| Research Article | ||
Open Vet. J.. 2025; 15(2): 646-659 Open Veterinary Journal, (2025), Vol. 15(2): 646-659 Research Article The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authenticationDwi Lestari1*, Hasyrul Hamzah1, Asri Dwi Endah Dewi Pramesthi1, Eka Siswanto Syamsul2, Putri Dela Safitri1, Rika Nurjunnah1 and Abdul Rohman31Faculty of Pharmacy, Universitas Muhammadiyah Kalimantan Timur, Samarinda, Indonesia 2Sekolah Tinggi Ilmu Kesehatan Samarinda, Samarinda, Indonesia 3Faculty of Pharmacy, Universitas Gadjah Mada, Yogyakarta, Indonesia * Correspondence Author: Dwi Lestari. Faculty of Pharmacy, Universitas Muhammadiyah Kalimantan Timur, Samarinda, Indonesia. dl792 [at] umkt.ac.id Submitted: 29/09/2024 Accepted: 31/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
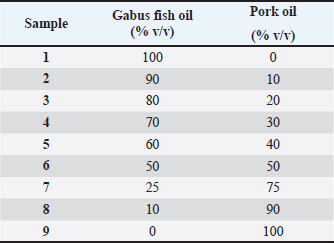
AbstractBackground: The muslim population is very concerned about halal food. Nowadays, there is a growing awareness among consumers regarding the adulteration of food. High-quality Gabus fish oil (halal) is very susceptible to being adulterated with Pork oil (non-halal) by unethical producers to gain greater profits. Aim: The research objective was to use Fourier Transform Infrared-Attenuated Total Reflection (FTIR-ATR) spectroscopy and gas chromatography–mass spectrometry (GC-MS) in combination with chemometrics for the analysis of pork oil adulteration in Gabus fish oil. Methods: Extraction of Gabus fish oil using the pressing method and pork oil using the soxhlet method. The oil components extracted were then analyzed using FTIR-ATR spectroscopy combined with chemometrics of linear discriminant analysis (LDA) and multivariate calibrations of partial least square (PLS) and principle component regression (PCR) using optimized conditions. The GC-MS data from methyl ester were processed using chemometrics principal component analysis (PCA) to group Gabus fish oil, pork oil, and palm oil. Results: The absorbance values at wavenumber regions of 1,500–1,000 cm–1 were selected for discrimination between Gabus fish oil and Gabus fish oil adulterated with pork oil using chemometrics of LDA. The LDA applied to the same wavenumber regions used in the quantitative analysis successfully classified Gabus fish oil, pork oil, and a Gabus-pork oil mixture with an accuracy of 100%. The prediction of pork oil was successfully determined using multivariate calibrations of PLS and PCR using optimized conditions. There are three fatty acid markers found in Gabus fish oil caprylic acid, pentadecanoic acid and arachidic acid. The PCA was applied for data GC-MS interpretation. An analysis by PCA was able to cluster and discriminate Gabus fish oil, pork oil, and palm oil. Conclusion: FTIR-ATR spectroscopy and GC-MS coupled with chemometrics is a rapid and accurate method for detecting and quantifying pork oil in Gabus fish oil for halal authentication. Keywords: Halal authentication, Gabus fish oil, FTIR spectroscopy, GC-MS, Chemometrics IntroductionHalal comes from Arabic and is a term used to describe any product that is permitted or allowed based on Sharia or Islamic law; these halal products include food, cosmetics, and medicines (Ahmad et al., 2018). Halal pharmaceuticals are a basic human need that must be considered because they can overcome health problems or disorders, maintain body resistance, and optimize the body’s metabolic processes. Therefore, the presence of halal products containing non-halal ingredients presents a significant issue within society (Subara and Jaswir, 2018; Nekha, 2024). Recently, the adulteration of food products, such as fats and oils, has surfaced as a significant issue motivated by the pursuit of increased profits. An instance is the adulteration of fish oil, which includes omega-3 fatty acids (Sakamoto et al., 2019). Omega-3 fatty acids serve as essential components of cell membranes (Cholewski et al., 2018). Several epidemiological and clinical studies have shown that omega-3 fatty acids are favorable for human health. Omega-3 fatty acids in fish oil confer health benefits such as primary and secondary protection against cardiovascular disease (Siscovick et al., 2017); the avoidance of cognitive impairment associated with Alzheimer’s disease may diminish the risk of dementia (Song et al., 2016; Putri et al., 2019a) and possess anti-inflammatory and antioxidant properties (Syifa et al., 2022). Gabus fish oil (Channa striata) is a vulnerable supplement to adulteration practices (Irnawati et al., 2021). This can involve adding or substituting high-quality oils, such as Gabus fish oil, with cheaper oils, such as pork oil (Windarsih et al., 2023). Gabus fish oil is rich in essential oils, composed of omega-3 and omega-6, which the human body cannot produce independently. Furthermore, fish oil has large amounts of vitamins A and D (Pasaribu et al., 2020). The fatty acid in Gabus fish oil is including C22:6, C20:4, C18:1, C18:0, and C16:0 (Nabila et al., 2020). Gabus fish oil contains polyunsaturated fatty acids (PUFA) and monounsaturated fatty acids (MUFA) those essential for brain and body growth and development (Rohman et al., 2021). Various analytical methods, including nano-real-time principle component regression (PCR), have been reported for authenticating oil. These methods use both simple and advanced instruments (He et al., 2013), nuclear magnetic resonance spectroscopy (Giese et al., 2019), liquid chromatography electrospray tandem ion-trap mass spectrometry (Zeng et al., 2010), gas chromatography flame ionization detector (Alinafiah et al., 2021), gas chromatography–mass spectrometry (Brotas et al., 2020), high-performance liquid chromatography–mass spectrometry (Suh et al., 2017), near-infrared spectroscopy (Cascant et al., 2018), and Raman spectroscopy (Killeen et al., 2017). A rapid, safe, also accurate approach is required for halal authenticity because most analytical techniques for fatty acid analysis in fish oil require energy, time, and hazardous chemicals. FTIR spectroscopy is a vibrational spectroscopic technique used for fingerprint analysis. It has several advantages, including fast analysis, minimal sample preparation, suitability for various types of samples (liquid and solid), and use of fewer solvents, supporting green chemistry (Lestari et al., 2022). FTIR spectroscopy is extensively used for the analysis of authentication and quality control of vegetable oils, such as fish oil. The analysis method using FTIR spectroscopy in concurrence with multivariate analysis chemometrics will serve as an effective application to authenticate the fish oil (Lestari et al., 2024). Chemometrics integrates statistical and mathematical methodologies for the analysis of chemical data (Irnawati et al., 2023). FTIR spectroscopy coupled with multivariate analysis or chemometrics is commonly used for authenticating vegetable oils in binary combinations, including the authenticity of olive oil with numerous vegetable oils (Lerma-García et al., 2010), the authentication of Patin fish oil (Putri et al., 2019b), and detecting the red fruit