| Research Article | ||
Open Vet. J.. 2025; 15(2): 640-645 Open Veterinary Journal, (2025), Vol. 15(2): 640-645 Research Article Biometrical parameters of internal female genitalia in western Libyan Barbary sheepFarg Aboashia1*, Fatma Alatrag2, Aboulgasem Elmarimi3 and Abdalhalim Suaiee41Department of Theriogenology, Faculty of Veterinary Medicine and Agriculture, Zawia University, Zawia, Libya 2Department of Physiology, Faculty of Medicine and Surgery, Zawia University, Zawia, Libya 3Department of Theriogenology, Faculty of Veterinary Medicine, University of Tripoli, Tripoli, Libya 4Department of Statistics, Faculty of Science, Zawia University, Zawia, Libya *Correspondence to: Farg Alhadi Aboashia. Department of Theriogenology, Faculty of Veterinary Medicine and Agriculture, Zawia University, Zawia, Libya. f.buaeshah [at] zu.edu.ly Submitted: 04/10/2024 Accepted: 20/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
AbstractBackground: In Libya, the Barbary sheep plays an important role as a source of meat and wool. Improving animal production requires knowledge and understanding of the female reproductive system. Aim: This study aimed to evaluate the biometry of the female genitalia of the Barbary sheep of Libya. Methods: Ninety-seven apparently healthy, non-pregnant female Barbary sheep genitalia were collected immediately after slaughter from the slaughterhouse. Biometric parameters, including the length and width of the genital organs, were measured using a vernier caliper. The biometric data were studied in two groups: the estrous phase or active ovary group (n = 47) and the inactive ovary group (n = 50). The active ovary group was subdivided into follicular phase (n = 24) and luteal phase (n = 23) groups. Results: The results showed that different dimensions of the genital organs between the ewes in the follicular and luteal phases of the estrous cycle were significantly affected (p < 0.05). In the inactive ovary group, no significant (p > 0.05) difference was observed in the mean length and width of both the ovaries and uterine horns, whereas the right and left oviduct lengths were significantly different (p < 0.05). Conclusion: Knowledge of the biometric parameters of female genitalia would help in the identification of various organ abnormalities, diagnosis of pregnancy, and infertility treatment of Barbary sheep in Libya. Keywords: Barbary sheep, Female genitalia, Follicular phase, Luteal phase, Biometry IntroductionAgreeing with the Food and Agriculture Organization, which stated in 2022 that there are 1 billion sheep in the world. Fat-tailed Barbary sheep represent ˜25% of the world’s sheep population. In Libya, Barbary sheep are the dominant breed; they represent about 95% of Libyan sheep (7,546,321 head) (Abdulkarim, 2015). The Barbary sheep originated in Egypt and were found along the northern coast of Libya (Mason, 1967). Barbary sheep are hardy animals that are perfectly adapted to thrive in arid and harsh conditions, such as drought, heat stress, disease, and poor management (Akraim et al., 2008; Abdulkarim, 2015). It is important to understand the breed’s reproductive anatomy to maintain good reproductive performance. The anatomy of female animals’ genitalia provides information about their general health, pregnancy diagnosis, and infertility treatment (Bhat et al., 2011). However, very little is known about the morphology of the reproductive tract of Barbary sheep in Libya. Therefore, this study was aimed to evaluate the biometry of genital organs in female Barbary sheep using abattoir samples collected in Western Libya, and this knowledge will contribute some baseline data to the reproductive biology of this breed. Materials and MethodsA total of 97 non-pregnant, clinically healthy genital tracts of female Barbary ewes were collected from the slaughterhouse of Sabratha city from August to November 2023. The animals would arrive at the slaughterhouse in the early morning, and they would be placed in a barn and supplied with water. The veterinarian will conduct a physical examination of the sheep. The animals were slaughtered at an age of less than 1 year; age was estimated by dentation (Wilson, 1992). During routine slaughtering, genital organs without visible abnormalities were selected for the present study. The genital organs were collected in a thermocol box containing ice packs. As soon as the genital organs were brought to the laboratory, they were washed with normal saline and cleaned, and the adhering tissues were removed. The biometric parameters of female genitalia were measured. The length and width of different segments of the genital tract were measured using a vernier caliper (Fig. 1) on a centimeter (Prasad et al., 2020; Supriya et al., 2021). Organs were divided into two groups based on the presence of ovarian structures [corpus luteum (CL)] and follicles) into the estrous group (n = 47) and the inactive ovary group (n = 50) (Fig. 2). The estrous group was subdivided into the follicular phase (n = 24) and the luteal phase (n = 23). The different segments of the genital tracts were evaluated. The length of the vagina was defined as the distance from the external os of the cervix to the ventral commissure of the vulva. The cervix length was defined as the distance between the internal and external os. The uterine body length was measured from the internal os of the cervix to the uterine bifurcation. The uterine horn length was measured from the bifurcation of the uterus to the apex of the horn. The oviduct length was measured from the infundibulum to the uterine junction. The ovarian length was measured from the cranial to the caudal surface, and the width was measured from the lateral to the medial surface, following the methods described by Bhat et al. (2011), Barwary (2011), and Jaji et al. (2013). The corpora lutea in each ovary were counted.
