| Review Article | ||
Open Vet. J.. 2025; 15(2): 482-503 Open Veterinary Journal, (2025), Vol. 15(2): 482-503 Review Article Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancerUlayatul Kustiati1,2, Dwi Aris Agung Nugrahaningsih3, Dwi Liliek Kusindarta4 and Hevi Wihadmadyatami4*1Post-Graduate School of Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 2Laboratory of Pharmacology, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia 3Department of Pharmacology and Therapy, Faculty of Medicine, Public Health and Nursing, Universitas Gadjah Mada, Yogyakarta, Indonesia 4Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia *Corresponding Author: Hevi Wihadmadyatami. Department of Anatomy, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia. heviwihadmadyatami [at] ugm.ac.id Submitted: 29/08/2024 Accepted: 03/12/2024 Published: 28/02/2025 © 2025 Open Veterinary Journal
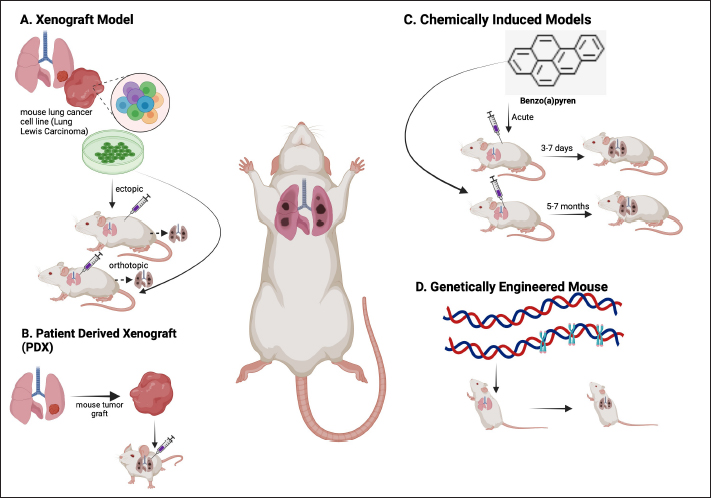
AbstractLung cancer is the leading cause of fatalities related to cancer globally. There are numerous ways to treat lung cancer, including surgery, chemotherapy, and radiation. Since these treatments have not yet shown satisfactory results, more research into the underlying mechanisms and different approaches to therapy and prevention are needed. Animal models are essential to the study of lung cancer because they offer priceless information about the etiology, course, and possible treatments for the illness. The therapeutic application of phytochemicals and medicinal plants to treat cancer-related compounds has gained attention subsequently. In addition to discussing the molecular carcinogenic and antitumor effects of the herbal treatment Ocimum sanctum (OS) in connection to lung cancer, this review will address the current awareness regarding lung cancer in animal models. The multitude of animal models used in lung cancer research—such as genetically modified mice, carcinogen-induced models, and xenograft induction—provides a solid foundation for understanding the illness. By easing the examination of the environmental and genetic factors involved and enhancing the analysis of possibilities for treatment, these models eventually assist in the further development of lung cancer therapy. Additionally, using the herb plant OS is essential for both treating and preventing lung cancer. Standardizing dosages and enforcing laws on the use of herbal medications require more in-depth investigation. Keywords: Animal model, Carcinogenesis mechanism, Lung cancer, Ocimum sanctum IntroductionCancer has been a major global public health concern in recent times. According to the National Cancer Institute, cancer is the abnormal growth of newly formed cells that surpass normal limits and then spread to other organs and the other side of the body (Brown et al., 2023). Oncogene activation and/or tumor suppressor gene inactivation often cause uncontrolled cell proliferation and the inhibition of the mechanism responsible for programmed cell death (Siegel et al., 2015). Lung cancer, frequently referred to as lung carcinoma, is a kind of malignant tumor that arises in the lung tissue as a result of an uncontrolled proliferation of cells (Mustafa et al., 2016). Approximately 9.6 million individuals worldwide succumbed to cancer in 2018. Curiously, genetic variables account for about 5%–10% of these cases, but lifestyle and environment have a significantly greater impact, making up 90%–95% (Anand et al., 2008). Research findings from 2012 indicated that Asia has the highest incidence of lung cancer. The World Health Organization reports that Asia has more cases than every other region, with a rise of more than 51.4%. Breast cancer, stomach cancer, liver cancer, and colon cancer are the most common cancer kinds in Asia. Lung cancer is the sixth among the top causes of death for women in Indonesia and has the highest mortality rate among men. Indonesia has the third highest rate of lung cancer, accounting for 8.6% of new cases and causing the highest number of deaths, which represents 12% of all cancer-related deaths (Arfan and Andarini, 2022). The elevated prevalence can likely be attributed to a multitude of unfavorable lifestyle factors, including smoking, inadequate dietary habits, air pollution, and insufficient public consciousness on early cancer detection. Smoking is responsible for almost 90% of lung cancer cases in Indonesia, making it the primary cause of cancer-related deaths in the country (Mustafa et al., 2016; Kristina et al., 2019). Unlike in humans, lung cancer is uncommon in pet animals (dogs and cats) because of restrictions on occurrence rates and prognosis. Animals suffering from lung cancer encounter difficulties in receiving therapy because of the presence of nonspecific clinical signs such as coughing, vomiting, difficulty breathing, fatigue, and vomiting. This leads to a lack of awareness of how lung cancer begins and develops in dogs and cats (Fowler et al., 2020). Presently, surgical procedures, biopsy, radiation, and chemotherapy are the cornerstones of cancer prevention and therapy for both people and animals (Miller and Zachary, 2017). The amount that the disease has spread is a crucial factor that impacts how effectively lung cancer treatments work. Surgical surgery is a successful treatment for malignancies that have not spread, but advanced-stage tumors present considerable therapeutic hurdles. In addition, cancer treatments are expensive and have side effects. Animal models are essential for the scientific investigation of lung cancer because they provide significant details on the biology, course, and possible therapies of the disease. By using these models, scientists gain insight into the complex interactions that occur between cancer cells and the tumor microenvironment, which is important to understand the dynamics of tumors and how well therapies work. The development of orthotopic lung cancer models, in which tumors are inserted directly into lung tissue, is one instance of this. In contrast with ectopic models, these kinds of models have been demonstrated to yield more realistic results that closely resemble lung cancer in humans. The development of animal models for lung cancer is important because it allows for an improved understanding of the complex process of carcinogenesis, precise evaluation of tumor growth and metastasis, and the efficacy of treatment medications in a setting that closely mimics the real disease environment. Fortunately, indigenous knowledge also gives us significant insights into the efficient use of natural resources. Nowadays, these materials are widely used as complementary therapies and prophylactics. Under medical supervision, they can also be used in addition to traditional therapies. As a popular herbal remedy in South and Southeast Asia, basil has a number of benefits, such as anti-inflammatory abilities, the ability to stimulate nerve cell proliferation, neuroprotection, and the ability to induce apoptosis in breast cancer (Kusindarta et al., 2016; Kusindarta et al., 2018; Kusindarta et al., 2018a; Raditya et al., 2020; Wihadmadyatami et al., 2023). The processes of molecular carcinogenesis, the development of animal models for the study of lung cancer, and the possible use of Ocimum sanctum (OS) as an herbal Ayurvedic medicine for lung cancer prevention and therapy will all be addressed in this review. MethodologyThe National Center for Biotechnology Information, PubMed, Science Direct, Google Scholar, Springer Link, and SAGE journal databases were all employed in a methodical and thorough evaluation of the online scientific literature. The research encompassed all citations from 2000 to 2022. The following keywords were used in the online search: OS, antioxidant properties, cytotoxicity, antitumor, anticancer, antitumor, antimicrobial, antibacterial, anti-inflammation, conventional ethnomedicine, and phytochemical substances, and pharmacological features. Lung cancerApproximately 13% of all cancer diagnoses are related to lung cancer, making it as one of the leading causes of mortality globally (Barta et al., 2019). Epigenetic alterations and hereditary DNA damage are the causes of lung cancer. Normal cell functions, such as DNA repair, programmed cell death (apoptosis), and cell growth, are impacted by these alterations. Lung cancer can be worsened or prevented by a number of risk factors, such as genetics, asbestos, air pollution, radon gas, and smoking (Larsen and Minna, 2011). The leading cause of cancer is still smoking (Yuan et al., 2009), and secondhand smoke exposure is a significant risk factor for lung cancer in both humans and animals (Brown et al., 2007; Kim et al., 2012). Lung cancer risk is raised through asbestos exposure in addition to air pollution, particularly from automobile emissions (Zhou et al., 2015). Lung cancer is a condition that is influenced by radon exposure (Fowler et al., 2020). Viral infections can lead to infectious lung cancer in sheep infected with the Jaagsiekte sheep retrovirus (Rai et al., 2000). Research is presently being done to determine whether retroviral infections contribute to lung cancer in humans (Miller et al., 2017). Lung cancer progresses in part due to chronic inflammation (Freeman et al., 2023). Proto-oncogenes, which encode growth-promoting proteins, and tumor suppressor genes, which encode molecules that negatively regulate cell proliferation, are the two categories of oncogenes that have been found. A protein with dominant behavioral effects can be produced by a mutation in one of the proto-oncogenic paired alleles. On the other hand, gene products that become cancerous cells require the activation of tumor suppressor genes before they may appear. Most patients with lung cancer have altered gene coding for proteins, which regulates or controls cell proliferation. Lung cancer etiology may be significantly influenced by these molecular alterations (Huang et al., 2021). Tumor suppressor gene inactivation or oncogene activation is the first step in the pathophysiology of lung cancer. Tumor invasion, angiogenesis, apoptosis, and cell proliferation are all regulated by the epidermal growth factor receptor (EGFR) (Mustafa et al., 2016). Lung cancer is classified histologically into two categories: non-small cell lung cancer (NSCLC) and small-cell lung cancer (SCLC). Small cells with minimal cytoplasm and no discernible nucleoli are the hallmarks of SCLC, which typically affects smokers. Because SCLC problems metastasis earlier and double more quickly, they are always diagnosed as systemic diseases. NSCLC comes in five different forms: the two most common histological subtypes of NSCLC are (1) squamous cell carcinoma, which develops in the central bronchi and is associated with smoking; (2) adenocarcinoma, which develops in the bronchioles and alveoli and is most common in women and non- smokers. (3) Large cell carcinoma, which arises from a lack of differentiation in small cells and glandular or squamous cells; (4) carcinoid tumors, which include two subtypes: typical and atypical (Travis et al., 2013; Siddiqua et al., 2022); and (5) adenosquamous carcinoma, which is defined as having more than 10% mixed glandular and squamous components—it has a worse prognosis than squamous carcinoma and adenocarcinoma. Adenocarcinoma usually develops from mucosal glands and represents approximately 40% of all lung cancer cases. The development of adenocarcinoma usually begins in the marginal area of the lung, and in many cases, it is found in scars or areas of chronic inflammation (Li and Lu, 2018), making it more difficult to attribute the cause to the bronchial wall. This cancer grows and spreads along the alveolar wall, using the alveolar wall as a starting point for growth (Travis et al., 2013). Cells from an adenocarcinoma may distribute locally, entering the pericardium, pleura, diaphragm, or bronchi. The spread will proceed to the mediastinum, big blood arteries, trachea, esophagus, spine, or neighboring lobes if problems from other disorders are present. Before spreading to the mediastinal or subcarinal lymph nodes and ultimately the contralateral lung, lymph node metastases start in the parabronchial lymph nodes. Extensions to the opposing lobe, pleural nodules, malignant pleural or pericardial effusions, and distant locations (liver, brain, or bones) are examples of remote metastases (Nana-Sinkam and Shojaee, 2017). EGFR mutations and oncogenic rearrangements of anaplastic lymphoma kinase fusion are present in a subgroup of NSCLCs and make them more sensitive to tyrosine kinase inhibitors (Shojaee and Nana-Sinkam, 2017). Thyroid transcription factor-1, cytokeratin-7, and napsin A are the traditional histochemical indicators of adenocarcinoma. There are three subtypes of pulmonary adenocarcinoma: acinar, papillary, and mixed. Adenocarcinoma can spread to the liver, bones, central nervous system, and adrenal glands, just like small cell carcinoma does. Adenocarcinoma is more likely to be confined than small cell carcinoma, particularly when it presents as peripheral lung nodules (Travis et al., 2013). Animal models of lung cancerAnimal models play a crucial role in lung cancer mechanism, prevention, and medication due to the intricate nature of oncology research and the ability of animal models to produce significant data with meaningful clinical implications, including disease processes, pharmacological treatments, and early detection approaches (Onaciu et al., 2020). Animal models are experimental animals that closely resemble human diseases (Li et al., 2021). Although in vitro studies offer valuable cellular insights, their conclusions could be more extensive in their capacity to capture the intricate pathogenic connections in vivo animals (Tratar et al., 2018). In vivo research using animal models is valuable for assessing possible treatments’ effectiveness, including traditional herbal remedies. Furthermore, the impact of signaling systems on lung cancer can also be demonstrated. One way to uncover biomarkers is by combining data from animal models with human clinical research (Domínguez-Oliva et al., 2023). External or exogenous factors, such as lifestyle and site of residence, communicate with internal factors, especially genetics, affecting the onset and progression of cancer (Rudolph et al., 2016). Therefore, integrating in vitro and in vivo studies yields more compelling findings (Tratar et al., 2018). A number of animal models have been developed to mimic lung cancer; four common approaches are used to generate lung cancer models in mice: the genetically modified mouse model, the chemically induced model, the cell line-derived xenograft (CDX) model, and the patient- derived xenograft (PDX) model (Fig. 1) (Onaciu et al., 2020). Using these models, scientists may study how lung cancer develops, evaluate cutting-edge therapeutic approaches, and investigate the effects of various treatments. The advantages and disadvantages of every method used to create the lung cancer model are listed in Table 1.