oil adulteration (Rohman et al., 2014), sesame adulterated with corn oil analysis (Fadzlillah et al., 2014), and virgin coconut oil with canola oil authentication (Kiyat et al., 2013). Another method for identifying adulterated fish oil is to examine its fatty acid composition. This method involves converting fatty acids into ester derivatives and then analyzed using gas chromatography–mass spectrometry (GC-MS). The GC-MS method has advantages, including not requiring a standard sample for analysis, being more sensitive, and not making it difficult to read the analysis results if there is noise in the analysis (Lestari et al., 2023). The GC-MS analysis in combination with chemometrics principal component analysis (PCA) seeks to minimize the variable numbers while preserving the information inherent in the initial data (Salamah et al., 2022). FTIR spectroscopy and GC-MS have distinct advantages regarding cost and accessibility in analyzing oils. FTIR is a more cost-effective option with lower initial investment and operational costs while offering high sample throughput and ease of use. GC-MS provides detailed compositional data at a lower cost than LC-MS. Whereas High Performance Liquid Chromatography (HPLC) is quite effective and more economical, but this technique provides less detailed analysis, especially in identifying complex compounds or small quantities than spectroscopic techniques. Methodologies should be guided by specific analytical needs, budget constraints, and laboratory expertise. For oil analyses where rapid and cost are critical factors, FTIR may be preferred, and GC-MS for detailed compositional studies where precision is essential (Syafri et al., 2022; Lestari et al., 2024). This study was conducted to detect the adulteration of Gabus fish oil mixed with pork oil using Fourier Transform Infrared-Attenuated Total Reflection (FTIR-ATR) spectroscopy, which is capable of acting as a fingerprint technique, and analysis using a gas chromatography–mass spectrometer (GC-MS) to confirm the fatty acids contained and as a specific marker of fatty acids in Gabus fish oil (C. striata). Methods and MaterialsGabus fish (C. striata) was obtained from the Ijabah market of Samarinda, Kalimantan Timur, Indonesia. The pork was collected from the Dayak market of Samarinda, Kalimantan Timur, Indonesia. Analytical grade solvents and other reagents were employed in the study (Merck). Extraction of Gabus fish oilThe extraction of Gabus fish oil was conducted using the pressing method with a slightly modified (Bako et al., 2017; Syamsul et al., 2024). The Gabus fish sample was encased in filter cloth, positioned within a press machine column, and subjected to a pressure of 100 kN for 2 minutes. The extracted oil was gathered in the flask. To eliminate water content and other contaminants, 10 g of Gabus fish oil was combined with 0.3 g of bentonite and 1 g of anhydrous sodium sulfate in a flask as a purification technique. Bentonite is used as an adsorbent in the fish oil purification process because of its ability to bind and absorb impurities such as free fatty acids, peroxide numbers, and other compounds contained in crude fish oil (Syamsul et al., 2024). If suspended particles remain in the extracted fish oil, they are removed using centrifugation at 5,000 rpm for 10 minutes. The upper phase, consisting of pure oil, was transferred to a new container, and a measured quantity of this pure oil was deposited in a separate, fresh container (Syifa et al., 2022). Acid hydrolysisThe 20 g of pork was hydrolyzed using 200 ml of 1 N hydrochloric acid. The sample was then cooked in a water bath at 60–65°C for 15–25 minutes before filtering. Acid hydrolysis may enhance extraction efficiency by liberating bound lipids attached to proteins and carbohydrates (Lestari et al., 2024). Extraction of pork oilSoxhlet extraction method: The extraction was conducted for 8 hours at 100°C (±50 cycles). A 50 g sample of pork after hydrolyzed acid was wrapped in filter paper and placed in the Soxhlet apparatus. A total of 438 ml of petroleum ether was used as the extracting solvent. To remove the water content, the lipid extract was combined with anhydrous sodium sulfate, stirred, and filtered using Whatman filter paper. Additionally, it was evaporated using a vacuum rotary evaporator at 40°C. A lipid extract was obtained and heated in a water bath until it was transformed into pork oil. (Lestari et al., 2022). Preparation of fatty acid methyl ester (FAME)Derivatization of fatty acids to FAME with slight modification method: Each sample consisted of 5–20 mg of Gabus fish oil, and pork oil was added with 1 ml of BF-methanol 10%, then heated in an ultrasonic bath, temperature 60°C, for 20 minutes, until cool. Furthermore, 2 mL of n-hexane is added, vortexed, and centrifuged at 4000 rpm for 10 minutes. The upper layer (n-hexane) is then collected, transferred into a collection vial, and injected into the GC-MS for analysis (Christie, 1993). Preparation of calibration and validation samplesGabus fish and pork oil were used as calibration and validation standards to observe the difference in lipid spectra at concentrations of 0%, 10%, 20%, 30%, 40%, 50%, 75%, 90%, and 100%. FTIR spectroscopySamples were analyzed using an Fourier Transform Infrared (FTIR) spectrometer (Thermo Scientific Nicolet iS10, Madison, WI), operated using Omnic software. The measurements were conducted in the mid-infrared region of 4000–650 cm-¹, using 32 scans at a resolution of 16 cm-¹. Background scanning was necessary to reduce the effect of the air’s reference spectrum. The data obtained were managed using the Total Quality (TQ) Analyst software. GC-MSOne µl of derivatized fatty acids was injected into the GC-MS Agilent (type: 8890 GC System, 5977B GC/MSD, 7693A Autosampler). The separation was carried out in a DB-FastFAME; 30 m x 250 µm x 0,25 µm column, oven profile is 60°C/minute rise 30°C/ minute to 150°C for 1 minute, increase 2°C/minute to 200°C for 5 minutes, increase 3°C/minute to 225°C for 5 minutes, injector temperature of 250ᵒC, the mobile phase flow rate is 1 ml/minute. The carrier gas is helium. ChemometricsMultivariate calibration and discriminant analysis for FTIR spectroscopy were conducted using TQ Analyst software version 9 (Thermo Fisher Scientific, Inc.). Multivariate calibrations employed for quantification were partial least squares (PLS) and PCR. Data analysis chemometrics for GC-MS employing PCA with SIMCA software. ResultsGabus fish oil and pork oil extractionFish oil is obtained using a pressing method without involving solvents, and pork oil is obtained using the Soxhlet method with slight modifications to produce quality oil. The oil produced from Gabus fish and pork exhibits a yellowish color. As a consequence, it is difficult to differentiate between pure Gabus fish oil and adulterated fish oil by visual inspection. Table 1 shows the adulteration of Gabus fish oil mixed with pork oil at various concentrations. Table 1. The binary mixtures of Gabus fish oil and pork oil used for FTIR analysis.