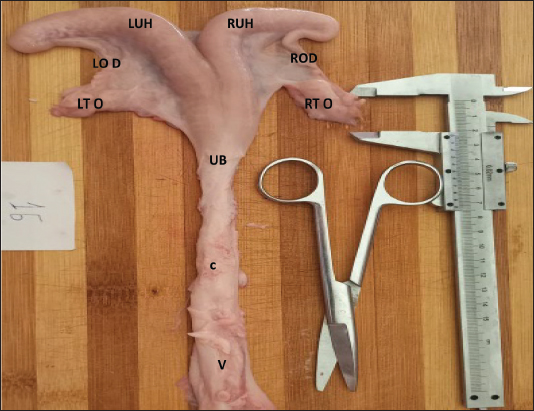
Fig. 1. Image of the internal genital tract of the western Libyan Barbary ewe (younger than one year). V: Vagina; CX: Cervix; UB: Uterine body; LUH: Left uterine horn; RUH: Right uterine horn; OD: Oviduct; LTO: Left ovary; RTO: Right ovary.
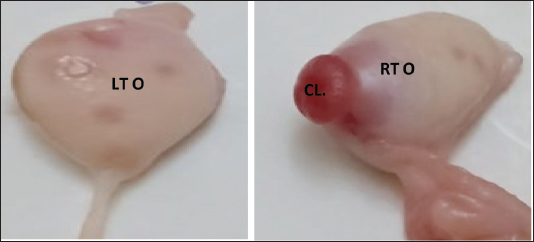
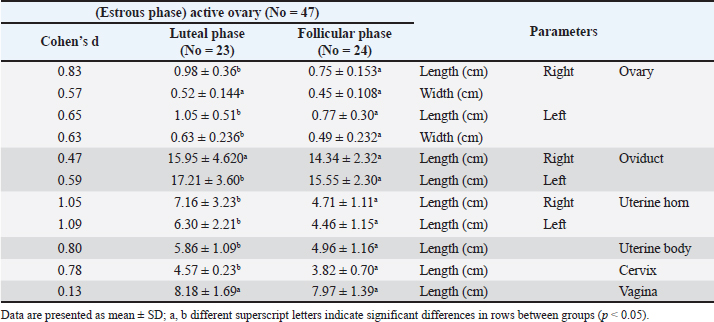
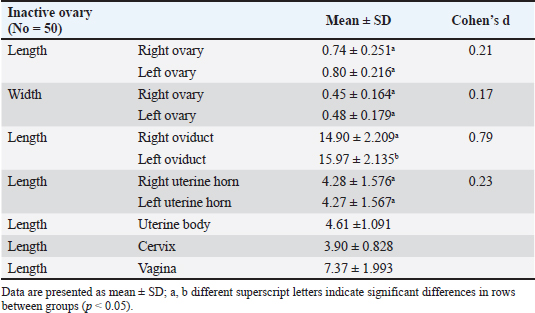
Fig. 2. Right ovary (RTO) of the Barbary ewe showing the CL. Statistical analysisThe data were analyzed using IBM SPSS Statistics (Version 27, IBM Corp., Armonk, NY). Data normality was assessed using the Shapiro–Wilk test for all variables. For variables following a normal distribution (p > 0.05), paired t-tests were used to compare right and left reproductive structures, and independent sample T tests were used to compare right luteal and follicular phases and also to compare left luteal and follicular phases. For non-normal variables (p < 0.05), the Wilcoxon signed-rank test was employed to ensure that assumptions were not violated. Sample sizes ranged from 30 to 50, and effect sizes (Cohen’s d or rank- biserial correlation) were calculated to complement the p-values. All tests were two-tailed with significance set at α = 0.05. This approach ensures the validity and reliability of statistical inferences while minimizing potential biases. ResultsThe genital organs of female Barbary ewes were examined, and biometric measurements were performed. A comparison of biometrical parameters between the two estrous phases: Length of the right and left ovaries showed a significant difference (p < 0.05) at the luteal phase compared to the follicular phase “(0.98 ± 0.36 vs. 0.75 ± 0.15; 1.05 ± 0.51 vs. 0.77 ± 0.30),” respectively. Moreover, the width of the left ovaries presented a significant (p < 0.05) difference between the two phases, whereas the width of the right ovaries showed no differences “(0.63 ± 0.23 vs. 0.49 ± 0.23; 0.52 ± 0.14 vs. 0.45 ± 0.10).” The length of the left oviduct showed significant (p < 0.05) differences at the luteal phase compared with the follicular phase “(17.21 ± 3.60 vs. 15.55 ± 2.30), “whereas the average length of the right oviducts revealed no difference “(15.95 ± 4.620 vs. 14.34 ± 2.32)„ between both estrous cycle phases. The lengths of the right horn were 7.16 ± 3.23 and 4.71± 1.11 cm, and those of the left uterine horn were 6.30 ±2.21 and 4.46 ± 1.15 cm at luteal and follicular phases. The lengths of the two horns differed significantly (p < 0.05) during the luteal and follicular phases. The uterine body and cervix length showed significant (p < 0.05) differences in the luteal phase compared with the follicular phase “(5.86 ± 1.09 vs. 4.96 ± 1.16; 4.58 ± 1.122 vs. 3.85 ± 0.702),” respectively. The mean vaginal length did not differ significantly (p > 0.05) difference “(8.18 ± 1.69 vs. 7.97 ± 1.39).„ The numbers of CL were 14 in the right ovaries and 16 in the left ovaries (Table 1). The study analyzed 50 genital organ measurements taken from an inactive ovary group and compared right-and left-side differences (Table 2). The results showed insignificant (p > 0.05) differences in organ length. However, there was a marked (p < 0.05) difference between the right and left oviducts “(14.90 ± 2.209; 15.97 ± 2.135),” respectively. Table 1 Comparison of biometric parameters between the follicular and luteal phases.