Fig. 1. The four most frequently used techniques to generate mouse lung cancer models include chemically induced models, PDX mode, CDX model, and genetically engineered mouse model. Table 1. Advantages and disadvantages of several methods to lung cancer animal model methods.
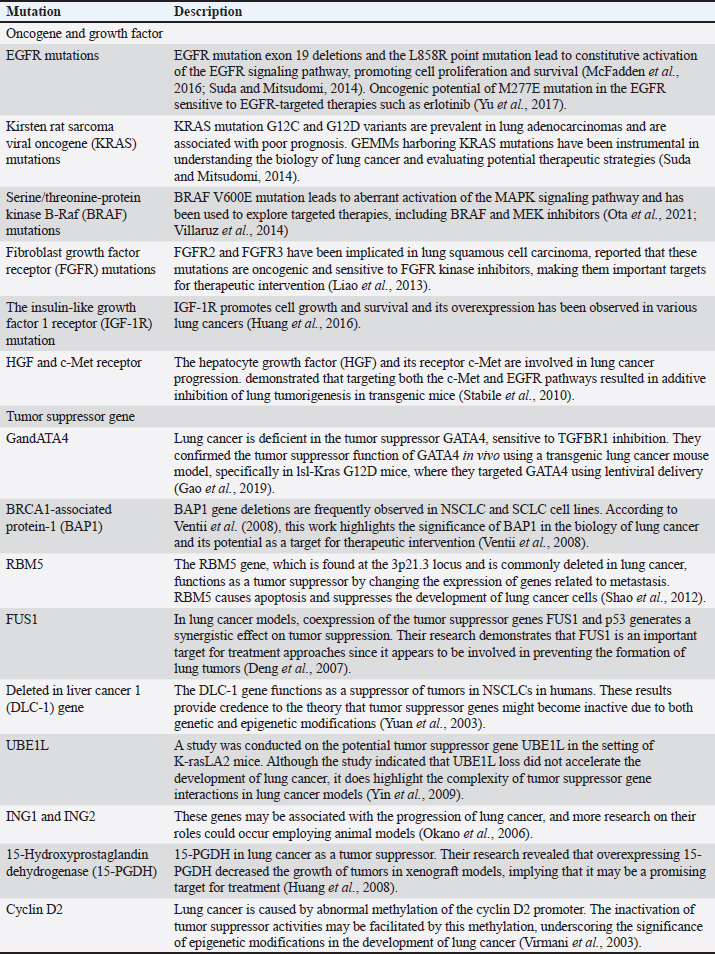
Chemically induced lung cancer animal modelChemically induced cancer animal models use carcinogens to transform normal cells into tumors. Chemically induced animal models have the advantage of mimicking the occurrence of human cancer from the beginning of the carcinogenic process (Liu and Johnston, 2002.) providing a valuable platform to study the pathogenesis, progression, and potential therapeutic interventions for lung cancer. However, the main drawback of this method is that it takes 30–50 weeks to form tumors after using carcinogens (Minicis et al., 2013). Various chemical substances have been employed in Several studies to induce lung carcinomas in animals through intraperitoneal injection such as N-nitroso diethylamine (NDEA) (Çiçek et al., 2022), 3-methylcholanthrene and diethylnitrosamine (DEN) (Zhu et al., 2017). In animal models of adenocarcinoma-type lung cancer, N-nitrosamines such as 4-(methylnitrosamino)-1-(3- pyridyl)-1-butanone (Brown et al., 2007) and DEN (Mervai et al., 2018) have been shown to selectively induce lung adenocarcinoma in animals so that the pathogenesis of lung cancer can be studied. In addition, chemical induction models using substances such as benzo(a)pyrene (BaP) have been developed to induce lung cancer in mice, providing insight into the effects of these carcinogens on lung tumourigenesis (Wang et al., 2021). Urethane, a chemical carcinogen present in tobacco smoke, has been shown to induce lung tumors in mice by causing activating mutations in Kras (Minowada and Miller, 2009). Cell lines for lung cancer induction in animal modelsA xenotransplantation paradigm in which cancer cell lines are injected into immunodeficient mice is known as the CDX model (Brennecke et al., 2014). For this aim, the NSCLC adenocarcinoma cell line A549 has been widely employed, created an orthotopic lung cancer animal model by implanting a variety of cells into BALB/c nude mice, demonstrating the adaptability and significance of the A549 cell line (Lee et al., 2010). Furthermore, a lung cancer animal model in nude mice has been established using the human large-cell lung cancer cell line H460 (Ye et al., 2016). Furthermore, the C57BL/6 mouse-derived murine lung cancer cell lines LLC and CMT167 are crucial for researching the tumor microenvironment and determining how sensitive lung cancers are to PD-1/programmed cell death ligand 1 (PD-L1) antibody blocking (Du et al., 2016). PDXs for lung cancerPDX models have become an invaluable tool in lung cancer research, providing a platform to study tumor biology, test therapeutic regimens, and identify predictive biomarkers. These models involve implanting patient-derived tumor tissue into immunocompromised mice to create xenografts that mimic the characteristics of native tumors, successfully created a broad panel of patient-derived tumor xenografts for NSCLC, despite the considerable time and distance between clinical and animal facilities, providing a stable and reliable preclinical model for human lung cancer research (Ilié et al., 2014). This PDX model recapitulates the histopathology and molecular diversity of NSCLC, offering a platform to study disease heterogeneity and test personalized treatment approaches. Furthermore, PDX models have been instrumental in predicting effective therapies for lung cancer, demonstrating the utility of PDX models in predicting effective heparanase-based therapies for lung cancer, highlighting the relevance of these models in guiding treatment strategies (Katz et al., 2018). Additionally, it emphasizes the clinical significance of PD-L1 in patients with lung cancer using PDX models, demonstrating the ability of these models to reproduce the biological characteristics of human cancers (Ma et al., 2016). Genetically engineered mouse model for lung cancerResearch on lung cancer has derived greatly from the use of genetically engineered mice models (GEMMs), which offer an invaluable platform for examining the development, spread, and effects of treatment. Animal models of genetically engineered lung cancer are produced by modifying (adding/removing) genes to produce desired functional alterations. Lung cancer that is both universal and unique to the pau will be the outcome of the change (Zhao et al., 2000). The KrasG12D/+ (Kirsten rat sarcoma viral oncogene homolog) mouse model of lung cancer is a well-known GEMM for the disease. It is a p53 Frt/Frt model in which conditional mutations in the p53 tumor suppressor gene and the K-ras oncogene cause adenocarcinoma to develop in the mouse lung (Barck et al., 2015). Utilization of GEMMs with conditional mutations in the K-ras oncogene and p53 tumor suppressor gene to induce lung tumors can exert circadian rhythm disruption effects on lung tumorigenesis (Papagiannakopoulos et al., 2016) and highlight histopathological changes using micro-CT imaging and automated analysis (Barck et al., 2015). Another method eliminates the SWI/SNF ATPase subunits BRM and BRG1 in promoting lung cancer development in GEMM. This study highlights the histopathological changes associated with lung cancer progression in GEMMs. Mutations in growth factors such as SP-C-IgEGF, SP-C-cMyc, and SpC-IgEGF will result in adenocarcinoma GEMM changes and provide insight into tumor evolution and progression (McFadden et al., 2016). Other GEMM studies can be seen in Table 2. Molecular carcinogenesis of lung cancerThe development of lung cancer involves a complex process known as molecular carcinogenesis, which includes genetic mutations, epigenetic changes, and interactions with the tumor microenvironment. Gaining a comprehensive understanding of these mechanisms is essential for the development of highly effective preventative and treatment strategies. Angiogenesis and vasculogenesisAngiogenesis is a common, complex physiological process that is managed by certain molecules that the body produces. Endogenous local or systemic biomolecular signals synchronize the actions of smooth muscle and endothelial cells to repair damaged blood vessels. Vascularization aids in the provision of nutrients in addition to serving as a pathway for neoplasm cell extravasation, or the movement of tumor cells to other organs through circulation (Rajabi and Mousa, 2017). The process of neoplasm cells generating blood vessels that invade and spread within the tumor environment is known as angiogenesis. Neoplasm cells require blood vessel development to survive since it provides them with nutrition and oxygen. Not only does angiogenesis encourage the growth and metastasis of neoplasm cells, but it is also a sign of cancer. Vasculogenesis, sprouting angiogenesis, intussusception, and vasculogenic mimicry are a few angiogenesis mechanisms (Liu et al., 2023). The genesis and progression of neovascularization are aided by endothelial progenitor cells or EPCs. Neoplasm cells provide proangiogenic substances, including vascular endothelial growth factor (VEGF) and cytokines, which subsequently attract dendritic cells coming from bone marrow and facilitate their proliferation and development. Endhotelial Progenitor Cells (EPC) mobilization merges into a single endothelium layer subsequent to matrix metalloprotease 9 (MMP 9) activation (Zuazo-Gaztelu and Casanovas, 2018). VEGF drives the development of EPCs into mature endothelial cells. Through a paracrine mechanism, EPCs also promote angiogenesis by expressing proangiogenic factors at the site of neovascularization of the tumor stroma or ischemic tissue (Marçola and Rodrigues, 2015; Urbich and Dimmeler, 2004). Table 2. Gene mutation related genetically engineered mouse model for lung cancer.