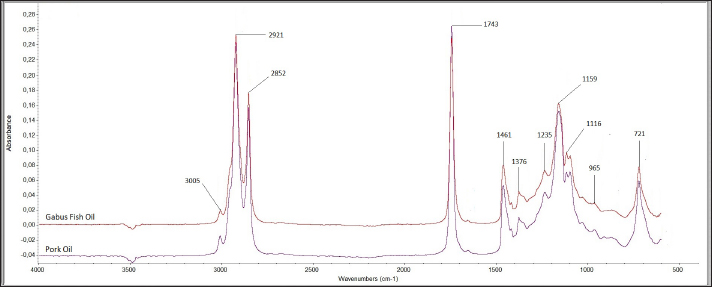
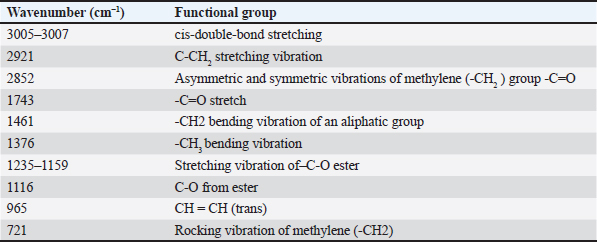
FTIR spectra analysisFTIR spectroscopy provides fingerprint spectra specific for Gabus fish oil, pork oil, and a mixture of Gabus fish oil and pork oil samples. The compounds resulting from the FTIR vibrations of the oil samples consisted mainly of triacylglycerols, triglycerides, and fatty acids. The spectra of Gabus fish oil 100% and pork oil 100% demonstrated almost the same pattern. The spectra measured at the wavenumber ranging from 4,000 to 600 cm–1 are depicted in Figure 1. Each shoulder and peak correspond to functional groups responsible for the absorption of infrared radiation. Notable distinctions can be observed in both spectra within the wavenumber range of 1,500–1,000 cm–1, particularly in the fingerprint region. The peak at about 3,005–3,007 cm–1 is due to the C-H strain vibration at = C-H cis. The –CH2 functional group peaks at 2,921 cm–1 and 2,852 cm–1, respectively, due to asymmetric and symmetrical vibrations. A peak indicates the carbonyl group (C = O) of the triglyceride ester at 1,743 cm–1. The absorption of carbonyl (C = O) ester bonds was observed at a frequency of 1,743 cm–1 with strong intensity because the electronegativity of the hydrogen and carbon atoms differs greatly. The bending vibrations of methylene and methyl groups are evident in the 1,461 cm–1 and 1,376 cm–1 regions. The vibrations of overlapping methylene shaking and out-of-plane bending of cissubstituted olefins give rise to the bands at 1,235, 1,159, 1,116, 965, and 721 cm–1.
