| Research Article | ||
Open Vet. J.. 2025; 15(2): 660-667 Open Veterinary Journal, (2025), Vol. 15(2): 660-667 Research article Molecular characterization of Leishmania infection in canineSamar Abbas Fadhil and Mansour Jadaan AliDepartment of Microbiology, College of Veterinary Medicine, University of Al-Qadisyiah, AL-Diwanyia, Iraq * Correspondence to: Mansour Jadaan Ali. Department of Microbiology, College of Veterinary Medicine, University of Al-Qadisyiah, AL-Diwanyia, Iraq. Mansoor.ali [at] qu.edu.iq Submitted: 07/10/2024 Accepted: 05/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
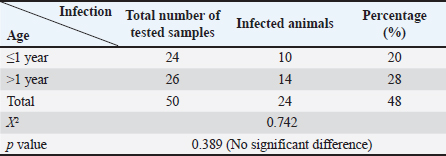
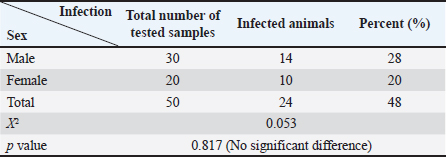
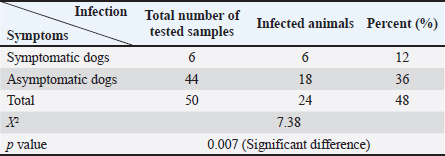
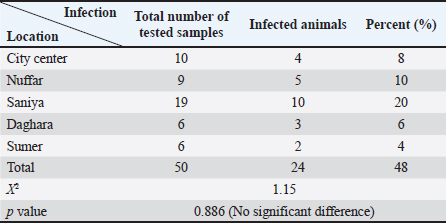
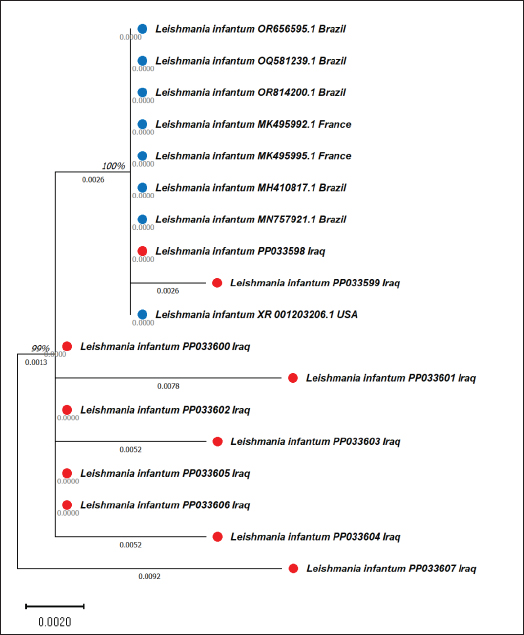
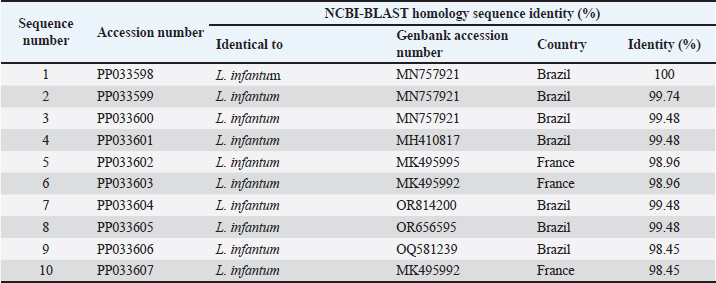
AbstractBackground: Canine leishmaniasis is an infectious protozoan disease transmitted by sand fly bites. The parasite infects the macrophages of vertebrates during the amastigote stage. Aim: This study aimed to detect the causative species for infections between dogs with Leishmania in Iraq and to determine the relationship between isolates that infect Iraqi animals and other animals worldwide. Methods: Fifty dogs of various breeds, ranging from 1 month to 7 years old of both sexes. Then, blood samples were collected from the cephalic vein into an EDTA tube; DNA was extracted using a commercial kit. Afterward, conventional PCR was performed using universal primers to detect 18S in Leishmania. Results: Most dog infections were caused by Leishmania infantum. The age of the infected dogs ranged from a few months to 7 years. Our results indicate that most infections with Leishmiania spp. occurred between older males and younger females. Most dogs were asymptomatic, and a small percentage exhibited clinical signs, such as skin lesions, rough coats, hair loss, and onychogryphosis. To confirm the results and gain insight into the origin of the positive L. infantum isolates, 10 samples were sequenced. Interestingly, L. infantum isolates were typical of those recorded in Brazil, France, and the USA, except for one isolate, which was slightly different from the detected isolates. Therefore, our results showed that most infections were between older males rather than younger females, and the DNA sequencing results indicated that only 1 isolate out of 10 had a different demographic origin. Conclusion: Dogs in this study were infected mainly with L. infantum without any clinical signs; most isolates resemble those recorded in Brazil, France, and the USA. Keywords: Leishmania, Canine, PCR, Phylogenetic analysis, Al-Qadisiyiah IntroductionCanine leishmaniasis is a zoonotic protozoan disease caused by Leishmania infantum, which is transmitted by phlebotomine sand flies. It is considered a relevant veterinary and public health problem in various countries, where dogs are considered the main reservoir (Albuquerque et al., 2017). Dogs can develop clinical signs or be asymptomatic; nevertheless, they can spread the protozoan within the dog population and remain a source of infection for sand flies, which transmit the parasite to other hosts (Solano-Gallego et al., 2017). The clinical form of the disease varies from cutaneous leishmaniasis (CL), mucocutaneous leishmaniasis, and visceral