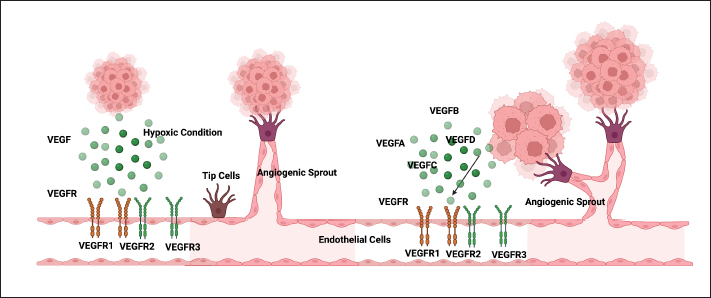
Sprouting angiogenesis is the first stage of vasculogenesis. Tumor cells, the microenvironment, and the extracellular matrix (ECM) are all involved in this pathway. Destabilization of endothelial-pericyte connections (maintaining the integrity and maintenance of blood vessels) is the first step in the sprouting angiogenesis stage. The mesenchymal-endothelial transition that endothelial cells go through improves their ability to migrate, invade, and proliferate. Mesenchymal-endothelial transition degrades the ECM and basement membrane with activated proteases (such as MMP), opening migration, and proliferation pathways. The lumen of blood vessels is formed by the polarization of endothelial cells (Urbich and Dimmeler, 2004; Lugano et al., 2020). This event provides a proangiogenic signal—the tip cell becomes invasive and motile. Endothelial cells respond to VEGF signals by expanding filopodia to form new blood vessels along the vascular bed. Endothelial cells in the sprouting angiogenesis stalk (stalk cells) have the potential to proliferate. The proliferative potential of stalk cells forms tubules, allowing expansion to form new blood vessels (Norton and Popel, 2016). The next step is intussusception microvascular growth. The intussusception microvascular growth mechanism begins with the division of the blood vessels into two new blood vessels, and a transvascular connective tissue column is formed within the lumen of the blood vessel, called a tissue pillar. Transendothelial cell bridges are formed subsequent to the induction of endothelial cells from the opposing wall. The endothelium layer forms as a result of the arrangement of the interendothelial connections. Interstitial pillars are created to reinforce the bridge, and mural cells are enlisted to line the newly created interstitial walls (Burri et al., 2004; Ali et al., 2019). There is vascularogenic mimicry. Tumor cell differentiation is the basis for this process. A matrix network like that of vasculogenesis is created by this process. Red blood cells and plasma make up this structure, which aids in blood circulation. Regardless of angiogenesis, endothelial cells that engage in vasculogenic mimicry can serve as a supplementary circulatory system for tumor cells. Vasculogenic mimicry is caused by low oxygen levels, the release of ECM components, and the activation of transmembrane Metalloproteinase (MP) (Luo et al., 2020). MMP, integrins, and VEGF are a few biomolecules involved in tumor cell angiogenesis. In addition to being the primary source of pathological angiogenesis, VEGF also causes inflammation, rheumatoid arthritis, diabetic retinopathy, and cancer (Mukhopadhyay et al., 2004). The Zn structure is essential to a class of endopeptidases known as MMPs. MMP plays a role in the disintegration of ECM macromolecules (Wang and Khalil, 2018). The role of VEGF in angiogenesisThe primary modulator of blood vessel development is homodimeric glycoprotein VEGF, sometimes referred to as VEGF-A. By starting the proteinase cascade that results in the creation of plasmin and MMP, VEGF generates feedback for VEGF activity (Liu et al., 2023). Some of the growth factors that may promote the synthesis of VEGF include platelet-derived growth factors (PDGF), fibroblast growth factors (FGF), epidermal growth factors, tumor necrosis factors (TNF), transforming growth factors, interleukin-1 (IL-1), and the hypoxic state of cells (Bao et al., 2009). Increased endothelial cell migration, increased endothelial cell mitosis, increased methane monooxygenase activity, increased αvβ3 activity, blood vessel lumen layout, and more are all related to VEGF’s function in angiogenesis, chemotaxis for granulocytes and macrophages, as well as indirect vasodilation via endothelial-derived relaxing factors or nitric oxide (NO) release (Lamalice et al., 2007). As a crucial modulator of normal physiological angiogenesis, VEGF expression abnormalities are a key marker of the illness process. Pathogenesis, which includes neoplasia and inflammatory diseases, is influenced by VEGF. Increased VEGF expression has been seen in neoplastic cell biopsies. In a variety of human cancer types, VEGF is known to be the starting point for carcinogenesis and disease development (Ceci et al., 2020). Neoplastic cells are essential for the formation of VEGF; without a sufficient vascular supply, they will not proliferate because they will not have enough oxygen and nutrients. As a key player in tumor angiogenesis, VEGF promotes the formation of new blood vessels from surrounding capillaries, giving tumors access to the nutrients and oxygen they require to proliferate (Lugano et al., 2020). The absence of adequate vascularization in neoplasm cells causes a hypoxic environment, which, in turn, triggers new vascularization. The tumor’s state also alters the microenvironment, resulting in elevated growth factor levels and neovascularization triggers tumor growth (Chen et al., 2023). The growing distance of dense cancerous cells from the nearest blood arteries causes the cells to undergo hypoxia. Von Hippel–Lindau does not suppress hypoxia-inducible factor-1 (HIF-1) or scatter in hypoxic circumstances (Kaelin, 2004). Because of this, HIF-1 can dimerize and attach to the VEGF gene promoter, which stimulates the production of VEGF. VEGFR on endothelial cells then receives this VEGF, potentially causing angiogenesis (Fig. 3).
Fig. 2. The OS structure. OS is a perennial plant with a medium stature and a woody base. OS grows to a height of 30–60 cm on a standing-up, multibranched stem with hairy, subquadrangular stems. The oval-shaped, green leaves are narrowed at both ends. The petioles are 1.5–3 cm long and very slender. The petals are clustered into bracteate clusters that are 15–20 cm long on little stems.
Fig. 3. The development of cancer cells is supported by the angiogenesis that is encouraged by the VEGF. Mutant cells constantly divide, growing bigger and more susceptible to hypoxia. Hypoxic conditions will promote microenvironment cells to secrete VEGF (VEGFA, VEGFB, VEGFC, VEGFD). VEGF will bind to its receptors (VEGFR1, VEGFR2, VEGFR3) and form tip cells on endothelial cells. Tip cells will develop branches (angiogenic sprouts) to form new blood vessel channels. The new blood vessel channels will continue to branch to form vascularization in cancer cells. The role of MMP in angiogenesisIn the process of angiogenesis, endothelial cells migrate, proliferate, and create new matrix components while new capillaries grow out of the endothelial vessels that are already there to facilitate the breakdown of the vascular basement membrane. MMPs are involved in metastasis, neovascularization of tumors, and illnesses. MMPs control transitional epithelium-mesenchyme, a molecular shift that controls the appearance and function of epithelial cells. These configurations include the division of the components of the basement membrane, the breakdown of the ECM, and the intercellular disintegration and adhesion characteristics of the cell matrix (Quintero-Fabián et al., 2019). MMPs, frequently referred to as matrixes, belong to the Zn2+ endopeptidase superfamily of metzincin proteases. Against different ECM substrates, MMPs have distinct proteolytic activity (Kelwick et al., 2015). The substrate’s specificity and the overall structural domain’s shape are used to categorize MMPs. Six subfamilies make up the categorization of MMPs: membrane-type MMP (MT)-MMPs, collagenases, gelatinases, stromelysins, matrilysins, and other MMPs (Quintero-Fabián et al., 2019). Proteases, which are precursors of all MMPs, must undergo proteolytic cleavage in a physiological setting in order to facilitate the release of propeptide domains or zymogen activation, which in turn produces MMPs (Jabłońska-Trypuć et al., 2016). Endogenous inhibitors and posttranslational proteolytic cleavage control MMP activity (Cabral-Pacheco et al., 2020). MMPs have a role not only in the abnormalities associated with cancer but also in the control of angiogenesis, vasculogenesis, and lymphangiogenesis. MMPs take role in the degradation of certain substrates and ECM. VEGF, basic fibroblast growth factor (bFGF), cytokines (IL-1and -6), tumor growth factor-α and -β (TGF), TNF-α, PDGF, angiogenin, and hormones are among the angiogenic agents that can activate MMPs. In order to control proteolytic activity and support invasive and morphogenic processes linked to angiogenesis, endothelial cells generate vesicles containing MMP-2 and MMP-9, which are activated by VEGF and FGF-2 (Fig. 5) (Taraboletti et al., 2007; Jabłońska-Trypuć et al., 2016).
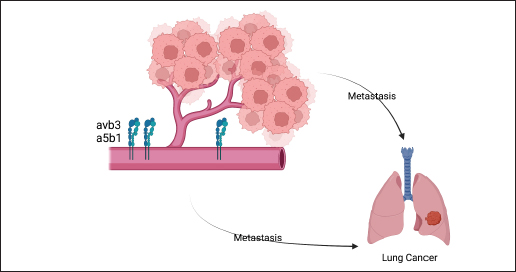
Fig. 4. The interaction of integrins αvβ3 and α5β1 with ECM regulates metastatic activity. The expression of integrin αvβ3 and α5β1 will significantly increase during the process of angiogenesis. Integrins will bind to the ECM and enhance cancer cell adhesion. Cancer cell adhesion will help cancer cells move to blood vessels that supply blood to cancer cells so that cancer cells can move to other organs suitable for growth.
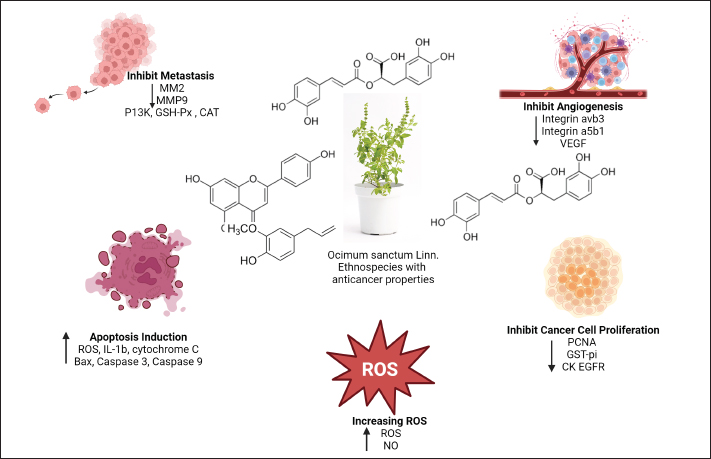
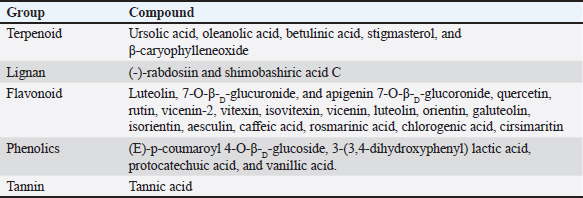
Fig. 5. OSs active ingredients inhibit cancer cells from proliferating and cause them to undergo apoptosis. Several active substances, including quercetin, eugenol, and rosmarinic acid, have been identified in OS Linn. Through the induction of cell death by an increase in ROS and NO, these bioactive substances will limit and shrink the growth of cancer cells. Bax, cytochrome C, caspase 3, caspase 9, and IL-1β all rise throughout the apoptotic process. Proliferating cell nuclear antigen (PCNA), glutathione S-transferase (GST-pi), and EGFR will all drop in response to a reduction in cancer cell proliferation. OS also inhibits metastasis, which is characterized by decreased expression of ECM (MMP-2, MMP-9), PI3K, glutathione peroxidase (GSH-Px), and catalase (CAT). It does this by reducing integrin αvβ3, integrin α5β1, and VEGF. The modulation of ECM remodeling (ECM remodeling) is influenced by MMP-2 and MMP-9. These molecules regulate laminin, glycosaminoglycans, fibronectin, aggrecan, and latent protein signaling. They are activated and deactivated by proteolytic cleavage, which releases biological activity that leads to cellular control (Mott and Werb, 2004). Gelatinases, such as MMP-2 and MMP-9, are categorized according to substrate specificity and are in charge of breaking down gelatin (Luchian et al., 2022). MMP-2, having a molecular weight of 72 kDa, is a member of the gelatinase type A subgroup. MMP-2 degrades fibronectin, elastin, Type IV, V, VII, and X nonfibrillar collagen, and the basement membrane (Chuliá-Peris et al., 2022). Cell migration, enhanced collagen affinity, elevated TGF-b bioavailability, neurodegeneration through neuron apoptosis, axon growth, mesenchymal cell differentiation with inflammatory phenotypes, anti- inflammatory effects, proliferation, and IGF-1 cleavage in the development of cancer are all functions of MMP-2 (Gonzalez-Avila et al., 2019). Tumor angiogenesis and MMP-2 expression are related. IL-8, an angiogenic agent, increases MMP-2 expression and activity in tumor cells to promote invasion. Through tumor neovascularization, metastasis, and ECM degradation, MMP-2 is crucial to the development of cancer (Quintero-Fabián et al., 2019). MMP-9, a 92 kDa type B gelatinase, is what it is called. MMP-9 degrades certain substrates, such as fibrillin, gelatin, and collagen and nonfibrillar collagen (Type IV, V, VII, X, and XIV), found in the basement membrane. MMP-9 biologically influences decreased IL-2 response, enhanced collagen affinity, and pro- and anti-inflammatory action. In addition to tumor cell resistance, TGF-b activation in cancer growth causes hypertrophic chondrocytes to undergo death and incorporates new osteoblast functional units. Additionally, MMP-9 was discovered when it was stimulated by VEGF during and after the metastasis of cancerous cells (Debbarma et al., 2020). Fortunately, during carcinogenesis, MMP-9 releases VEGF, which causes the angiogenic release and encourages the migration of endothelial cells. Increased vascular permeability brought on by MMP-9 expression stimulates the production of neovascularization in malignant tissue and regulates angiogenic factors, both of which contribute to the progression of cancer (Wihadmadyatami et al., 2020). It has been shown in several investigations that MMP- 2 and MMP-9 may diminish collagen type IV, despite being often imposed in human malignancies. Moreover, the effects of MMP-2 and MMP-9 were demonstrated using an in vivo experimental paradigm that produced the angiogenic phenotype and keratinocyte tumor invasion. Tumor growth and invasion are accelerated by MMP-2 and MMP-9 activity, which causes cancer angiogenesis by cleaving latent TGF-β in a way that is reliant on CD44. The aforementioned results may explain the pivotal function that MMP-2 and MMP-9 MP play in tumor angiogenesis by activating proangiogenic factors (Webb et al., 2017; Quintero-Fabián et al., 2019; Wihadmadyatami et al., 2020). The role of integrin a5β1 and avβ3 in