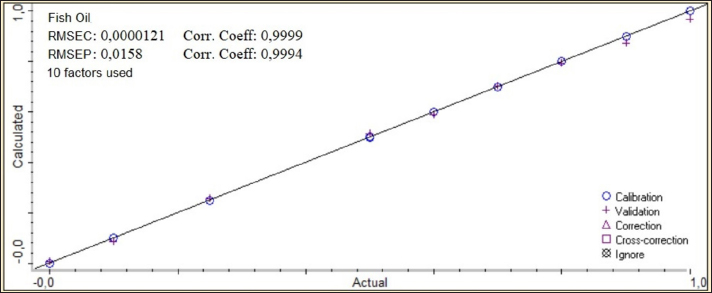
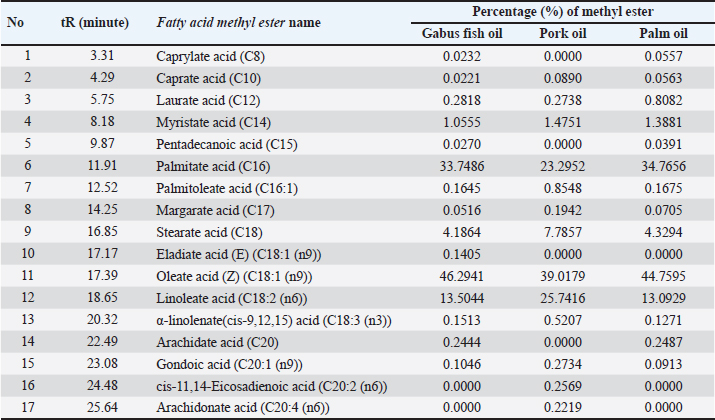
Fig. 1 FTIR spectra of the lipid extracted from Gabus fish oil and pork oil at a concentration of 100%. GC-MS analysisDerivatization changes fatty acid compounds in oil (non-volatile) converted into FAME volatile compounds. Derivatization was carried out on Gabus fish, pork, and palm oil, respectively. A total of 17 compounds are found in Gabus fish, pork, and palm oil, respectively. Three fatty acid compounds, caprylate acid (C8), pentadecanoic acid (C15), and arachidonate acid (C20), were found in Gabus fish oil but not in pork oil. Derivatization was carried out on Gabus fish, pork, and palm oil, respectively. There are three fatty acid compounds found in Gabus fish oil but are not found in pork oil, namely caprytic acid (C8), pentadecanoic acid (C15), and Arachidonate (C20) acid. While the fatty acids contained in pork oil are CIS-11,14-eicosadienoic acid (C20: 2 (N6)) and Arachidonate Acid (C20: 4 (N6)) and the two fatty acids are not found in Gabus fish oil and oil palm oil. ChemometricsThe class membership of unknown samples (Gabus fish oil, pork oil, and a combination of both) is predicted using linear discriminant analysis (LDA). The lipid components extracted by Gabus fish oil, pork oil, and a combination of both were distinguished using the absorbance values in wavenumber ranges of 1,500–1,000 cm–1. The LDA successfully distinguished between Gabus fish oil, pork oil, and Gabus–pork oil mixture. The quantitative analysis of oil adulteration using two multivariate calibrations of PLS and PCR. Based on the optimization, PLS using the normal spectrum in the wavenumber region 1,500–1,000 cm–1 provides the best model, with an R2 calibration value of 0.9999, Root Mean Square Error of Calibration (RMSEC) value of 0.0000121, R2 validation value of 0.9994, and Root Mean Square Error of Prediction (RMSEP) value of 0.0158. PCA is a sophisticated analytical technique for constructing multivariate linear models from complex datasets. According to the PCA chemometric data, the fatty acid profile of Gabus fish oil was in a different quadrant than that of pork oil and palm oil on the fatty acid score plot. DiscussionGabus fish oil and pork oil extractionThe pressing method used to extract Gabus fish oil offers numerous benefits. This technique preserves the natural omega-3 fatty acids, vitamins (such as A and D), and antioxidants found in the fish oil. Unlike other extraction methods, pressing does not subject the oil to high temperatures, which can degrade delicate compounds like omega-3. The pressing method is ecofriendly that does not require chemical solvents (Ivanovs and Blumberga, 2017). The selection of pork oil as an oil adulterant depended to three reasons: first, with the fact that the price of pork oil is cheaper than Gabus fish oil (Putri et al., 2019a); second, the similarity of Gabus fish oil and pork oil in the FTIR spectra and score plot of the first principle component and second principle component through the PCA (Irnawati et al., 2021); and third, for the reason of halal authentication (Irnawati et al., 2023). FTIR spectra analysis and chemometricsThe FTIR spectrum produces a distinctive fingerprint pattern. Each fatty acid does not have the same spectrum regarding the peak number, intensity, or wavenumber at each peak, so FTIR spectroscopy can extract differences between fatty acids (Pebriana et al., 2017). Figure 1 shows that Gabus fish oil and pork oil have similar spectrum patterns, namely typical spectra that indicate the presence of triglycerides (fats). Both are compounds rich in glycerol and fatty acids. Triglycerides are the main components with a common chemical structure in all edible fats and oils (Rohman, 2019). Gabus fish oil and pork oil are included in the edible oil group because both oils consist of fatty acids such as saturated, MUFA, and PUFA, although the proportions and types of fatty acids may differ (Rohman et al., 2012). The functional groups and vibration models of Gabus fish and pork oil responsible for IR absorption are shown in Table 2. Figure 2 demonstrates the representative spectra of fatty acids acquired from the extraction of Gabus fish oil and pork oil with different concentrations. The spectra are physically similar when observed using the unaided eyes and reveal the general characteristics of the absorption bands for triglycerides, the main component of which is contained in fatty acids. Table 2. The functional groups and vibration modes from Gabus fish oil and pork oil.
Fig. 2. FTIR spectra of the lipid extracted from Gabus fish oil and pork oil at a concentration of 0–100%. The LDA is a supervised learning technique that exploits class labels and aims to maximize separation between classes while reducing dimensionality. The LDA points to finding a linear combination of features that escalates separation between classes in the data while minimizing variation within the same class (Miller et al., 2018). In this study, LDA, known as a supervised pattern recognition method, is used to predict the class representative samples of Gabus fish oil, pork oil, and Gabus–pork oil mixture using FTIR spectra measurements at specific wavenumber regions as variables. The fingerprint technique approach to halal authentication of Gabus fish oil is used in the differences between the spectra seen in the shift in the absorption band (wavenumber) (Arifah et al., 2022). This grouping is based on the absorbance value of each peak of the lipid spectrum at a certain wavenumber region. The absorbance is then converted into Mahalanobis distance and used as a grouping variable for Gabus fish oil, pork oil, and Gabus–pork oil mixture to form a Cooman’s plot at the optimal wavenumber (Lestari et al., 2022). Figure 3 clearly indicates that both groups are separated, without observing classification objects. The wavenumber area between 1,500 and 1,000 cm–1 is a characteristic that distinguishes the fatty acids spectra of Gabus fish oil and pork oil.
