect pest management. Uttar. Pradesh J. Zool. 45(13), 73–99. Briggs, V., Evans, P., Klovanish, C. and Mazzotti, F.J. 2022. A species bioprofile for the Asian water monitor (Varanus salvator). Southeastern Nat. 21(3), 187–210. Crole, M.R. and Soley, J.T. 2010. Surface morphology of the emu (Dromaius novaehollandiae) tongue. Anat. Histol. Embryol. 39(4), 355–365. Delheusy, V., Toubeau, G. and Bels, V. 1994. Tongue structure and function in Oplurus cuvieri (Reptilia: iguanidae). Anat. Rec. 238(2), 263–276. El-Mansi, A.A., Alkahtani, M.A., Abumandour, M.M.A. and Ahmed, A.E. 2020. Structural and functional characterization of the tongue and digestive tract of Psammophis sibilans (squamata, lamprophiidae): adaptive strategies for foraging and feeding behaviors. Microsc. Microanal. 26(3), 524–541. El-Sayyad, H.I.H., Sabry, D.A., Khalifa, S.A., Abo u-El-Naga, A.M. and Foda, Y.A. 2011. Estudios sobre la lengua de las especies de reptiles Psammophis sibilans, Tarentola annularis y Crocodylus niloticus. Intl J. Morphol. 29(4), 1139–1147. Harlow, H., Purwandana, D., Jessop, T. and Phillips, J. 2010. Body temperature and thermoregulation of komodo dragons in the field. Therm. Biol. 35(7), 338–347. Herrel, A., Canbek, M., Özelmas, Ü., Uyanoğlu, M. and Karakaya, M. 2005. Comparative functional analysis of the hyolingual anatomy in lacertid lizards. The Anat. Rec. Part. A. Discov. Mol. Cell. Evol. Biol. 284A(2), 561–573. Herrel, A., Meyers, J.J., Nishikawa, K.C. and Vree, F.D. 2001. Morphology and histochemistry of the hyolingual apparatus in chameleons. J. Morphol. 249(2), 154–170. Howard, S. 2018. Keeping the java hump-headed lizard. Reptiles Magazine. Igado, O.O., Adebayo, A.O., Oriji, C., Aina, O.O. and Oke, B.O. 2021. Macroscopic and microscopic analysis of the tongue of the greater cane rat (Thryonomys swinderianus, temminck). J. Morphol. Sci. 38, 9–15. Iwasaki, S. 1990. Fine structure of the dorsal lingual epithelium of the lizard, Gekko japonicus (Lacertilia, Gekkonidae). Am. J. Anat. 187(1), 12–20. Iwasaki, S.I., Yoshizawa, H. and Kawahara, I. 1996. Three-dimensional ultrasctructure of the surface of the tongue of the rat snake, Elaphe climacophora. Anat. Rec. 245(1), 9–12. Jamniczky, H.A., Russell, A.P., Johnson, M.K., Montuelle, S.J. and Bels, V.L. 2009. Morphology and histology of the tongue and oral chamber of Eublepharis macularius (Squamata: Gekkonidae), with special reference to the foretongue and its role in fluid uptake and transport. Evol. Biol. 36(4), 397–406. Martins, D.M., Pinheiro, L.L., Ferreira, V.C., Costa, A.M., Lima, A.R., Ricci, R.E.G., Miglino, M.A. and Branco, É. 2014. Tongue papillae morphology of brown-throated sloth Bradypus variegatus (Schinz, 1825). Arq. Bras. Med. Vet. Zootec. 66(5), 1479–1486. Meyers, J.J., Herrel, A. and Nishikawa, K.C. 2002. Comparative study of the innervation patterns of the hyobranchial musculature in three iguanian lizards: Sceloporus undulates, Pseudotrapelus sinaitus, and Chamaeleo jacksonii. J. Exp. Biol. 267(2), 178–189. Schwenk, K. 1985. Occurrence, distribution and functional significance of taste buds in lizards. Copei. 1985(1), 91–101. Schwenk, K. 2000. Feeding: form, function, and evolution in tetrapod vertebrates. Elsevier. Schwenk, K. 2021. Tongue morphology in horned lizards (Phrynosomatidae: Phrynosoma) and its relationship to specialized feeding and diet. Russ. J. Herpetol. 