angiogenesisThe transmembrane glycoprotein subunits that makeup integrin, known as alpha (α) and beta (ß), are not covalently bonded and interact with the ECM. The ß subunit binds to the cytoskeleton and affects several signaling pathways, whereas the α subunit controls the specificity of the integrin ligand. By connecting cytoskeletal actin to ECM molecules, integrins serve as cell surface protein receptors that regulate cell adhesion to the ECM. This process produces mechanical adhesion. Signal transduction pathways govern many fundamental cell functions, including cell division, proliferation, and migration, which set the stage for the interaction between integrin components and ECM. Proper cell attachment and localization are ensured by the substantial participation of integrins in ECM components, which provide signals related to cell survival (Kim et al., 2010; Bachmann et al., 2019; Valdembri and Serini, 2021). Arginine-glycine aspartic acid (RGD) strands make up short-knot fibronectin peptides, which integrins α5β1 and αvβ3 react with (Ludwig et al., 2021). The vitronectin receptor, or integrin αvβ3, is made up of two subunits: the 105-kDa β3 subunit and the 125- kDa αv subunit. Integrin αvβ3 interacts to a variety of ECM molecules that have the triple-peptide ArgGly Asp (RGD) motif, such as polarized versions of collagen and laminin (Hsu et al., 2017; Du et al., 2020), fibronectin, fibrinogen, von Willebrand factor, and vitronectin. Integrin αvβ3 plays a part in angiogenesis, pathological neovascularization, osteoclast-mediated bone resorption, and neoplasia cell metastasis. Among all the integrins, integrin αvβ3 is crucial to the angiogenesis process. A high level of αvβ3 integrin expression on cancerous cells promotes angiogenesis and makes it easier for the cells to proliferate, invade, and undergo apoptosis. bFGF and TNF-α are required for αvβ3 to start αvβ3 integrin angiogenesis, according to in vivo angiogenesis tests (Chandra Kumar, 2003; Chen et al., 2008). Additionally, involved in angiogenesis is integrin α5β1. By directly binding to either angiopoietin-171 or VEGFR-1.72, integrin α5 has been found to increase cell migration. Additionally, it has been demonstrated to cross-communicate with the Tie-2 endothelial receptor (Cascone et al., 2005). Angiogenesis depends on B1 integrins; nonetheless, it remains apparent how particular ϱ5β1 integrins function in this process. Endothelial cells, endothelial support cells, and pericytes from both normal and angiogenic vessels express integrin β1. It is believed that pericytes’ production of β1 integrin contributes to blood vessel stabilization (Carnevale et al., 2006). α5β1 interacts with a potent antiangiogenic endostatin molecule in addition to its beneficial effect in angiogenesis (Fig. 4) (Sudhakar et al., 2003) (Fig. 4). During endothelial cells undergoing neoplastic cell angiogenesis, integrin α5β1 expression is highly elevated, although it is very low in normal endothelial cells. In endothelial cells, the transcription factor HoxD3, which is associated with the homeobox family, regulates the production of α5β1. bFGF activates HoxD3 (Avraamides et al., 2008). The ECM proteins Del-1, bFGF, and IL-8, but not VEGF, were the factors that caused integrin α5β1 to express. The α5β1 integrin-mediated adhesion promotes endothelial cell motility and neoplasm cell survival by inhibiting protein kinase A activity. Therefore, during angiogenesis, α5β1 integrins preserve the survival of endothelial cells (Zhu et al., 2017). Elevated levels of α5β1 were shown to be significantly correlated with lymph node metastases of NSCLC. The survival rate was worse for patients with NSCLC displaying excess α5β1 integrin than for those with NSCLC displaying normal levels of α5β1. α5β1 integrin promotes the development of NSCLC and the spread of metastases (Aksorn and Chanvorachote, 2019; Sökeland and Schumacher, 2019). OS: prevention and medication against lung cancerOS is a native plant of the tropics and other warm climates, known by several names such as tulsi (Hindi), holy basil (English), and kemangi (Indonesia) (Joseph and Nair, 2013). Indonesians use this fragrant herb as a vegetable (Cohen, 2014; Wihadmadyatami et al., 2019). This plant has a wide range of morphological traits, which are impacted by where it grows. These plants are often fragrant, herbaceous, sun shrubs with branching, and hairy subquadrangular branches that are between 30 and 60 cm long. The plant has simple, green, or purple leaves that can be ovate, elliptic-oblong, obtuse, or acute. The leaves have pubescent edges on both surfaces and can be slightly toothed with all substrate or dentate margins. The leaves are also dotted with tiny glands and have slender, hairy petioles. Volatile oils are produced by the glandular hairs on the stalked and sessile glands on the aerial section. The flowers are purplish-white in hue and hermaphrodite, grouped in elongated racemes in tight whorls. The seeds are globose–subglobose in form and are brownish-reddish yellow (Malav et al., 2015) (Fig. 2). The classification of OS: Kingdom : Plantae (Unranked) : Angiospermae (Unranked) : Eudicots (Unranked) : Asterids Order : Lamiales Family : Lamiaceae Genus : Ocimum Species : OS L. OS has a variety of applications in different parts of Asia, including religious and therapeutic uses. It is used to treat inflammation, stomach pains, headaches, malaria, and the common cold in traditional medicine (Yamani et al., 2016). OS has been shown in prior research to have antibacterial, antiviral, antidiabetic, antioxidant, antidepressant, wound healing, anti- inflammatory, hepatoprotective, cardioprotective, hypolipidemic, immunomodulatory, radioprotective, and neurodegenerative protection functions (Raghavendra et al., 2009; Yamani et al., 2016; Almatroodi et al., 2020). OS has an abundance of chemical compounds. The ethanolic leaf extract of OS contains tolunea, camphene, octane, benzene, citronellal, sabinene, limocene, ledol, dimetilbenzena, etil-2-metilbutirat, eugenol, terpinolene, β-elemene, isocaryophyllene, iso-eugenol, α-amorf, α-guaiene, α-humulen, α-terpineol, borneol, calamine, nerolidol, carvacrol, geraniol, humulene, oksida, elemol, tetradecanal, (EZ)-famesol, cis- sesquisainenehydrate, α-bisbolol, selin-11-en-4-α-ol, α-murolene, 14-hidroksi-α-humulen, carbohydrates, tannins, flavonoids, saponins, glycosides, terpenoids, and alkaloids. Many chemical components are formed when the leaf is extracted with methanol, including fatty acids, carbohydrates, phenol, tannins, flavonoids, saponin, glycosides, terpenoids, alkaloids, and fixed oils (Singh and Chaudhuri, 2018; Kustiati, et al., 2022a). Furthermore, of the others, phenol was said to be the most prevalent. Using gas chromatography- mass spectrometry and liquid chromatography/ mass spectromethry (LC/MS) analysis, methanol was extracted and eugenol, α-farnesene, benzene, 1,2-dimethoxy-4-(1-propenyl), cyclohexane, and 1,2,4-triethyl were found (Borah and Biswan, 2018). The primary source of fixed oils, including oleic acid, stearic acid, hexuronic acid, palmitic acid, linodilinolin, and linolenic acid, is the seeds of this plant. Other phytochemical components found in the aqueous and alcoholic extracts of defatted OS seeds include tannins (tannic acid) and flavonoids (quercetin and rutin) (Sharma et al., 2020). Phenolic substances such apigenin, circimaritin, isothymucin, eugenol, and rosameric acid are produced when the stem is extracted (Bhowmik et al., 2008). After analyzing the chemical composition of these leaves’ volatile oils, 22 components were identified: α-pinene, α-thujene, α-camphene, β-pinene, α-mycrene, d-limonene, eucalyptol, cis-α-terpineol, sabinene, borneol, bornyl acetate, camphor, eugenol, selinene, caryophyllene oxide, β-guaiene, phytol, and oleic acid (Padalia and Verma, 2011; Dohare et al., 2012; Wihadmadyatami et al., 2019; Kustiati 2022a). A mixture including two lignans, four phenolics, and five terpenoids was obtained by extracting the aerial portion of OS with dichloromethane and methane (Table 3) (Flegkas et al., 2019). Table 3. The summary of phytochemical compound of the OS.
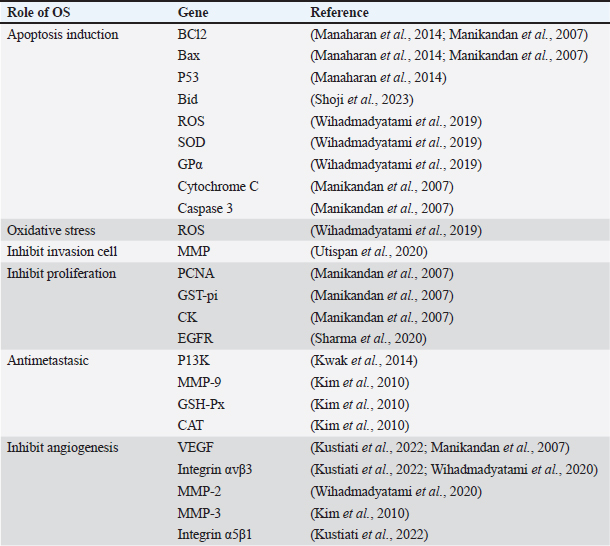
OS leaves, seeds, stems, fruits, flowers, and roots are almost all used as extracts or medications in traditional herbal remedies. However, in Indonesia, the leaves are the most commonly used herb in medicine, followed by the seeds, aerial parts, and entire plants. Similar to previous worldwide ethnobotanical studies conducted in Ghana, Lebanon, Morocco, and Turkey, an ethnobotanical reference study found that leaves were the main dominant plant component used in the composition of traditional treatments. Several studies have been carried out to comprehend OS’s impact on cancer. Ethanol extract of OS (EEOS) has been studied in vivo and in vitro for its anticancer properties. To determine the molecular target of EEOS, a study was carried out on a rat model of MNN-induced stomach carcinogenesis. As genes that promote proliferation and angiogenesis, the study found that eugenol, ursolic acid, and carvacrol extracted from the OS leaf were able to lower the expression of PCNA, GST-pi, BCl-2, CK, and VEGF in rats with stomach carcinogenesis caused by MNN. According to this discovery, substances also raise the expression of apoptosis-inducing genes such as caspase 3, cytochrome C, and Bax (Manikandan et al., 2007). The study also revealed that the expression of EGFR, Kelch-like ECH-associated protein 1 (KEAP1), and β2- adrenergic receptor (β2-AR) was successfully inhibited by quercetin, rutin, and tannic acid that were separated from defatted OS seed extract. The G protein-coupled receptors, which are seven transmembrane receptors, including the β2-AR, are involved in the development and spread of oral cancer. It was discovered that norepinephrine-stimulated β2-AR enhanced IL-6 production and cell proliferation in oral squamous cell carcinoma cell lines (Sharma et al., 2020). The lung is one of the tissues with high levels of NRF2 expression where detoxifying processes take place. It interacts with KEAP1, its own negative regulator, under normal physiological settings. The oxidative stress sensor KEAP1 mediates Nrf2 degradation (Bauer et al., 2013). According to research on an A549 human lung cancer cell, EEOS raises TUNEL 1 positive and the apoptotic sub-G1 in A549 cells, suggesting that enough DNA has been ruined by DNA fragmentation. According to a different study, EEOS’s polyphenols and flavonoids cause A549 cells to separate from the ECM, which lowers the cells’ viability. Furthermore, the capacity of EEOS to induce apoptosis raises the quantity of intracellular reactive oxygen species (ROS) and the interference activity of superoxide dismutase (SOD) and GPα (Fig. 5) (Wihadmadyatami et al., 2019). In A549 cells, angiogenesis is inhibited by downregulating integrin αvβ3, a5b1, MMP-2, and MMP-9 (Wihadmadyatami et al., 2020; Kustiati et al., 2022). The anti-metastasis action of EEOS was demonstrated to be mediated by inhibition of phosphatidylinositide 3-kinase (P13K) in research conducted on NCI-H460 NSCLC cells (Kwak et al., 2014). According to Utispan et al. (2019), rosmarinic acid in the extract of EEOS may reduce the activity of MP after preventing cell invasion (Utispan et al., 2020) (Fig. 5). The (-)-rabdosiin of EEOS has been shown to stimulate antiproliferation in human cancer cell lines in another investigation (Flegkas et al., 2019). Applying EEOS to human breast cancer cells has the potential to increase the expression of P53 and Bid (BH3 interacting domain death agonist) as well as the Bax/Bcl2 protein ratio (Manaharan et al., 2013). Singhal et al. (2017) reported that vicenin-2, a flavonoid derived from tulsi (holy basil), inhibits the proliferation of prostate cancer (CaP) and increases the antitumor properties of docetaxel in vitro and in vivo when compared to other active substances of OS Linn, such as erientin and luteolin. Vicenin-2 raises p53 levels, decreases hTERT, PCNA, and pRB, and suppresses pEGFR, pAkt, and the mTOR signaling mediator Pp70S6K (Singhal et al., 2017). Inducing apoptosis, preventing cell invasion, limiting proliferation, restricting metastasis, and inhibiting angiogenesis are among the functions of OS in lung cancer (Fig. 5). Multiple investigations have indicated that OS’s ability to fight cancer is significantly influenced by intrinsic mitochondrial apoptosis. Cytochrome C is released into the cytosol when mitochondrial malfunction brought on by the cleavage of Bid by the intrinsic apoptosis pathway triggers apoptosis. Apoptotic protease activating factor-1, or Apaf1, is an adaptor protein that is activated when cytochrome C in the cytosol attaches to procaspase. This process creates the apoptosome, which in turn attracts procaspase 9. Apoptosis is brought on by the activation of caspase-9 and caspase-3, which causes cell death and DNA fragmentation, among other apoptotic hallmarks (Manaharan et al., 2013, Manaharan et al., 2014; Wihadmadyatami et al., 2019, 2020). Table 4 provides a summary of the OS’s function. Table 4. The role of OS ethanolic extract to prevent cancer metastasis.