Fig. 3. The Cooman’s plot for discriminant analysis from 100% Gabus fish oil (green), 100% pork oil (blue), and Gabus–pork oil mixture at different concentrations (red). Multivariate PLS and PCR calibration are the most frequently used quantitative analysis techniques in chemometrics. The statistical parameters used as the criteria for the best multivariate calibration model are a high coefficient of determination (R2) between the actual value and the predicted value of FTIR for accuracy evaluation and a low RMSEC value, RMSEP for precision evaluation. The selection of FTIR spectra conditions is optimized at certain wave numbers to produce a coefficient of determination (R2) value close to 1 and RMSEC and RMSEP values close to 0 (Rahayu et al., 2018). The PLS and PCR analysis from lipid components extracted from Gabus fish oil and pork oil is shown in Table 3. The optimization analysis of Gabus fish oil with the PLS method from 1,500–1,000 cm–1 wavelength using the normal spectra with R2 calibrated and predicted is 0.9999 and 0.9994, respectively, and also RMSEC and RMSEP are 0.0000121 and 0.0158, respectively. Figure 4 shows the correlation between the actual value (X-axis) and the predicted FTIR value (Y-axis) of Gabus fish oil and pork oil. The figure explains the linear correlation between the actual value and the predicted FTIR value resulting in a high R2 value approaching 1 (Riyanta et al., 2020). The low RMSEC and RMSEP values indicate that the model formed is getting better because of the low error. These results are in by previous studies on the quantification of Gabus fish oil, palm oil, and corn oil, where the best wave number for quantification was obtained, namely 3,200–600 cm–1 with an R2 value of 0.9972, RMSEC 0.014 for calibration, and R2 0.9818 RMSEP 0.036 for prediction (Irnawati et al., 2021). Table 3. The optimization of multivariate calibrations of partial least square regression (PLS) and principle component regression (PCR) as well as wavenumber regions for quantitative analysis of Gabus fish oil, pork oil, and mixed Gabus–pork oil.
Fig. 4. The correlation between the actual value and FTIR predicted values is facilitated by partial least squares calibrations. GC-MS analysis and chemometricsThe GC-MS approach is used to analyze methyl esters from the derivatization of pork oil and Gabus fish oil. This device integrates gas chromatography with a mass spectrometer detector (Nurjuliana et al., 2011). Gas chromatography (GC) is highly effective in separating volatile components in a mixture or those that may be transformed into volatiles, such as FAMEs. Esterification is used in fatty acid analysis to transform fatty acids into a volatile state suitable for GC analysis (Lestari et al., 2023). The aim of GC is the detection and quantification of various types of fatty acids in a sample, even if their concentrations are very low. Mass spectrometry (MS) provides the ability to identify compounds based on their molecular mass and the fragment ions produced during ionization. Each fatty acid has a unique mass spectrum, allowing for highly accurate identification of the different types of fatty acids in a sample. The combination of GC for separation and MS for identification provides a powerful ability to identify fatty acid components, including isomers that may be difficult to distinguish from other techniques (Guntarti et al., 2020). Gas chromatography is used to separate fatty acid content in the form of methyl esters as shown in Figure 5. The results of the mass spectrometer of Gabus fish oil can be seen in Figure 6. Palm oil methyl esters are also analyzed to see the differences in fatty acids found in animals and plants. Figure 6 shows that there are three fatty acids found in Gabus fish oil but do not exist in pork oil, specifically caprylate acid, pentadecanoic acid, and arachidate acid. Figure 7 shows that there are two fatty acids found in pork oil but do not exist in Gabus fish oil or palm oil, specifically cis-11,14-Eicosadienoic acid and arachidonate acid. This shows that there is a difference in the type of methyl ester contained between pork oil (non-halal) and Gabus fish oil and palm oil (halal). The composition of methyl esters can be seen in Table 4 which explains the retention time, type of methyl ester found in Gabus fish oil, pork oil and palm oil, and the category of fatty acids. This is similar to previous research regarding GC-MS for the evaluation of fish oil authenticity. The fatty acid consisted of 25 fish oil samples was examined using gas chromatography with a single quadrupole detector. Derivatizations were conducted before GC-MS analysis by generating FAME using Boron trifluoride (BF3) (Abdulkadir and Tsuchiya, 2008).
Fig. 5. Chromatogram of gas chromatography from Gabus fish oil and pork oil.
Fig. 6. Spectrogram of mass spectrometry from Gabus fish oil; caprylate acid, pentadecanoic acid, and arachidate acid.
Fig. 7. Spectrogram of mass spectrometry from pork oil; cis-11,14-Eicosadienoic acid and arachidonate acid. Table 4. The composition of methyl ester content in Gabus fish oil, pork oil, and palm oil.
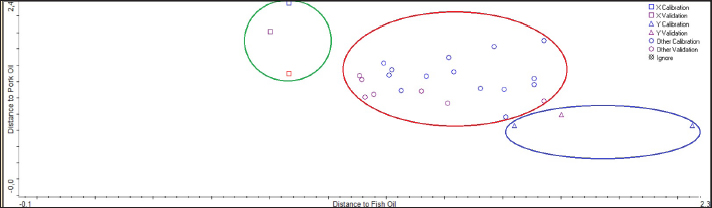
PCA is an unsupervised learning technique for reducing dimensionality based on the overall variability of the data without considering class labels. PCA looks for the direction of the greatest variance in the data in general (Miller et al., 2018). The principal component analysis is very useful for analyzing the pattern visualization and correlation. The matrix is the kind of interaction between the animal and the fatty acids. PCA can show each animal’s fat’s fatty acid concentration and distribution, both in similarities and differences. Data on the types of fatty acids present in different animals were variables for the PCA (Miller et al., 2018). Figure 8 shows the PCA scores plot generated by SIMCA. The classification performance results from the SIMCA model are used to distinguish between Gabus fish oil, pork oil, and palm oil. The SIMCA model was constructed and optimized. The data in the different quadrants, if they are in the same quadrants, show similarities in the physical–chemical properties of the fatty acid contents.