Table 2 Biometrical parameters of female genitalia in Barbary ewes of inactive ovary group.
DiscussionIn the current study, the length and width of the right and left ovaries were significantly different in the luteal phase compared with the follicular phase. A higher biometric value was recorded for the length and width of the left ovary than for the right ovary. Despite the differences in ages between animals in this study and those of Rajput and Sharma (1997) and Bhat et al. (2011), our results were in accordance with theirs. On the other hand, EL-Saigh et al. (2011) found that the right ovary was more active than the left ovary. Meanwhile, Alsafy and El-Shaha (2011), Hyacinth et al. (2016), Prasad et al. (2020), Supriya et al. (2021), and Reddy et al. (2023) observed insignificant differences between the length and width of the right and left ovaries (Table 1). The significant increase in the size of the ovary during the luteal phase may be due to the presence of corpora lutea on the surface (Noakes et al., 2001; Alsafy and El- Shaha, 2011). In the ewe, increased CL size and weight are due to hypertrophy and hyperplasia of luteal cells as CL develops. In addition to changes in steroidogenic and non-steroidogenic cell numbers during the estrous cycle (Noakes et al., 2001; Sangha et al., 2002; Senger, 2003), there was a significant (p < 0.05) difference between the mean lengths of the left oviduct observed in the luteal and follicular phases. This result is in agreement with the findings of Rajput and Sharma (1997), Saleem et al. (2017), and Rennak et al. (2024). Although other researchers (Suri et al., 2013; Prasad et al., 2020) did not find significant variation in the parameters of the right and left oviducts, the average length of the uterine body during the luteal phase was significantly different from that during the follicular phase. This finding is consistent with that of EL-Saigh et al. (2006). However, no significant difference was reported by Prasad et al. (2020). The mean lengths of the right and left uterine horns were significantly different during the luteal phase compared with the follicular phase. This finding is consistent with those of Suri et al. (2013) and Saleem et al. (2017). Other studies have indicated that the left uterine horn was longer than the right horn, although this difference was not statistically significant (EL-Saigh et al., 2006). Prasad et al. (2020) reported no significant differences in the right and left uterine horns between the luteal and follicular phases. The cervix was longer at the luteal phase in this study. The results are consistent with Kershaw et al. (2005) and Saleem et al. (2017). Moreover, some previous studies (EL-Shaha and Alsafy, 2009; Abbasabadi et al., 2019; Prasad et al., 2020; Abril-Parreno et al., 2021) reported that there was no difference in cervical lengths during different phases of the estrous cycle. This found no significant difference in the average vaginal length between the luteal and follicular phases. This finding is consistent with the findings of Hyacinth et al. (2016). The CL and the number of follicles have a local effect on the ovary diameter (Bartlewski et al., 2001). We noticed no significant difference in the average number of CL between the right and left ovaries. This result was similar to that of Hoque et al. (2016), who reported that the average number