Future perspectiveThe results in this review indicate that mouse models predominantly used in cancer research, particularly in the study of lung cancer. The use of mice as a lung cancer model is because mice have the advantage of genetic tractability and the ability to modify different biological pathways. Nevertheless, these models possess notable constraints that can impact the applicability of results to human patients. A significant drawback of mice models is their inability to completely mimic the genetic and phenotypic diversity exhibited in human lung cancer. Moreover, the immune system of mice, especially in models with reduced immunity, fails to replicate the immunological response observed in humans accurately. The constraint above can substantially influence the examination of interactions between tumors and the immune system and the effectiveness of immunotherapies (Weiss et al., 2015). In order to address this constraint, there is a continuous effort to establish a wider range of animal species as models for studying lung cancer. This novel method is essential for enhancing the disease’s comprehension mechanism and therapy solutions. Moreover, applying genetic engineering, environmental factors, and lifestyle factors in these models will increase their significance and effectiveness in preclinical research, ultimately leading to improved outcomes for individuals with lung cancer. Understanding the interactions between tumor cells, stroma, and microenvironment, which are included in molecular carcinogenesis, is critical for developing effective therapies. Research into the role of immune cells, fibroblasts, and ECM components in lung cancer can provide insights into how these interactions influence tumor growth and metastasis (Pullamsetti et al., 2017). Targeting these interactions may enhance existing therapies’ effectiveness and lead to new treatment strategies. Moreover, as a new approach to treatment strategies, the use of herbal medicine, it is possible to employ phytoconstituents of OS in lung cancer, deeper research involving the use of various animal models and new technologies such as organoids and organs on a chip, is needed related to the functional mechanism, standardization of dosages, and regulations on the use of herbal medication. ConclusionRecently, mice have been used extensively as experimental models for lung cancer. Chemically induced mice, lung cancer cell line induction, PDXs, and genetically modified mice are some of the manufacturing techniques used. It is crucial to apply the strategy of using molecular carcinogenesis interactions, such as tumor microenvironment, genetic mutation, and epigenetics, to improve the efficacy of therapy and encourage the development of novel treatment modalities, particularly those involving herbal medicine. One of the most widely used herbal remedies in Southeast and South Asia is OS. Lung cancer therapy and prevention heavily depend on OS. OS possesses antioxidant, anti-inflammatory, immunomodulatory, antiproliferative, proapoptotic, antiangiogenic, and antimetastatic qualities, which account for its reported chemopreventive and anticancer therapeutic benefits. Furthermore, OS has the ability to control enzymatic activity, including GSH, GST, GSH-Px, and several antioxidant enzymes such as SOD and CAT, as well as signal transduction pathways. AcknowledgmentsThe authors would like to thank the Integrated Laboratory for Research and Testing, Universitas Gadjah Mada, for providing the facilities for this study. This study was supported by grants from the Directorate of Research, Universitas Gadjah Mada through Thesis Recognition Grant 2024 (Rekognisi Tugas Akhir-RTA 2024) with grant number 5286/UN1. P1/PT.01.03/2024. Conflict of interestThe authors declare that they have no conflict of interest FundingThis study was supported by grants from the Directorate of Research, Universitas Gadjah Mada through Thesis Recognition Grant 2024 (Rekognisi Tugas Akhir-RTA 2024) with grant number 5286/UN1.P1/PT.01.03/2024. Authors’ contributionsConceptualized and Supervision: H.W., D.L.K., D and .A.A.N., Methodology: H.W., Writing-original draft: U.K., H.W., D.L.K., and D.A.A.N.; Writing-review and editing: H.W. and U.K.; Visualized: U.K. and H.W. All authors have read and approved the final manuscript. Ethical approvalSince this review does not use materials obtained directly from humans or animals, there is no need for ethical clearance. ReferencesAksorn, N. and Chanvorachote, P. 2019. Integrin as a molecular target for anti-cancer approaches in lung cancer. Anticancer Res. 39(2), 541–548. Ali, Z., Mukwaya, A., Biesemeier, A., Ntzouni, M., Ramsköld, D., Giatrellis, S., Mammadzada, P., Cao, R., Lennikov, A., Marass, M., Gerri, C., Hildesjö, C., Taylor, M., Deng, Q., Peebo, B., Peso, L. del, Kvanta, A., Sandberg, R., Schraermeyer, U., Andre, H., Steffensen, J.F., Lagali, N., Cao, Y., Kele, J. and Jensen, L.D. 2019. Intussusceptive vascular remodeling precedes pathological neovascularization. Arterioscler. Thromb. Vasc. Biol. 39(7), 1402–1418. Almatroodi, S.A., Alsahli, M.A., Almatroudi, A. and Rahmani, A.H. 2020. Ocimum sanctum: role in diseases management through modulating various biological activity. Phcog. J. 12(5), 1198–1205. Anand, P., Kunnumakara, A.B., Sundaram, C., Harikumar, K.B., Tharakan, S.T., Lai, O.S., Sung, B. and Aggarwal, B.B. 2008. Cancer is a preventable disease that requires major lifestyle changes. Pharm. Res. 25(9), 2097–2116. Arfan, A. and Andarini, S.L. 2022. Role of miRNA in non-small cell lung carcinoma. Res. Sci. 2(3), 165–174. Avraamides, C.J., Garmy-Susini, B. and Varner, J.A. 2008. Integrins in angiogenesis and lymphangiogenesis. Nat. Rev. Cancer. 8(8), 604–617. Bachmann, M., Kukkurainen, S., Hytönen, V.P. and Wehrle-Haller, B. 2019. Cell adhesion by integrin. Physiol. Rev. 99, 1655–1699. Bao, P., Kodra, A., Tomic-Canic, M., Golinko, M.S., Ehrlich, H.P. and Brem, H. 2009. The role of vascular endothelial growth factor in wound healing. J. Surg. Res. 153(2), 347–358. Barck, K., Bou-Reslan, H., Rastogi, U., Sakhuja, T., Long, J.E., Molina, R., Lima, A., Hamilton, P., Junttila, M.R., Johnson, L. and Carano, R.A. 2015. Quantification of tumor burden in a genetically engineered mouse model of lung cancer by micro-CT and automated analysis. Trans. Oncol. 8(2), 126–135. Barta, J.A., Powell, C.A. and Wisnivesky, J.P. 2019. Global epidemiology of lung cancer. Ann. Glob. Health. 85(1), 8. Bauer, A.K., Hill, T. and Alexander, C.M. 2013. The involvement of NRF2 in lung cancer. Oxid. Med. Cell Longev. 2013, 746432. Bhowmik, S., Chowdhury, S.D., Kabir, M.H. and Ali, M.A. 2008. Chemical composition of some medicinal plant products of indigenous origin. Bangladesh Vet. 25(1), 32–39. Borah, R. and Biswas, S.P. 2018. Tulsi (Ocimum sanctum), excellent source of phytochemical. Int. J. Environ. Agr. Biotech. 3(5), 1732–1738. Brennecke, P., Arlt, M.J.E., Campanile, C., Husmann, K., Gvozdenovic, A., Apuzzo, T., Thelen, M., Born, W. and Fuchs, B. 2014. CXCR4 antibody treatment suppresses metastatic spread to the lung of intratibial human osteosarcoma xenografts in mice. Clin. Exp. Metastasis. 31(3), 339–349. Brown, J.S., Amend, S.R., Austin, R.H., Gatenby, R.A., Hammarlund, E.U. and Pienta, K.J. 2023. Updating the definition of cancer. Mol. Cancer Res. 21(11), 1142–1147. Brown, P.J., Bedard, L.L., Reid, K., Petsikas, D. and Massey, T.E. 2007. Analysis of CYP2A contributions to metabolism of 4-(Methylnitrosamino)-1-(3-Pyridyl)-1-butanone in human peripheral lung microsomes. Drug Metab. Dispos. 35(11), 2086–2094. Burri, P.H., Hlushchuk, R. and Djonov, V. 2004. Intussusceptive angiogenesis: its emergence, its characteristics, and its significance. Dev. Dyn. 231(3), 474–488. Cabral-Pacheco, G.A., Garza-Veloz, I., La Rosa, C.C.D., Ramirez-Acuña, J.M., Perez-Romero, B.A., Guerrero-Rodriguez, J.F., Martinez-Avila, N. and Martinez-Fierro, M.L. 2020. The roles of matrix metalloproteinases and their inhibitors in human diseases. Int. J. Mol. Sci. 21(24), 1–53. Carnevale, E., Fogel, E., Aplin, A.C., Gelati, M., Howson, K.M., Zhu, W.-H. and Nicosia, R.F. 2006. Regulation of postangiogenic neovessel survival by β1 and β3 integrins in collagen and fibrin matrices. J. Vas. Res. 44(1), 40–50. Cascone, I., Napione, L., Maniero, F., Serini, G. and Bussolino, F. 2005, Stable interaction between α5β1 integrin and Tie2 tyrosine kinase receptor regulates endothelial cell response to Ang-1. J. Cell Bio. 170(6), 993–1004. Ceci, C., Atzori, M.G., Lacal, P.M. and Graziani, G. 2020. Role of VEGFs/VEGFR-1 signaling and its inhibition in modulating tumor invasion: experimental evidence in different metastatic cancer models. Int. J. Mol. Sci. 21(4), 388. Chandra Kumar, C. 2003. Integrin αvβ3 as a therapeutic target for blocking tumor-induced angiogenesis. Curr. Drug Target. 4(2), 123-131. Chen, Q., Manning, C.D., Millar, H., McCabe, F.L., Ferrante, C., Sharp, C., Shahied-Arruda, L., Doshi, P., Nakada, M.T. and Anderson, G.M. 2008. CNTO 95, a fully human anti αv integrin antibody, inhibits cell signaling, migration, invasion, and spontaneous metastasis of human breast cancer cells. Clin. Exp. Metastasis. 25(2), 139–148. Chen, Z., Han, F., Du, Y., Shi, H. and Zhou, W. 2023. Hypoxic microenvironment in cancer: molecular mechanisms and therapeutic interventions. Signal Transduct. Target Ther. 8(1), 70. Chong, Y.J., Jung, Y.C., Hwang, E., Cho, H.J., Kang, M.-W. and Na, M.H. 2021. Circulating tumor cell detection in lung cancer animal model. J. Chest Surg. 54(6), 460–465. Chuliá-Peris, L., Carreres-Rey, C., Gabasa, M., Alcaraz, J., Carretero, J. and Pereda, J. 2022. Matrix metalloproteinases and their inhibitors in pulmonary fibrosis: EMMPRIN/CD147 comes into play. Int. J. Mol. Sci. 23(13), 6894. Çiçek, B., Hayme, S., Kuzucu, M., Çetin, A., Yeni, Y., Genç, S., Yıldırım, S., Bolat, İ., Kantarcı, M., Gül, M. and Hacımüftüoğlu, A. 2022. Sorafenib alleviates inflammatory signaling of tumor microenvironment in lung cancer. Pharmaceuticals (Basel). 16(2), 221. Cohen, M.M. 2014. Tulsi-Ocimum sanctum: a herb for all reasons. J. Ayurveda Integr. Med. 5(4), 251–259. De Minicis, S., Kisseleva, T., Francis, H., Baroni, G.S., Benedetti, A., Brenner, D., Alvaro, D., Alpini, G. and Marzioni, M. 2013. Liver carcinogenesis: rodent models of hepatocarcinoma and cholangiocarcinoma. Dig. Liver Dis. 45(6), 450–459. Debbarma, P., Mondal, T., Manna, C., Kumar, K., Mukherjee, J., Das, B.C., Bag, S. and Das, K. 2020. Post-calving umbilical cord tissue offcut: a potential source for the isolation of bovine mesenchymal stem cells. Vet. World. 