Fig. 8. Chemometric principal component analysis analysis of Gabus fish oil, pork oil, and palm oil fatty acid profiles. The potential for interference in FTIR–ATR spectroscopy and GC-MS measurements must be considered. The interference that may arise is as follows: first, in terms of sample quality such as the presence of other components such as water or solvents so that we need purification; second, data interpretation problems such as producing overlapping peaks; and third, problems in instrumentation such as inadequate instrument resolution can cause difficulties in identifying small peaks. These interferences must be avoided in order to obtain accurate results both qualitatively and quantitatively (Khan et al., 2016; Syafri et al., 2022). ConclusionThe analysis for halal authentication of Gabus fish oil adulterated with pork oil is very important to ensure product quality and consumer protection. FTIR spectroscopy, which can be used as a fingerprint, is a rapid and authentic analysis technique for the authentication of Gabus fish oil. The combination with LDA chemometrics can be used to distinguish Gabus fish oil and pork oil and can classify Gabus fish oil (halal product) and Gabus–pork oil mixture (non-halal) with 100% accuracy. Meanwhile, PLS and PCR multivariate calibration offers a fast and reliable method to authenticate Gabus fish oil from pork oil by applying FTIR spectral absorbance as a variable with acceptable accuracy and precision. The gas chromatography–mass spectroscopy method can be used to authenticate Gabus fish oil with fatty acid content. Two specific fatty acids in pork oil do not exist in Gabus fish oil and palm oil, specifically cis-11,14-Eicosadienoic acid and arachidonate acid. Chemometrics PCA Gabus fish oil, pork oil, and fat can be grouped. FTIR spectroscopy and GC-MS methods can be used for halal authentication and rapid detection of adulteration of pork oil in Gabus fish oil. Further research can be carried out using market samples or unknown samples based on the chemometric results in this study and determine the characterization of Gabus fish oil. AcknowledgmentsThe authors thank Kementerian Pendidikan, Kebudayaan, Riset dan Teknologi, Hibah Penelitian Dosen Pemula 2024. Conflict of interestThe authors state no conflict of interest. FundingPDP DIKTI Contract number: 113/E5/PG.02.00. PL/2024, 11 Juni 2024; LLDIKTI XI Kalimantan Contract number: 61/LL11/KM/20244, 19 Juni 2024; LPPM Contract number: 167.3/LPPM/A.4.C/2024, 20 Juni 2024. Authors’ contributionsDL: Planned the study and drafted and revised the manuscript. HH, ADEDP, ESS, PDS, RN and AR: Performed the extraction and analyzed the data. All authors have read, reviewed, and approved the final manuscript. Data availabilityAll data supporting the findings of this study are available within the manuscript and no additional data sources are required. ReferencesAbdulkadir, S. and Tsuchiya, M. 2008. One-step method for quantitative and qualitative analysis of fatty acids in marine animal samples. J. Exp. Mar. Bio. Ecol. 354(1), 1–8. Ahmad, A.N., Ungku Zainal Abidin, U.F, Othman, M. and Abdul Rahman, R. 2018. Overview of the halal food control system in Malaysia. Food Control 90, 352–363. Alinafiah, S.M., Azlan, A., Ismail, A. and Rashid, N.K.M.A. 2021. Method development and validation for omega-3 fatty acids (Dha and epa) in fish using gas chromatography with flame ionization detection (gc-fid). Molecules 26(21), 6592. Arifah, M.F., Irnawati, Ruslin, Nisa, K., Windarsih, A. and Rohman, A. 2022. The application of FTIR spectroscopy and chemometrics for the authentication analysis of horse milk. Int. J. Food Sci. 2022, 7643959. Bako, T., Umogbai, V.I. and Awulu, J.O. 2017. Criteria for the extraction of fish oil. Agric. Eng. Int. CIGR J. 19(3), 120–132. Brotas, M.S.C., Carvalho, G.A. and Pereira, P.A.P. 2020. Determination, through derivatization and GC-MS analysis, of Omega-3 and Omega-6 fatty acids in fish oil capsules sold in Salvador, Bahia. J. Braz. Chem. Soc. 31(3), 447–455. Cascant, M.M., Breil, C., Fabiano-Tixier, A.S., Chemat, F., Garrigues, S. and de la Guardia, M. 2018. Determination of fatty acids and lipid classes in salmon oil by near infrared spectroscopy. Food Chem. 239, 865–871. Cholewski, M., Tomczykowa, M. and Tomczyk, M. 2018. A comprehensive review of chemistry, sources and bioavailability of omega-3 fatty acids. Nutrients 10(11), 1662. Christie, W.W. 1993. Preparation of ester derivatives of fatty acids for chromatographic analysis. Adv. Lipid Methodol. 2–43. Fadzlillah, N.A., Che Man, Y.B. and Rohman, A. 2014. FTIR spectroscopy combined with chemometric for analysis of sesame oil adulterated with corn oil. Int. J. Food Prop. 17(6), 1275–1282. Giese, E., Rohn, S. and Fritsche, J. 2019. Chemometric tools for the authentication of cod liver oil based on nuclear magnetic resonance and infrared spectroscopy data. Anal. Bioanal. Chem. 411(26), 6931–6942. Guntarti, A., Gandjar, I.G. and Jannah, N.M. 2020. Authentication of wistar rat fats with gas chromatography mass spectometry combined by chemometrics. Potravin. Slovak J. Food Sci. 14, 52–57. He, J., Xu, W., Shang, Y., Zhu, P., Mei, X., Tian, W. and Huang, K. 2013. Development and optimization of an efficient method to detect the authenticity of edible oils. Food Control 31(1), 71–79. Irnawati, Riyanto, S. and Rohman, A. 2021. Adulteration of Gabus (Channa striata) fish oil with corn oil and palm oil: the use of FTIR spectra and chemometrics. Food Res. 5(2), 184–190. Irnawati, I., Windarsih, A., Indrianingsih, A.W., Apriyana, W., Ratnawati, Y.A., Nadia, L.O.M.H. and Rohman, A. 2023. Rapid detection of tuna fish oil adulteration using FTIR-ATR spectroscopy and chemometrics for halal authentication. J. Appl. Pharm. Sci. 13(4), 231–239. Ivanovs, K. and Blumberga, D. 2017. Extraction of fish oil using green extraction methods: a short review. Energy Procedia. 