of CL was almost the same in both ovaries. In the inactive group, there were no significant differences in the mean length and width of the ovaries. This result was in accordance with the previous studies by Hyacinth et al. (2016), Prasad (2020), Supriya et al. (2021), and Reddy et al. (2023). Islam et al. (2018) observed that the average length and width of the right ovary surpassed those of the left ovary. In contrast, Rajput and Sharma (1997) observed that the mean length and width of the left ovary exceeded those of the right ovary. The study found a significant difference in the mean length of the left oviduct compared with the right oviduct, consistent with previous research (Rajput and Sharma, 1997). Bhat et al. (2011) reported that the right oviduct was significantly longer than the left oviduct, whereas Reddy et al. (2023) found no difference between the lengths of the two oviducts. The average lengths of the uterine horns were not different. This result agreed with Hyacinth et al. (2016). Some studies reported that the left uterine horn was significantly longer than the right horn (Reddy et al., 2023), whereas Bhat et al. (2011) and Shehan et al. (2019) found that the right horn was longer than the left horn. In this study, the mean lengths of the uterine body, cervix, and vagina were similar to those reported by Hyacinth et al. (2016) and slightly higher than those reported by Bhat et al. (2011). However, the discrepancy between the findings of the present study and those of earlier studies might be due to differences in breed, age, reproductive status of the animals, genetic and endocrine profiles of the animals, nutrition, and climate. ConclusionThis study established basic measurements of the female genital tract of Barbary sheep. These findings will facilitate the identification of various organ abnormalities, pregnancy detection, and infertility treatment in this animal breed. AcknowledgmentWe would like to thank the staff members of the slaughterhouse in Sabratha City for their help. Conflict of interestThere are no conflicts of interest. FundingNo funding to declare. Authors’ contributionsAll authors contributed equally and read and approved the final manuscript. Data availabilityThe data supporting the findings of this study are available and can be obtained from the corresponding author. ReferencesAbbasabadi, M., Kochakzadeh, B.H. and Aski, K. 2019. Evaluating gross anatomy of cervix in zel sheep. Ana. Sci. 16(1), 39–44. Abdulkarim, A. 2015. Small ruminant contribution In meat production in Libya. Egyptian J. Sheep Goat Sci. 10(1), 13–18. Abril-Parreno, L., Krogenæs, A.K., Byrne, C.J., Donovan, A., Stuen, S., Caldas, E., Diskin, M., Druart, X. and Fair, S. 2021. Differences in ewe breeds in cervical anatomy and cervicovaginal mucus properties: an international study. Theriogenology. 15(160), 18–25. Akraim, F., Milad, I.S., Abdulkarim, A. and Ganem, M. 2008. Wool characteristics of Libyan Barbary sheep in north-eastern Libya: I. fiber diameter and staple length. Livestock Res. Rural Develop. 