leishmaniasis (VL); CL is the most prevalent form of the disease (Eshetu et al., 2016). VL, also known as kala-azra, is caused by both L. infantum and L donovani. Moreover, several signs and symptoms are associated with its pathogenesis, such as fever, weight loss, paleness, and enlargement of the spleen and liver (hepatosplenomegaly) (Najafi et al., 2021; Costa et al., 2023). Leishmaniasis is endemic in Iraq, and both cutaneous and visceral forms have been reported (AlSamarai and Al Obaidi, 2009; Gani et al., 2010). Diagnosis of Leishmania is based on the detection of amastigote in biopsy/sample smears of lymphoid organs such as bone marrow, lymph nodes, and spleen, as well as liver and skin (Tafuri et al., 2001; Sundar and Rai, 2002). Molecular diagnosis is based on the amplification of several targets, including ITS- 1, hsp70, 18S, and cathepsin L-like, by PCR, as referred by da Silva et al., (2019); Giraldo-Martínz et al., (2022); and Osorio-Peralta et al., (2024), which allow phylogeny and taxonomy studies. Thus, the aim of this study was to detect species that mainly infect dogs and to determine the phylogenetic relationship between the detected isolates and other globally recorded species. Materials and MethodsStudy area and sample collectionThe investigation was performed from February to October 2023 in Al-Diwaniyah, Iraq. A total of 50 blood samples were collected from randomly stray and domestic dogs via cephalic venipuncture under sterile conditions. Aggressive dogs were sedated with ketamine 10%/xylazine 2% (Alfasan, Holland) in 1:1/2 ml i.m. Blood was collected in tubes with EDTA as anticoagulant and stored in a cooling box with ice until delivery to the biological laboratory for analysis and stored at –20○C until DNA extraction. DNA extractionGenomic DNA was extracted from whole blood using a tissue DNA extraction kit (AddBio, Korea), and the DNA concentration was measured using a QuantusTM Fluorometer (Promega, USA) following the manufacturer’s instructions. The extracted DNA samples were stored at –20°C until use. Molecular diagnosis was performed by conventional PCR using universal primers targeting the partial regions of the small ribosomal RNA gene 18S (F-5’ GCCTCTAGGCTACCGTTTCG ’3; R- 5’ GTAAGGTGGTAAAAGCGGGC ‘3). Primers were designed using the National Center for Biotechnology Information (NCBI) service with the relevant accession numbers of (MN757921) related to Leishmania spp. The PCR was run in 20 µl total reaction volume in which 10 µl master mix (AddBio, Korea), 2 µl of each primer (forward and reverse), 4 µl of nuclease-free water, and 2 µl of template DNA. The thermal conditions were achieved in a thermal cycler system (BioRad, USA) with an initial denaturation of 95°C for 3 minutes, followed by 39 cycles of denaturation at 95°C for 30 seconds, 55°C for annealing, and 72°C for 30 seconds as an extension step and final extension at 72°C for 1 minutes. PCR products were analyzed by agarose gel electrophoresis, and amplicons were selected from positive PCR samples for sequencing using the Sanger method at Macrogen Inc. (South Korea). Sequence data were deputed and submitted to GenBank (NCBI) for assignment of accession numbers. A phylogenetic tree was built using Molecular Evolutionary Genetics Analysis version 10 (Mega x) and multiple sequence alignment analysis based on