128, 477–483. Khan, Y., Ansari, H., Rinki, Chauhan, R., Tamboli, E. and Ahmad, S. 2016. Comparative gas chromatography-mass spectroscopy, Fourier transform infrared spectroscopy, and high-performance liquid chromatography analysis of essential oils extracted using 4 methods from the leaves of Eucalyptus globulus L. L. Drug Dev. Ther. 7(2), 81. Killeen, D.P., Marshall, S.N., Burgess, E.J., Gordon, K.C. and Perry, N.B. 2017. Raman spectroscopy of fish oil capsules: polyunsaturated fatty acid quantitation plus detection of ethyl esters and oxidation. J. Agric. Food Chem. 65(17), 3551–3558. Kiyat, W. El, Monica, A., Qomariyah, N. and Manurung, B.S. 2013. Analysis of Canola Oil in Virgin Coconut Oil Using FTIR Spectroscopy and Chemometrics. J. Food Pharm. Sci. 1(1), 1–6. Lerma-García, M.J., Ramis-Ramos, G., Herrero-Martínez, J.M. and Simó-Alfonso, E.F. 2010. Authentication of extra virgin olive oils by Fourier-transform infrared spectroscopy. Food Chem. 118(1), 78–83. Lestari, D., Rohman, A., Syofyan, S., Yuliana, N.D., Bt, K., Bakar, A. and Hamidi, D. 2022. Analysis of beef meatballs with rat meat adulteration using Fourier Transform Infrared (FTIR) spectroscopy in combination with chemometrics. Int. J. Food Prop. 25(1), 1446–1457. Lestari, D., Syamsul, E.S., Wirnawati, Syafri, S., Syofyan, S., Rohman, A., Yuliana, N.D., Abu Bakar, N.K.B. and Hamidi, D. 2024. Rapid detection of rat meat adulteration in beef sausages using Ftir-Atr spectroscopy and chemometrics for halal authentication. Int. J. Appl. Pharm. 16(1), 82–88. Lestari, D., Syamsul, E.S., Wirnawati, Syofyan, S., Rohman, A. and Hamidi, D. 2023. Authentication of rattus norvegicus fat and other animal fats using gas chromatography-mass spectrometry (Gc-Ms) and principal component analysis (Pca). Int. J. Appl. Pharm. 15(1), 39–44. Miller, J.N., Miller, J.C. and Miller, R.D. 2018. Statistics and chemometrics for analytical chemistry, 7th ed. Pearson Education Limited, Harlow, United Kingdom. Nabila, Ade Puspa, S. and Trisnawaty, K. 2020. Minimum inhibitory and fungicidal concentration tests of snakehead fish (Channa striata) oil extracts against Candida albicans: an in vitro study. J. Dent. 1(1), 84–94. Nekha, A. 2024. Halal pharmaceutical development in Indonesia. Islam. Econ. Methodol. 2(2), 1–10. Nurjuliana, M., Che Man, Y.B., Mat Hashim, D. and Mohamed, A.K.S. 2011. Rapid identification of pork for halal authentication using the electronic nose and gas chromatography mass spectrometer with headspace analyzer. Meat Sci. 88(4), 638–644. Pasaribu, Y.P., Buyang, Y., Suryaningsih, N.L.S., Dirpan, A. and Djalal, M. 2020. Effect of steaming and pressurized boiling process to the nutrient profile of Papuan cork fish Channa striata as potential protein-rich food to prevent stunting. Med. Clin. Pract. 3, 100120. Pebriana, R.B., Rohman, A., Lukitaningsih, E. and Sudjadi. 2017. Development of FTIR spectroscopy in combination with chemometrics for analysis of rat meat in beef sausage employing three lipid extraction systems. Int. J. Food Prop. 20(2), 1995–2005. Putri, A.R., Rohman, A. and Riyanto, S. 2019a. Authentication of patin (Pangasius micronemus) fish oil adulterated with palm oil using ftir spectroscopy combined with chemometrics. Int. J. Appl. Pharm. 11(3), 195–199. Putri, A.R., Rohman, A. and Riyanto, S. 2019b. Comparative study of fatty acid profiles in patin (Pangasius micronemus) and gabus (Channa striata) fish oil and its authentication using ftir spectroscopy combined with chemometrics. Int. J. Appl. Pharm. 11(6), 55–60. Rahayu, S.W., Martono, S., Sudjadi, and Rohman, A. 2018. The potential use of infrared spectroscopy and multivariate analysis for differentiation of beef meatball from dog meat for Halal authentication analysis. J. Oleo Sci. Adv. Vet. Anim. Res. 5(3), 307–314. Riyanta, A.B., Riyanto, S., Lukitaningsih, E. and Rohman, A. 2020. The employment of fourier transform infrared spectroscopy (FTIR) and chemometrics for analysis of candlenut oil in binary mixture with grape seed oil. Food Res. 4(1), 184–190. Rohman, A. 2019. The employment of Fourier transform infrared spectroscopy coupled with chemometrics techniques for traceability and authentication of meat and meat products. J. Adv. Vet. Anim. Res. 6(1), 9–17. Rohman, A., Putri, A.R., Irnawati, Windarsih, A., Nisa, K. and Lestari, L.A. 2021. The employment of analytical techniques and chemometrics for authentication of fish oils: a review. Food Control 124(1), 107864. Rohman, A., Riyanto, S., Sasi, A.M. and Yusof, F.M. 2014. The use of FTIR spectroscopy in combination with chemometrics for the authentication of red fruit (Pandanus conoideus Lam) oil from sunflower and palm oils. Food Biosci. 