20(8), 1–6. Alsafy, M. and El-Shahat, K.H. 2011. Morphological evaluation of ovary in relation to recovery, quality of Oocytes and Steroid production in sheep. J. Appl. Biol. Sci. 5(3), 27–31. Bartlewski, P.M., Beard, A.P. and Rawlings, N.C. 2001. Ultrasonographic study of the effects of the corpus luteum on antral follicular development in unilaterally ovulating western white-faced ewes. Anim. Repro. Sci. 65(3–4), 231–244. Barwary, M.S.Q. Seasonal physiological activity of genital system in local sheep and goat in Kurdistan Region. The 3rd International conference on sustainable anim. Agr. for Develop. countries (SAADC), 2011, Thai, 26–29. Bhat, F.A., Bhattacharyya, H.K. and Khan, M.Z. 2011. Biometry of female genitalia and in vitro tubal patency tests in local sheep of kashmir valley. India J. Small Rum. 17(2), 170–173. El-Saigh, M.N., Hatif, S.A. and Hussin, A.M. 2006. Morpho-anatomial changes of awassi ewes genitalia at different phase of estrus 2-The uterine. Bas. J. Vet. Res. 5(1), 64–69. El-Saigh, M.N., Hussin, A.M. and Hatif, S.A. 2011. Morpho-anatomial changes of awassi ewes genitalia at different phase of estrus 1-the ovary. Bas. J. Vet. Res. 10(1), 82–87. EL-Shaha, K.H. and Alsafy, M.A.M. 2009. The anatomical structures of sheep cervix and its influence on the transcervical passage of an inseminating catheter into the uterine lumen. Vet. Med. J. 57 (2), 211–222. Food and Agriculture Organization, 2022. Agriculture Data. Food and Agriculture Organization Statistics. http://www.fao.org/statistics/en. Hoque, S.A., Islam, M.D., Abu, S. and Selim, M.D. 2016. Interspecies differences on ovarian parameters between black bngal goat and indigenous Bengal sheep in view of in vitro maturation. Adv. Life. Sci. 6(3), 54–60. Hyacinth, A., Terzungwe, A., Daniel, O.L.D. and Oluwabamise, J. 2016. Biometrical study of the genitalia of kano brown goats and yankasa sheep. Int. J. Agro Vet. Sci. Res. 10(1), 3–8. Islam, M.d.R., Khaton, R., Islam, M.d.H., Ullah, M.d.N., Nishi, S.A., Paul, B.R. and Sarder, M.d.J.U. 2018. Biometry of ovary in different ruminant animals. Bangladesh Livest. J. 1, 44–48. Jaji, Z., Buduwara, R., Akanmu, A., Elelu, N., Kigir, E. and Mahre, M.B. 2013. Pregnancy- related biometrical changes in the ovaries and uterus of the balami sheep. Glob. J. Med. Res. Vet. Sci. Vet. Med. 13(12), 1–6. Kershaw, C.M., Khalid, M., Mcgowan, M.R., Ingram, K., Wax, S.L.G. and Scaramuzzi, R.J. 2005. The anatomy of the sheep cervix and its influence on the transcervical passage of an inseminating pipette into the uterine lumen. Theriogenology 64(5), 1225–1235. Mason, I.L. 1967. The sheep breeds of the Mediterranean. Commonwealth Agricultural Bureaux International, Wallingford, Oxon, UK. pp. 215. Noakes, D.E., Parkinson, T.J. and England, G.C. 2001. Arthur’s veterinary reproduction and Obstetrics, 8th ed. Philadelphia, PA: W.B. Saunders, pp: 22–26. Prasad, B.S., Srinivas, M.G., Naidu, V. and Rao, V.V. 2020. Studies on biometry of female genitalia in Nellore sheep of Andhra Pradesh. J. Entomol. Zool. Stud. 8(5), 2192–2194. Rajput, R. and Sharma, D.N. 1997. Biometry of female genitalia of Gaddi sheep. Ind. J. Anim. Sci. 67(12), 1073. Reddy, B., Nageswara, S.M., Shanawaz, K.A. and Krishna, N.V. 2023. Biometric study on female reproductive tract of Nellore sheep. Int. J. Vet. Sci. Anim. Husb. 8(4), 82–85. Rennak, K., Chikhaoui1, M., Mahouz, F. 2024. Biometrical changes in reproductive tract of Arbia goats according to Age, body condition, and pregnancy. Iranian J. Vet. Med. 18(3), 345–358. Saleem, R., Shalini, S. and Sarma, K. 2017. Gross and biometrical studies on internal female genitalia of adult Bakerwali Goat in different phases of estrus cycle. Indian J. Vet. Anat. 