13(12), 2772. Deng, W.G., Kawashima, H., Wu, G., Jayachandran, G., Xu, K., Minna, J.D., Roth, J.A. and Ji, L. 2007. Synergistic tumor suppression by coexpression of FUS1 and p53 is associated with down-regulation of murine double minute-2 and activation of the apoptotic protease-activating factor 1–dependent apoptotic pathway in human non–small cell lung cancer cells. Cancer Res. 67(2), 709–717. Dohare, S.L., Shuaib, M., Ahmad, M.I. and Naquvi, K.J. 2012. Chemical composition of volatile oil of Ocimum sanctum Linn. Int. J. Biomed. Adv. Res. 3(2), 129–131. Domínguez-Oliva, A., Hernández-Ávalos, I., Martínez-Burnes, J., Olmos-Hernández, A., Verduzco-Mendoza, A. and Mota-Rojas, D. 2023. The importance of animal models in biomedical research: current insights and applications. Animals 13(7), 1223. Du, Q., Wang, W., Liu, T., Shang, C., Huang, J., Liao, Y., Qin, S., Chen, Y., Liu, P., Liu, J. and Yao, S. 2020. High expression of integrin α3 predicts poor prognosis and promotes tumor metastasis and angiogenesis by activating the c-Src/extracellular signal-regulated protein kinase/focal adhesion kinase signaling pathway in cervical cancer. Front. Oncol. 10, 36. Du, S., Lockamy, V., Zhou, L., Xue, C., LeBlanc, J., Glenn, S., Shukla, G., Yan, Y., Dicker, A.P., Leeper, D.B., Lü, Y. and Lü, B. 2016. Stereotactic body radiation therapy delivery in a genetically engineered mouse model of lung cancer. Int. J. Radiat. Oncol. Biol. Phys. 96(3), 529–537. Flegkas, A., Milosević Ifantis, T., Barda, C., Samara, P., Tsitsilonis, O. and Skaltsa, H. 2019. Antiproliferative activity of (-)-rabdosiin isolated from Ocimum sanctum L. Medicines 6(1), 37. Fowler, B.L., Johannes, C.M., O’Connor, A., Collins, D., Lustgarten, J., Yuan, C., Weishaar, K., Sullivan, K., Hume, K.R., Mahoney, J., Vale, B., Schubert, A., Ball, V., Cooley-Lock, K., Curran, K.M., Nafe, L., Gedney, A., Weatherford, M. and LeVine, D.N. 2020. Ecological level analysis of primary lung tumors in dogs and cats and environmental radon activity. J. Vet. Intern. Med. 34(6), 2660–2670. Freeman, B., Mamallapalli, J., Bian, T., Ballas, K., Lynch, A.M., Scala, A., Huo, Z., Fredenburg, K.M., Bruijnzeel, A.W., Baglole, C.J., Lü, J., Salloum, R.G., Malaty, J. and Xing, C. 2023. Opportunities and challenges of kava in lung cancer prevention. Int. J. Mol. Sci. 24(11), 9539. Fu, S., Zhao, J., Bai, H., Duan, J., Wang, Z., An, T. and Wang, J. 2015. High-fidelity of non-small cell lung cancer xenograft models derived from bronchoscopy-guided biopsies. Thorac. Cancer 7(1), 100–110. Gao, L., Hu, Y., Tian, Y., Fan, Z., Wang, K., Li, H., Zhou, Q., Zeng, G., Hu, X., Yu, L., Zhou, S., Tong, X., Huang, H., Chen, H., Liu, Q., Liu, W., Zhang, G., Zeng, M., Zhou, G., He, Q., Ji, H. and Chen, L. 2019. Lung cancer deficient in the tumor suppressor GATA4 is sensitive to TGFBR1 inhibition. Nat. Commun. 10(1), 1665. Garcia-Oliveira, P., Fraga-Corral, M., Pereira, A.G., Lourenço-Lopes, C., Jimenez-Lopez, C., Prieto, M.A. and Simal-Gandara, J. 2020. Scientific basis for the industrialization of traditionally used plants of the Rosaceae family. Food Chem. 330, 127197. Gautam, A., Li, Z.R. and Bepler, G. 2003. RRM1-induced metastasis suppression through PTEN-regulated pathways. Oncogene 22(14), 2135–2142. Gonzalez-Avila, G., Sommer, B., Mendoza-Posada, D.A., Ramos, C., Garcia-Hernandez, A.A. and Falfan-Valencia, R. 2019. Matrix metalloproteinases participation in the metastatic process and their diagnostic and therapeutic applications in cancer. Crit. Rev. Oncol. Hematol. 137, 57–83. Han, A., Song, Z., Tong, C., Hu, D., Bi, X., Augenlicht, L.H. and Yang, W. 2008. Sulindac suppresses Β-catenin expression in human cancer cells. Eur. J. Pharmacol. 583(1), 26–31. Houdebine, L.M. 2014. Impacts of genetically modified animals on the ecosystem and human activities. Global Bioethics 25(1), 3–18. Hsu, L.H., Chu, N.M. and Kao, S.H. 2017. Estrogen, estrogen receptor and lung cancer. Int. J. Mol. Sci. 18(8), 1713. Huang, C., Williamson, S.K., Neupane, P., Taylor, S., Allen, A., Smart, N.J., Uypeckcuat, A.M., Spencer, S., Wick, J., Smith, H.D., Veldhuizen, P.J.V. and Kelly, K. 2016. Impact study: MK-0646 (Dalotuzumab), insulin growth factor 1 receptor antibody combined with pemetrexed and cisplatin in stage IV metastatic non-squamous lung cancer. Front. Oncol. 5, 301. Huang, G., Eisenberg, R., Yan, M., Monti, S., Lawrence, E., Fu, P., Walbroehl, J., Löwenberg, E.C., Golub, T.R., Merchan, J.R., Tenen, D.G., Markowitz, S.D. and Halmos, B. 2008. 15-hydroxyprostaglandin dehydrogenase is a target of hepatocyte nuclear factor 3β and a tumor suppressor in lung cancer. Cancer Res. 68(13), 5040–5048. Huang, L., Guo, Z., Wang, F. and Fu, L. 2021. KRAS mutation: from undruggable to druggable in cancer. Signal Transduct. Target Ther. 6(1), 386. Ilié, M., Nunes, M., Blot, L., Long-Mira, E., Butori, C., Selva, É., Merino-Trigo, A., Vénissac, N., Mouroux, J., Vrignaud, P. and Hofman, P. 2014. Setting up a wide panel of patient-derived tumor xenografts of non–small cell lung cancer by improving the preanalytical steps. Cancer Med. 4(2), 201–211. Jabłońska-Trypuć, A., Matejczyk, M. and Rosochacki, S. 2016. Matrix metalloproteinases (MMPs), the main extracellular matrix (ECM) enzymes in collagen degradation, as a target for anticancer drugs. J. Enzyme Inhib. Med. Chem. 31, 177–183. Janker, F., Weder, W., Jang, J. and Jungraithmayr, W. 2018. Preclinical, non-genetic models of lung adenocarcinoma: a comparative survey. Oncotarget 9(55), 30527–30538. Joseph, B. and Nair, V.M. 2013. Ethanopharmacological and phytochemical aspects of Ocimum sanctum Linn-the elixir of life. J. Pharm. Res. Int. 3, 273–292. Kaelin Jr, W.G. 2004. The Von Hippel-Lindau tumor suppressor gene and kidney cancer. Clin. Cancer Res. 10(18), 6290S–6295S. Katz, A., Barash, U., Boyango, I., Feld, S., Zohar, Y., Hammond, E., Ilan, N., Kremer, R. and Vlodavsky, I. 2018. Patient derived xenografts (PDX) predict an effective heparanase-based therapy for lung cancer. Oncotarget 9(27), 19294–19306. Kelwick, R., Desanlis, I., Wheeler, G.N. and Edwards, D.R. 2015. The ADAMTS (A Disintegrin and Metalloproteinase with Thrombospondin motifs) family. Genome Biol. 16(1), 113. Khan, A., Alsahli, M.A., Aljasir, M., Maswadeh, H., Mobark, M.A., Azam, F., Allemailem, K.S., Alhumaydhi, F.A., Almatroudi, A., Alsuhaymi, N. and Khan, M.A. 2022. Experimental and theoretical insights on chemopreventive effect of the liposomal thymoquinone against benzo[a]pyrene-induced lung cancer in Swiss albino mice. J. Inflamm. Res. 15, 2263–2280. Kim, K.H. 2024. Tumor immunogenicity regulates host immune responses, and conventional dendritic cell type 2 uptakes the majority of tumor antigens in an orthotopic lung cancer model. Cancer Immunol. Immunother. 73(12), 237. Kim, S., Yoon, J.I., Tommasi, S. and Besaratinia, A. 2012. New experimental data linking secondhand smoke exposure to lung cancer in nonsmokers. FASEB J. 26(5), 1845–1854. Kim, S.C., Magesh, V., Jeong, S.J., Lee, Hyo Jung, Ahn, K.S., Lee, Hyo Jeong, Lee, E.O., Kim, Sun Hyung, Lee, M.H., Kim, J.H. and Kim, Sung Hoon. 2010. Ethanol extract of Ocimum sanctum exerts anti-metastatic activity through inactivation of matrix metalloproteinase-9 and enhancement of anti-oxidant enzymes. Food Chem. Toxicol. 48(6), 1478–1482. Kristina, S.A., Wiedyaningsih, C., Masrida, W.O., Andriani, Y. and Puspawati, P.R. 2019. Estimation of tobacco related cancers deaths and costs of productivity lost in Indonesia. Int. Res. J. Pharm. 10(9), 43–46. Ku, T.K., Nguyen, D.C., Karaman, M.W., Gill, P.S., Hacia, J.G. and Crowe, D.L. 2007. Loss of P53 expression correlates with metastatic phenotype and transcriptional profile in a new mouse model of head and neck cancer. Mol. Cancer. Res. 5(4), 351–362. Kusindarta, D.L., Wihadmadyatami, H. and Haryanto, A. 2016. Ocimum sanctum Linn. stimulate the expression of choline acetyltransferase on the human cerebral microvascular endothelial cells. Vet. World. 9(12), 1348–1354. Kusindarta, D.L., Wihadmadyatami, H. and Haryanto, A. 2018. The analysis of hippocampus neuronal density (CA1 and CA3) after Ocimum sanctum ethanolic extract treatment on the young adulthood and middle age rat model. Vet. World. 11(2), 135–140. Kusindarta, D.L., Wihadmadyatami, H., Jadi, A.R., Karnati, S., Lochnit, G., Hening, P., Haryanto, A., Auriva, M.B. and Purwaningrum, M. 2018a. Ethanolic extract Ocimum sanctum. enhances cognitive ability from young adulthood to middle aged mediated by increasing choline acetyl transferase activity in rat model. Res. Vet. Sci. 118, 431–438. Kustiati, U., Ergün, S., Karnati, S., Nugrahaningsih, D.A.A., Kusindarta, D.L. and Wihadmadyatami, H. 2022. Ethanolic extract of Ocimum sanctum Linn. inhibits cell migration of human lung adenocarcinoma cells (A549) by downregulation of integrin αvβ3, α5β1, and VEGF. Sci. Pharm. 90(4), 69. Kustiati, U., Wihadmadyatami, H. and Kusindarta, D.L. 2022a. Dataset of phytochemical and secondary metabolite profiling of holy basil leaf (Ocimum sanctum Linn) ethanolic extract using spectrophotometry, thin layer chromatography, Fourier transform infrared spectroscopy, and nuclear magnetic resonance. Data Brief 40, 107774. Kwak, T.K., Sohn, E.J., Kim, S., Won, G., Choi, J.U., Jeong, K., Jeong, M., Kwon, O.S. and Kim, S.H. 2014. Inhibitory effect of ethanol extract of Ocimum sanctum on osteopontin mediated metastasis of NCI-H460 non-small cell lung cancer cells. BMC Complement. Altern. Med. 14, 419. Lamalice, L., Le Boeuf, F. and Huot, J. 2007. Endothelial cell migration during angiogenesis. Cir. Res. 100(6), 782–794. Larsen, J.E. and Minna, J.D. 2011. Molecular biology of lung cancer: clinical implications. Clin. Chest Med. 32(4), 703–740. Lee, J., Han, Y.A., Yang, H.S., Song, J.A., Yang, Y.S., Kwon, S., Kang, M.S., Lee, K., Heo, J.D., Cho, K.H. and Song, C.W. 2010. The incidence rate and severity of orthotopic lung cancer in an animal model depends on the number of A549 cells and transplantation period. Lab. Animal Res. 26(4), 369. Li, C. and Lu, H. 2018. Adenosquamous carcinoma of the lung. Onco Targets Ther. 11, 4829–4835. Li, Y., Qu, P., Wu, L., Li, B., Du, H. and Yan, C. 2011. Api6/Aim/Spα/Cd5l overexpression in alveolar type II epithelial cells induces spontaneous lung adenocarcinoma. Cancer Res. 71(16), 5488–5499. Li, Z., Zheng, W., Wang, H., Cheng, Y., Fang, Y., Wu, F., Sun, G., Sun, G., Lv, C. and Hui, B. 2021. Application of animal models in cancer research: recent progress and future prospects. Cancer Manag. Res. 13, 2455–2475. Liao, R.G., Jung, J., Tchaicha, J.H., Wilkerson, M.D., Sivachenko, A., Beauchamp, E.M., Liu, Q., Pugh, T.J., Pedamallu, C.S., Hayes, D.N., Gray, N.S., Getz, G., Wong, K.K., Haddad, R.I., Meyerson, M. and Hammerman, P.S. 2013. Inhibitor-sensitive FGFR2 and FGFR3 mutations in lung squamous cell carcinoma. Cancer Res. 73(16), 5195–5205. Liu, D., Pan, L., Zhai, H., Qiu, H.J. and Sun, Y. 2023. Virus tracking technologies and their applications in viral life cycle: research advances and future perspectives. Front. Immunol. 