7, 64–70. Rohman, A., Triyana, K., Sismindari, and Erwanto, Y. 2012. Differentiation of lard and other animal fats based on triacylglycerols composition and principal component analysis. Food Res. J. 19(2), 475–479. Sakamoto, A., Saotome, M., Iguchi, K. and Maekawa, Y. 2019. Marine-derived omega-3 polyunsaturated fatty acids and heart failure: current understanding for basic to clinical relevance. Int. J. Mol. Sci. 20(16), 4025. Salamah, N., Guntarti, A., Lestari, P.A. and Gandjar, I.G. 2022. Fat analysis of house rat (Rattus tanezumi) in meatball using gas chromatography-mass spectrometry (GC-MS) combined with principal component analysis. J. Pharm. 33(2), 208–214. Siscovick, D.S. Barringer, T.A., Fretts, A.M., Wu, J.H.Y., Lichtenstein, A.H., Costello, R.B., Kris-Etherton, P.M., Jacobson, T.A., Engler, M.B., Alger, H.M., Appel, L.J. and Mozaffarian, D. 2017. Omega-3 polyunsaturated fatty acid (fish oil) supplementation and the prevention of clinical cardiovascular disease: a science advisory from the american heart association. Circulation 135(15), 867–884. Song, C., Shieh, C.H., Wu, Y.S., Kalueff, A., Gaikwad, S. and Su, K.P. 2016. The role of omega-3 polyunsaturated fatty acids eicosapentaenoic and docosahexaenoic acids in the treatment of major depression and Alzheimer’s disease: acting separately or synergistically? Prog. Lipid Res. 62, 41–54. Subara, D. and Jaswir, I. 2018. Gold nanoparticles: synthesis and application for Halal authentication in meat and meat products. Int. J. Adv. Sci. Eng. Inf. Technol. 8(4–2), 1633–1641. Suh, J.H., Ho, C.T. and Wang, Y. 2017. Evaluation of carbonyl species in fish oil: an improved LC–MS/MS method. Food Control 78, 463–468. Syafri, S., Jaswir, I., Yusof, F., Rohman, A., Ahda, M. and Hamidi, D. 2022. The use of instrumental technique and chemometrics for essential oil authentication: a review. Results Chem. 4, 100622. Syamsul, E.S., Lestari, D., Hamidi, D., Umar, S., Wahyuni, F.S., Martien, R., Rohman, A. and Rusita, Y.D. 2024. Characterization and anti-aging activities of Haruan (Channa striata) fish oil. Food Res. 8(6), 1–6. Syifa, F., Hidayah, N., Lukitaningsih, E., Irnawati, and Rohman, A. 2022. Physicochemical properties, fatty acid composition and FTIR Spectra of Gabus (Channa striata) fish oil. Food Res. 6(2), 219–224. Windarsih, A., Indrianingsih, A.W., Apriyana, W. and Rohman, A. 2023. Rapid detection of pork oil adulteration in snakehead fish oil using FTIR - ATR spectroscopy and chemometrics for halal authentication. Chem. Pap. 77, 2859–2870. Zeng, Y.X., Araujo, P., Du, Z.Y., Nguyen, T.T., Frøyland, L. and Grung, B. 2010. Elucidation of triacylglycerols in cod liver oil by liquid chromatography-electrospray tandem ion-trap mass spectrometry. Talanta 82(4), 1261–1270. | ||
| How to Cite this Article |
| Pubmed Style Lestari D, Hamzah H, Pramesthi ADED, Syamsul ES, Safitri PD, Nurjunnah R, Rohman A. The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authentication. Open Vet. J.. 2025; 15(2): 646-659. doi:10.5455/OVJ.2025.v15.i2.13 Web Style Lestari D, Hamzah H, Pramesthi ADED, Syamsul ES, Safitri PD, Nurjunnah R, Rohman A. The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authentication. https://www.openveterinaryjournal.com/?mno=222401 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.13 AMA (American Medical Association) Style Lestari D, Hamzah H, Pramesthi ADED, Syamsul ES, Safitri PD, Nurjunnah R, Rohman A. The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authentication. Open Vet. J.. 2025; 15(2): 646-659. doi:10.5455/OVJ.2025.v15.i2.13 Vancouver/ICMJE Style Lestari D, Hamzah H, Pramesthi ADED, Syamsul ES, Safitri PD, Nurjunnah R, Rohman A. The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authentication. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 646-659. doi:10.5455/OVJ.2025.v15.i2.13 Harvard Style Lestari, D., Hamzah, . H., Pramesthi, . A. D. E. D., Syamsul, . E. S., Safitri, . P. D., Nurjunnah, . R. & Rohman, . A. (2025) The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authentication. Open Vet. J., 15 (2), 646-659. doi:10.5455/OVJ.2025.v15.i2.13 Turabian Style Lestari, Dwi, Hasyrul Hamzah, Asri Dwi Endah Dewi Pramesthi, Eka Siswanto Syamsul, Putri Dela Safitri, Rika Nurjunnah, and Abdul Rohman. 2025. The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authentication. Open Veterinary Journal, 15 (2), 646-659. doi:10.5455/OVJ.2025.v15.i2.13 Chicago Style Lestari, Dwi, Hasyrul Hamzah, Asri Dwi Endah Dewi Pramesthi, Eka Siswanto Syamsul, Putri Dela Safitri, Rika Nurjunnah, and Abdul Rohman. "The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authentication." Open Veterinary Journal 15 (2025), 646-659. doi:10.5455/OVJ.2025.v15.i2.13 MLA (The Modern Language Association) Style Lestari, Dwi, Hasyrul Hamzah, Asri Dwi Endah Dewi Pramesthi, Eka Siswanto Syamsul, Putri Dela Safitri, Rika Nurjunnah, and Abdul Rohman. "The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authentication." Open Veterinary Journal 15.2 (2025), 646-659. Print. doi:10.5455/OVJ.2025.v15.i2.13 APA (American Psychological Association) Style Lestari, D., Hamzah, . H., Pramesthi, . A. D. E. D., Syamsul, . E. S., Safitri, . P. D., Nurjunnah, . R. & Rohman, . A. (2025) The employment of FTIR-ATR spectroscopy and GC-MS combined with chemometrics for rapid detection of adulteration of pork oil in Gabus fish oil (Channa striata) for halal authentication. Open Veterinary Journal, 15 (2), 646-659. doi:10.5455/OVJ.2025.v15.i2.13 |