29(2), 23–25. Sangha, G.K., Sharma, R.K. and Guraya, S.S. 2002. Biology of corpus luteum in small ruminants. J. Small Rumin. Res. 43(1), 53–64. Senger, P.L. 2003. Pathway to pregnancy and parturition. In Cadmus professional communications, 2nd ed. Pullman, Washington. pp: 190–198. Shehan, N.A., Kareem, D.A., Hussein, H.A., ALkhalad, W.J. and Jassem, E.S. 2019. Morphological and histological study of uterus in domestic Iraqi sheep. Al-Qadisiyah J. Vet. Med. Sci. (18), 1. Supriya, B., Jamuna, K.V., Ramayya, P.J., Prasad, R.V., Sathyanarayana, M.L. and Kumar, S. 2021. Morphologic and morphometric exploration of the ovarian follicles in the Nellore sheep (Ovis aries). Int. J. Chem. Stud. 9(1), 699–702. Suri, L.S., Sarma, K. and Tagge, R.K. 2013. Biometry of the internal female genital organs of different age groups of Bakerwali goat (Kaghani Goat). Indian J. Vet. Ana. 25(1), 41–42. Wilson, A. 1992. Practical meat inspection. In Sex Characteristic and estimation of age, 5th ed. Blackwell sci, pp: 79–105. | ||
| How to Cite this Article |
| Pubmed Style Aboashia FA, Alatrag FA, Elmarimi AA, Suaiee AM. Biometrical parameters of internal female genitalia in western Libyan Barbary sheep. Open Vet. J.. 2025; 15(2): 640-645. doi:10.5455/OVJ.2025.v15.i2.12 Web Style Aboashia FA, Alatrag FA, Elmarimi AA, Suaiee AM. Biometrical parameters of internal female genitalia in western Libyan Barbary sheep. https://www.openveterinaryjournal.com/?mno=223251 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.12 AMA (American Medical Association) Style Aboashia FA, Alatrag FA, Elmarimi AA, Suaiee AM. Biometrical parameters of internal female genitalia in western Libyan Barbary sheep. Open Vet. J.. 2025; 15(2): 640-645. doi:10.5455/OVJ.2025.v15.i2.12 Vancouver/ICMJE Style Aboashia FA, Alatrag FA, Elmarimi AA, Suaiee AM. Biometrical parameters of internal female genitalia in western Libyan Barbary sheep. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 640-645. doi:10.5455/OVJ.2025.v15.i2.12 Harvard Style Aboashia, F. A., Alatrag, . F. A., Elmarimi, . A. A. & Suaiee, . A. M. (2025) Biometrical parameters of internal female genitalia in western Libyan Barbary sheep. Open Vet. J., 15 (2), 640-645. doi:10.5455/OVJ.2025.v15.i2.12 Turabian Style Aboashia, Farg Alhadi, Fatma Ayad Alatrag, Aboulgasem Ahmed Elmarimi, and Abdalhalim Meloud Suaiee. 2025. Biometrical parameters of internal female genitalia in western Libyan Barbary sheep. Open Veterinary Journal, 15 (2), 640-645. doi:10.5455/OVJ.2025.v15.i2.12 Chicago Style Aboashia, Farg Alhadi, Fatma Ayad Alatrag, Aboulgasem Ahmed Elmarimi, and Abdalhalim Meloud Suaiee. "Biometrical parameters of internal female genitalia in western Libyan Barbary sheep." Open Veterinary Journal 15 (2025), 640-645. doi:10.5455/OVJ.2025.v15.i2.12 MLA (The Modern Language Association) Style Aboashia, Farg Alhadi, Fatma Ayad Alatrag, Aboulgasem Ahmed Elmarimi, and Abdalhalim Meloud Suaiee. "Biometrical parameters of internal female genitalia in western Libyan Barbary sheep." Open Veterinary Journal 15.2 (2025), 640-645. Print. doi:10.5455/OVJ.2025.v15.i2.12 APA (American Psychological Association) Style Aboashia, F. A., Alatrag, . F. A., Elmarimi, . A. A. & Suaiee, . A. M. (2025) Biometrical parameters of internal female genitalia in western Libyan Barbary sheep. Open Veterinary Journal, 15 (2), 640-645. doi:10.5455/OVJ.2025.v15.i2.12 |