14, 1204730. Liu, J. and Johnston, M. 2002. Animal models for studying lung cancer and evaluating novel intervention strategies. Surg. Oncol. 11(4), 217–227. Luchian, I., Goriuc, A., Sandu, D. and Covasa, M. 2022. The role of matrix metalloproteinases (MMP-8, MMP-9, MMP-13) in periodontal and peri-implant pathological processes. Int. J. Mol. Sci. 23(3), 1806. Ludwig, B.S., Kessler, H., Kossatz, S. and Reuning, U. 2021. RGD-binding integrins revisited: how recently discovered functions and novel synthetic ligands (re-)shape an ever-evolving field. Cancers 13(7), 1711. Luettich, K., Xiang, Y., Iskandar, A., Sewer, A., Martin, F., Talikka, M., Vanscheeuwijck, P., Berges, A., Veljkovic, E., González-Suárez, I., Schlage, W.K., Hoeng, J. and Peitsch, M.C. 2014. Systems toxicology approaches enable mechanistic comparison of spontaneous and cigarette smoke-related lung tumor development in the A/J mouse model. Interdiscip. Toxicol. 7(2), 73–84. Lugano, R., Ramachandran, M. and Dimberg, A. 2020. Tumor angiogenesis: causes, consequences, challenges and opportunities. Cell. Mol. Life Sci. 77(9), 1745–1770. Luo, Q., Wang, J., Zhao, W., Peng, Z., Liu, X., Li, B., Zhang, H., Shan, B., Zhang, C. and Duan, C. 2020. Vasculogenic mimicry in carcinogenesis and clinical applications. J. Hematol. Oncol. 13(1), 19. Ma, Y., Zhang, P., An, G., Zhang, X., Zhang, L., Si, J., Zhang, J. and Yang, Y. 2016. Induction of patient-derived xenograft formation and clinical significance of programmed cell death ligand 1 (PD-L1) in lung cancer patients. Med. Sci. Monit. 22, 4017–4025. Malav, P., Pandey, A., Bhatt, K.C., Gopala Krishnan, S. and Bisht, I.S. 2015. Morphological variability in holy basil (Ocimum tenuiflorum L.) from India. Genet. Resour. Crop Evol. 62(8), 1245–1256. Manaharan, T., Ming, C.H. and Palanisamy, U.D. 2013. Syzygium aqueum leaf extract and its bioactive compounds enhances pre-adipocyte differentiation and 2-NBDG uptake in 3T3-L1 cells. Food Chem. 136(2), 354–363. Manaharan, T., Thirugnanasampandan, R., Jayakumar, R., Ramya, G., Ramnath, G. and Kanthimathi, M.S. 2014. Antimetastatic and anti-inflammatory potentials of essential oil from edible Ocimum sanctum leaves. Sci. World J. 2014, 239508. Manikandan, P., Murugan, R.S., Abbas, H., Abraham, S.K. and Nagini, S. 2007. Ocimum sanctum Linn. (Holy Basil) ethanolic leaf extract protects against 7,12-dimethylbenz[a]anthracene-induced genotoxicity, oxidative stress, and imbalance in xenobiotic-metabolizing enzymes. J. Med. Food. 10(3), 495–502. Marçola, M. and Rodrigues, C.E. 2015. Endothelial progenitor cells in tumor angiogenesis: another brick in the wall. Stem Cells Int. 2015, 832649. McFadden, D.G., Politi, K., Chen, F.K., Song, X., Pirun, M., Santiago, P.M., Kim, C., Platt, J.T., Lee, E., Hodges, E., Rosebrock, A.P., Bronson, R.T., Socci, N.D., Hannon, G.J., Jacks, T. and Varmus, H. 2016. The mutational landscape of EGFR-, MYC-, and Kras-driven genetically-engineered mouse models of lung adenocarcinoma. Proc. Natl. Acad. Sci. 113(42), E6409. Mervai, Z., Egedi, K., Kovalszky, I. and Baghy, K. 2018. Diethylnitrosamine induces lung adenocarcinoma in FVB/N mouse. BMC Cancer 18(1), 157. Miller, A.D., Heras, M.D.L., Yu, J., Zhang, F., Liu, S.L., Vaughan, A.E., Vaughan, T.L., Rosadio, R., Rocca, S., Palmieri, G., Goedert, J.J., Fujimoto, J. and Wistuba, I.I. 2017. Evidence against a role for jaagsiekte sheep retrovirus in human lung cancer. Retrovirology 14(1), 3. Miller, M.A. and Zachary, J.F. 2017. Mechanisms and morphology of cellular injury, adaptation, and death. In Pathologic basis of veterinary disease. Eds., Zachary, J.F. Maryland Heights, MO: Mosby, p. 2–43, 219. Minowada, G. and Miller, Y.E. 2009. Overexpression of sprouty 2 in mouse lung epithelium inhibits urethane-induced tumorigenesis. Am. J. Respir. Cell Mol. Biol. 40(1), 31–37. Mott, J.D. and Werb, Z. 2004. Regulation of matrix biology by matrix metalloproteinases. Curr. Opin. Cell Biol. 16(5), 558–564. Mukhopadhyay, D., Zeng, H. and Bhattacharya, R. 2004. Complexity in the vascular permeability factor/vascular endothelial growth factor (VPF/VEGF)-receptors signaling. Mol. Cell. Biochem. 264(1–2), 51–61. Mustafa, M., Azizi, AR.J., IIIzam, EL., Nazirah, A., Sharifa, S. and Abbas, SA. 2016. Lung cancer: risk factors, management, and prognosis. IOSR J. Dent. Med. Sci. 15(10), 94–101. Nolan, K., Verzosa, G., Cleaver, T., Tippimanchai, D., DePledge, L.N., Wang, X.J., Young, C., Le, A., Doebele, R., Li, H. and Malkoski, S.P. 2020. Development of syngeneic murine cell lines for use in immunocompetent orthotopic lung cancer models. Cancer Cell Int. 20(1), 217. Norton, K.A. and Popel, A.S. 2016. Effects of endothelial cell proliferation and migration rates in a computational model of sprouting angiogenesis. Sci. Rep. 6, 36992. Okano, T., Gemma, A., Hosoya, Y., Hosomi, Y., Nara, M., Kokubo, Y., Yoshimura, A., Shibuya, M., Nagashima, M., Harris, C.C. and Kudoh, S. 2006. Alterations in novel candidate tumor suppressor genes, ING1 and ING2 in human lung cancer. Oncol. Rep. 15(3), 545–549. Onaciu, A., Munteanu, R., Munteanu, V.C., Gulei, D., Raduly, L., Feder, R.I., Pirlog, R., Atanasov, A.G., Korban, S.S., Irimie, A. and Berindan-Neagoe, I. 2020. Spontaneous and induced animal models for cancer research. Diagnostics 10(9), 660. Ormandy, E.H., Dale, J. and Griffin, G. 2011. Genetic engineering of animals: ethical issues, including welfare concerns. Can. Vet. J. 52(5), 544–550. Ota, T., Okabayashi, A. and Fukuoka, M. 2021. Rapid and dramatic responses to dabrafenib and trametinib in BRAF V600e-mutated lung adenocarcinoma. Resiprol. Case Rep. 9(10), e0841. Padalia, R.C. and Verma, R.S. 2011. Comparative volatile oil composition of four Ocimum species from northern India. Nat. Prod. Res. 25(6), 569–575. Papagiannakopoulos, T., Bauer, M.R., Davidson, S.M., Heimann, M., Subbaraj, L., Bhutkar, A., Bartlebaugh, J., Vander Heiden, M.G. and Jacks, T. 2016. Circadian rhythm disruption promotes lung tumorigenesis. Cell Metab. 24(2), 324–331. Pullamsett, S.S., Kojonazarov, B., Storn, S., Gall, H., Salazar, Y., Wolf, J., Weigert, A., El-Nikhely, N., Ghofrani, H.A., Krombach, G.A., Fink, L., Gattenlöhner, S., Rapp, U.R., Schermuly, R.T., Grimminger, F., Seeger, W. and Savai, R. 2017. Lung cancer–associated pulmonary hypertension: role of microenvironmental inflammation based on tumor cell–immune cell cross-talk. Sci. Transl. Med. 9(416), eaai9048. Quintero-Fabián, S., Arreola, R., Becerril-Villanueva, E., Torres-Romero, J.C., Arana-Argáez, V., Lara-Riegos, J., Ramírez-Camacho, M.A. and Alvarez-Sánchez, M.E. 2019. Role of matrix metalloproteinases in angiogenesis and cancer. Front. Oncol. 9, 1370. Raditya, M.N., Bagus, A.M., Kustiati, U., Wihadmadyatami, H. and Kusindarta, D.L. 2020. Data of the expression of serotonin in Alzheimer’s disease (AD) rat model under treatment of ethanolic extract Ocimum sanctum Linn. Data Brief 30, 105654. Raghavendra, M., Maiti, R., Kumar, S. and Acharya, S.B. 2009. Role of Ocimum sanctum in the experimental model of Alzheimer′s disease in rats. Int. J. Green Pharm. 3(1), 6–15. Rai, S.K., DeMartini, J.C. and Miller, A.D. 2000. Retrovirus vectors bearing jaagsiekte sheep retrovirus Env transduce human cells by using a new receptor localized to chromosome 3p21.3. J. Virol. 74(10), 4698–4704. Rajabi, M. and Mousa, S.A. 2017. The role of angiogenesis in cancer treatment. Biomedicines 5(2), 34. Rudolph, A., Chang-Claude, J. and Schmidt, M.K. 2016. Gene-environment interaction and risk of breast cancer. Br. J. Cancer 114(2), 125–133. Shao, C., Zhao, L., Wang, K., Xu, W., Zhang, J. and Yang, B. 2012. The tumor suppressor gene RBM5 inhibits lung adenocarcinoma cell growth and induces apoptosis. World J. Surg. Oncol. 10(1), 160. Sharma, Y., Bharadwaj, M., Srivastava, N., Kaur, A., Kumar, M., Agarwal, M., Bahl, Y. and Bala, K. 2020. In vitro antioxidant activity of defatted seed extracts of Ocimum sanctum on rat PC-12 cells and its inhibitory efficacy with receptors of oral squamous cell carcinoma. Ind. Crops Prod. 154, 112668. Shojaee, S. and Nana-Sinkam, P. 2017. Recent advances in the management of non-small cell lung cancer. F1000Res. 6, 2110. Shoji, C., Kikuchi, K., Yoshida, H., Miyachi, M., Yagyu, S., Tsuchiya, K., Nakaya, T., Hosoi, H. and Iehara, T. 2023. In ovo chorioallantoic membrane assay as a xenograft model for pediatric rhabdomyosarcoma. Oncol. Rep. 49(4), 76. Siddiqua, A., Hahladakis, J.N. and Al-Attiya, W.A.K.A. 2022. An overview of the environmental pollution and health effects associated with waste landfilling and open dumping. Environ. Sci. Pollut. Res. Int. 29(39), 58514–58536. Siegel, R.L., Miller, K.D. and Jemal, A. 2015. Cancer statistics 2015. CA Cancer J. Clin. 65(1), 5–29. Singh, D. and Chaudhuri, P.K. 2018. A review on phytochemical and pharmacological properties of Holy basil (Ocimum sanctum L.). Ind. Crops Prod. 118(S1), 367–382. Singhal, S.S., Jain, D., Singhal, P., Awasthi, S., Singhal, J. and Horne, D. 2017. Targeting the mercapturic acid pathway and vicenin-2 for prevention of prostate cancer. Biochim. Biophys. Acta 868(1), 167–175. Sökeland, G. and Schumacher, U. 2019. The functional role of integrins during intra- and extravasation within the metastatic cascade. Mol. Cancer. 