28(5), 309–317. Selan, Y., Wihadmadyatami, H., Haryanto, A. and Kusindarta, D. 2023. The tongue morphology of Pteropus vampyrus from timor island, Indonesia: new insights from scanning electron and light microscopic studies. Biodiversitas 24(6), 3512–3518. Sheren, A.Z.A., Nasr, E.S. and Hassan, S.S. 2018. Light and scanning electron microscopic observations on the tongue of Nile monitor, Varanus niloticus. Int. J. Adv. Res. Biol. Sci. 5(4), 1–11. Singh, A., Philpott, J.M., Patel, N. and Mochloulis, G. 2000. Proliferative myositis arising in the tongue. J. Laryngol. Otol. 114(12), 978–979. Taha, A. 2013. Comparative anatomical, histological and histochemical study of tongue in two species of insectivorous vertebrates. AJBAS 7(1), 401–410. Tiwari, A. 2024. IPM essentials: combining biology, ecology, and agriculture for sustainable pest control. J. Adv. Biol. 27(2), 39–47. Yang, C. and Wang, L. 2016. Histological and morphological observations on tongue of Scincella tsinlingensis (Reptilia, Squamata, Scincidae). Micron 80, 24–33. Zghikh, L.N., Vangysel, E., Nonclerq, D., Legtand, A., Blairon, B., Berri, C., Bordeau, T., Remy, C., Burtea, C., Montuelle, A.J. and Bels, V. 2014. Morphology and fibre-type distribution in the tongue of the Pogona vitticeps lizard (Iguania, Agamidae). J Anat. 225(4), 377–389. | ||
| How to Cite this Article |
| Pubmed Style Theqla AD, Karnati S, Kusindarta DL. Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopy. Open Vet. J.. 2025; 15(2): 1032-1042. doi:10.5455/OVJ.2025.v15.i2.51 Web Style Theqla AD, Karnati S, Kusindarta DL. Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopy. https://www.openveterinaryjournal.com/?mno=215304 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.51 AMA (American Medical Association) Style Theqla AD, Karnati S, Kusindarta DL. Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopy. Open Vet. J.. 2025; 15(2): 1032-1042. doi:10.5455/OVJ.2025.v15.i2.51 Vancouver/ICMJE Style Theqla AD, Karnati S, Kusindarta DL. Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopy. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 1032-1042. doi:10.5455/OVJ.2025.v15.i2.51 Harvard Style Theqla, A. D., Karnati, . S. & Kusindarta, . D. L. (2025) Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopy. Open Vet. J., 15 (2), 1032-1042. doi:10.5455/OVJ.2025.v15.i2.51 Turabian Style Theqla, Amanda Dominica, Srikanth Karnati, and Dwi Liliek Kusindarta. 2025. Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopy. Open Veterinary Journal, 15 (2), 1032-1042. doi:10.5455/OVJ.2025.v15.i2.51 Chicago Style Theqla, Amanda Dominica, Srikanth Karnati, and Dwi Liliek Kusindarta. "Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopy." Open Veterinary Journal 15 (2025), 1032-1042. doi:10.5455/OVJ.2025.v15.i2.51 MLA (The Modern Language Association) Style Theqla, Amanda Dominica, Srikanth Karnati, and Dwi Liliek Kusindarta. "Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopy." Open Veterinary Journal 15.2 (2025), 1032-1042. Print. doi:10.5455/OVJ.2025.v15.i2.51 APA (American Psychological Association) Style Theqla, A. D., Karnati, . S. & Kusindarta, . D. L. (2025) Detailed morphological study of the tongue of forest dragon (Gonocephalus chamaeleontinus) by scanning electron and light microscopy. Open Veterinary Journal, 15 (2), 1032-1042. doi:10.5455/OVJ.2025.v15.i2.51 |