18(1), 12. Stabile, L.P., Rothstein, M.E., Keohavong, P., Lenzner, D., Land, S.R., Gaither-Davis, A., Kim, K.J., Kaminski, N. and Siegfried, J.M. 2010. Targeting of both the C-Met and EGFR pathways results in additive inhibition of lung tumorigenesis in transgenic mice. Cancer 2(4), 2153–2170. Suda, K. and Mitsudomi, T. 2014. Successes and limitations of targeted cancer therapy in lung cancer. Prog. Tumor Res. 41, 62–77. Sudhakar, A., Sugimoto, H., Yang, C., Lively, J., Zeisberg, M. and Kalluri, R. 2003. Human tumstatin and human endostatin exhibit distinct antiangiogenic activities mediated by v3 and 51 integrins. Proc. Natl. Acad. Sci. 100(8), 4766–4771. Taraboletti, G., Ascenzo, S.D., Borsotti, P., Giavazzi, R., Pavan, A. and Dolo, V. 2007. Shedding of the matrix metalloproteinases MMP-2, MMP-9, and MT1-MMP as membrane vesicle-associated components by endothelial cells. Am. J. Pathol. 160(2), 673–80. Tratar, U.L., Horvat, S. and Cemazar, M. 2018. Transgenic mouse models in cancer research. Front. Oncol. 8, 268. Travis, W.D., Brambilla, E., Noguchi, M., Nicholson, A.G., Geisinger, K., Yatabe, Y., Ishikawa, Y., Wistuba, I., Flieder, D.B., Franklin, W., Gazdar, A., Hasleton, P.S., Henderson, D.W., Kerr, K.M., Petersen, I., Roggli, V., Thunnissen, E. and Tsao, M. 2013. Diagnosis of lung cancer in small biopsies and cytology: implications of the 2011 international association for the study of lung cancer/American Thoracic Society/European respiratory society classification. Arch. Pathol. Lab. Med. 137(5), 668–684. Uner, O.E., Gandrakota, N., Azarcon, C.P. and Grossniklaus, H.E. 2022. Animal models of uveal melanoma. Ann. Eye Sci. 7, 7. Urbich, C. and Dimmeler, S. 2004. Endothelial progenitor cells. Circ. Res. 95(4), 343–353. Utispan, K., Niyomtham, N., Yingyongnarongkul, B.E. and Koontongkaew, S. 2020. Ethanolic extract of Ocimum sanctum leaves reduced invasion and matrix metalloproteinase activity of head and neck cancer cell lines. Asian Pac. J. Cancer Prev. 21(2), 363–370. Valdembri, D. and Serini, G. 2021. The roles of integrins in cancer. Fac. Rev. 10, 45. Ventii, K.H., Devi, N.S., Friedrich, K.L., Chernova, T.A., Tighiouart, M., Meir, E.G.V. and Wilkinson, K.D. 2008. BRCA1-associated protein-1 is a tumor suppressor that requires deubiquitinating activity and nuclear localization. Cancer Res. 68(17), 6953–6962. Vertrees, R.A., Quast, M.J., Lightfoot, K., Boor, P.J. and Zwischenberger, J.B. 2000. Development of a human to murine orthotopic xenotransplanted lung cancer model. J. Invest. Surg. 13(6), 347–356. Villaruz, L.C., Socinski, M.A., Abberbock, S., Berry, L.D., Johnson, B.E., Kwiatkowski, D.J., Iafrate, A.J., Varella-Garcia, M., Franklin, W.A., Camidge, D.R., Sequist, L.V, Haura, E.B., Ladanyi, M., Kurland, B.F., Kugler, K., Minna, J.D., Bunn, P.A. and Kris, M.G. 2014. Clinicopathologic features and outcomes of patients with lung adenocarcinomas harboring BRAF mutations in lung cancer mutation consortium. Cancer 121(3), 448–456. Virmani, A., Rathi, A., Heda, S., Sugio, K., Lewis, C., Tonk, V., Takahashi, T., Roth, J.A., Minna, J.D., Euhus, D.M. and Gazdar, A.F. 2003. Aberrant methylation of the cyclin D2 promoter in primary small cell, nonsmall cell lung and breast cancers. Int. J. Cancer 107(3), 341–345. Walrath, J.C., Hawes, J.J., Dyke, T.V. and Reilly, K.M. 2010. Genetically engineered mouse models in cancer research. Adv. Cancer Res. 106, 113–164. Wang, X. and Khalil, R.A. 2018. Matrix metalloproteinases, vascular remodeling, and vascular disease. Adv. Pharmacol. 81, 241–330. Wang, X., Veeraraghavan, V.P., Mohan, S.K. and Lv, F. 2021. Anticancer and immunomodulatory effect of rhaponticin on benzo(a)pyrene-induced lung carcinogenesis and induction of apoptosis in A549 cells. Saudi J. Biol. Sci. 28(8), 4522–4531. Webb, A.H., Gao, B.T., Goldsmith, Z.K., Irvine, A.S., Saleh, N., Lee, R.P., Lendermon, J.B., Bheemreddy, R., Zhang, Q., Brennan, R.C., Johnson, D., Steinle, J.J., Wilson, M.W. and Morales-Tirado, V.M. 2017. Inhibition of MMP-2 and MMP-9 decreases cellular migration, and angiogenesis in in vitro models of retinoblastoma. BMC Cancer 17(1), 434. Weiss, I.D., Ella, E., Dominsky, O., Smith, Y., Abraham, M., Wald, H., Shlomai, Z., Zamir, G., Feigelson, S.W., Shezen, E., Bar-Shai, A., Alon, R., Izhar, U., Peled, A., Shapira, O.M. and Wald, O. 2015. In the hunt for therapeutic targets: mimicking the growth, metastasis, and stromal associations of early-stage lung cancer using a novel orthotopic animal model. J. Thorac. Oncol. 10(1), 46–58. Weng, C.J., Yang, Y.T., Ho, C.T. and Yen, G.C. 2009. Mechanisms of apoptotic effects induced by resveratrol, dibenzoylmethane, and their analogues on human lung carcinoma cells. J. Agri. Food Chem. 57(12), 5235–5243. Wihadmadyatami, H., Karnati, S., Hening, P., Tjahjono, Y., Rizal, Maharjanti, F., Kusindarta, D.L., Triyono, T. and Supriatno. 2019. Ethanolic extract Ocimum sanctum Linn. induces an apoptosis in human lung adenocarcinoma (A549) cells. Heliyon 5(11), e02772. Wihadmadyatami, H., Hening, P., Kustiati, U., Kusindarta, D.L., Triyono, T. and Supriatno, S. 2020. Ocimum sanctum Linn. ethanolic extract inhibits angiogenesis in human lung adenocarcinoma (a549) cells. Vet. World 13(9), 2028–2032. Wihadmadyatami, H., Karnati, S., Ergún, S., Kustiati, U., Tirtosari, D.R., Kusindarta, D.L. and Tjahjono, Y. 2023. Ocimum sanctum Linn. ethanolic extract promotes an antiproliferative and apoptosis activity in MCF-7 and T47D breast cancer cell lines mediated by upregulation of ROS/RNS, Caspase 9, and Caspase 3: an in silico and in vitro study. F1000Res. 12, 136. Wislez, M., Spencer, M.L., Izzo, J.G., Juroske, D.M., Balhara, K., Cody, D.D., Price, R.E., Hittelman, W.N., Wistuba, I.I. and Kurie, J.M. 2005. Inhibition of mammalian target of rapamycin reverses alveolar epithelial neoplasia induced by oncogenic K-Ras. Cancer Res. 65(8), 3226–3235. Yamani, H.A., Pang, E.C., Mantri, N. and Deighton, M.A. 2016. Antimicrobial activity of Tulsi (Ocimum tenuiflorum) essential oil and their major constituents against three species of bacteria. Front. Microbiol. 7, 681. Ye, L., He, M., Huang, Y., Lei, Y., Zhou, Y. and Chen, X. 2016. Tree shrew as a new animal model for the study of lung cancer. Oncol. Lett. 11(3), 2091–2095. Yin, X., Cong, X., Yan, M. and Zhang, D.E. 2009. Deficiency of a potential 3p21.3 tumor suppressor gene UBE1L (UBA7) does not accelerate lung cancer development in K-rasLA2 mice. Lung Cancer 63(2), 194–200. Yu, S.J., Zhang, Y., Pan, Y., Cheng, C., Sun, Y. and Chen, H. 2017. The non-small cell lung cancer EGFR extracellular domain mutation, M277E, is oncogenic and drug-sensitive. Onco. Targets Ther. 10, 4507–4515. Yuan, B.Z., Jefferson, A.M., Baldwin, K.T., Thorgeirsson, S.S., Popescu, N.C. and Reynolds, S.H. 2003. DLC-1 operates as a tumor suppressor gene in human non-small cell lung carcinomas. Oncogene 23(7), 1405–1411. Yuan, J.M., Koh, W.P., Murphy, S.E., Fan, Y., Wang, R., Carmella, S.G., Han, S., Wickham, K., Gao, Y.T., Yu, M.C. and Hecht, S.S. 2009. Urinary levels of tobacco-specific nitrosamine metabolites in relation to lung cancer development in two prospective cohorts of cigarette smokers. Cancer Res. 69(7), 2990–2995. Zhao, B., Magdaleno, S., Chua, S., Wang, Y.L., Burcin, M., Elberg, D., Finegold, M., Tsai, S. and Demayo, F.J. 2000. Transgenic mouse models for lung cancer. Exp. Lung Res. 26(8), 567–79. Zhao, L., Ni, X., Zhao, L., Zhang, Y., Jin, D., Yin, W., Wang, D. and Zhang, W. 2018. MiroRNA-188 acts as tumor suppressor in non-small-cell lung cancer by targeting MAP3K3. Mol. Pharm. 15(4), 1682–1689. Zhou, F., Li, S., Jia, W., Lv, G., Chen, S., Kang, C. and Zhang, Q. 2015. Effects of diesel exhaust particles on microRNA-21 in human bronchial epithelial cells and potential carcinogenic mechanisms. Mol. Med. Rep. 12(2), 2329–2335. Zhu, H., Chen, A., Li, S., Tao, X., Sheng, B., Chetry, M. and Zhu, X. 2017. Predictive role of galectin-1 and integrin α5β1 in cisplatin-based neoadjuvant chemotherapy of bulky squamous cervical cancer. Biosci. Rep. 37(5), BSR20170958. Zuazo-Gaztelu, I. and Casanovas, O. 2018. Unraveling the role of angiogenesis in cancer ecosystems. Front. Oncol. 8, 248. | ||
| How to Cite this Article |
| Pubmed Style Kustiati U, Nugrahaningsih DAA, Kusindarta DL, Wihadmadyatami H. Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancer. Open Vet. J.. 2025; 15(2): 482-503. doi:10.5455/OVJ.2025.v15.i2.1 Web Style Kustiati U, Nugrahaningsih DAA, Kusindarta DL, Wihadmadyatami H. Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancer. https://www.openveterinaryjournal.com/?mno=217818 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.1 AMA (American Medical Association) Style Kustiati U, Nugrahaningsih DAA, Kusindarta DL, Wihadmadyatami H. Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancer. Open Vet. J.. 2025; 15(2): 482-503. doi:10.5455/OVJ.2025.v15.i2.1 Vancouver/ICMJE Style Kustiati U, Nugrahaningsih DAA, Kusindarta DL, Wihadmadyatami H. Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancer. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 482-503. doi:10.5455/OVJ.2025.v15.i2.1 Harvard Style Kustiati, U., Nugrahaningsih, . D. A. A., Kusindarta, . D. L. & Wihadmadyatami, . H. (2025) Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancer. Open Vet. J., 15 (2), 482-503. doi:10.5455/OVJ.2025.v15.i2.1 Turabian Style Kustiati, Ulayatul, Dwi Aris Agung Nugrahaningsih, Dwi Liliek Kusindarta, and Hevi Wihadmadyatami. 2025. Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancer. Open Veterinary Journal, 15 (2), 482-503. doi:10.5455/OVJ.2025.v15.i2.1 Chicago Style Kustiati, Ulayatul, Dwi Aris Agung Nugrahaningsih, Dwi Liliek Kusindarta, and Hevi Wihadmadyatami. "Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancer." Open Veterinary Journal 15 (2025), 482-503. doi:10.5455/OVJ.2025.v15.i2.1 MLA (The Modern Language Association) Style Kustiati, Ulayatul, Dwi Aris Agung Nugrahaningsih, Dwi Liliek Kusindarta, and Hevi Wihadmadyatami. "Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancer." Open Veterinary Journal 15.2 (2025), 482-503. Print. doi:10.5455/OVJ.2025.v15.i2.1 APA (American Psychological Association) Style Kustiati, U., Nugrahaningsih, . D. A. A., Kusindarta, . D. L. & Wihadmadyatami, . H. (2025) Lung cancer: Animal model of lung cancer, molecular carcinogenesis of lung cancer, and antitumor effect of Ocimum sanctum against lung cancer. Open Veterinary Journal, 15 (2), 482-503. doi:10.5455/OVJ.2025.v15.i2.1 |