Clustal W alignment analysis using sequences reported in GenBank. This analysis was performed using the software provided by Stecher et al. (2020): Molecular Biology Evolutionary Genetics Analysis for macOS from Molecular Biology and Evolution (http://doi.org/10.1093/molbev/msz312). Statistical analysisAll the statistical analysis depending on chi square was done by using Statistical Package for the Social Science (SPSS) program (version-10) (SPSS, 2006). In all cases, p < 0.05 was considered significant. Ethical approvalThe study protocol was approved for animal care and use by the College of Veterinary Medicine, University of Al-Qadisiyah, Al-Diwaniyah province, Iraq. The study was approved on November 22, 2022 (No. 1890). ResultsThe dog population consisted of 50 random dogs domesticated by rural people; almost all the animals were healthy except 6 dogs exhibited scattered clinical signs, such as skin lesions, particularly in the hips, legs, around the eyes, and tail, with a rough coat, dehydration, segregation, and skin wounds in different parts of the body due to fighting with other dogs in the herd. Occasionally, a high temperature, redness of one or both eyes, anemia, thickening of paws hyperkeratinization, loss of hair in the infected area, and onychogryphosis. Age was determined after asking the owner or by examining the teeth of the calming animals. PCR amplification showed that 24 animals were positive for Leishmania spp. by amplification of the 18S gene. Regarding age, a higher prevalence rate was detected in dogs older than 1 year old, mostly males compared to females. All animals with symptoms were infected and almost all asymptomatic dogs. The highest distribution rate of infected dogs was in the Saniya district at 20% (Figures 1 and 2); Table (1–4). Results showed that 10 Iraqi isolate records (PP033598; PP033599; PP033600; PP033601; PP033602; PP033603; PP033604; PP033605; PP033606; and PP033607) of L. infantum submitted to GenBank were based on phylogenetic analysis of similarly nucleotide sequence recorded isolates; these isolates were as follows: Brazilian (MN757921.1; MH410817.1; OR814200.1; OR656595.1; and OQ581239.1) (Marcili et al., 2020; Rogerio et al., 2023); French (MK495995.1 and MK495992.1) (Medkour et al., 2019); and American (XR_001203206.1) (Peacock et al., 2007), as seen in Figure 3. Depending on the phylogenetic tree analysis of the S18 sequence in Leishmania, we can notice the typical identity range (100%) of isolates PP033598 and PP033599 (remarked with a red circle) to the global isolates such as MN757921.1_Brazil; MH410817.1_ Brazil; OR814200.1_Brazil; OR656595.1_Brazil; OQ581239.1_Brazil; MK495995.1_France; and XR_001203206.1_USA (remarked with a blue circle), while the isolates PP0033600, PP0033602, PP0033605, and PP0033606 showed a wide identity range (99%) for the previously recorded isolates in Brazil, France, and USA. The isolates PP0033601, PP0033603, and PP0033604 have a slight identity (98%–99%), and PP0033607 has little difference from all other isolates, as shown in Figure 3 of the phylogenetic tree. When compared to the new isolates, such as PP033598 to other cosmopolitan isolates such as MN757921.1_ Brazil showed 100% identity, while PP033599, PP033600, PP033601, PP033604, and PP033605 showed 99.74%, 99.48%, 99.48%, 99.48%, and 99.48% identity, respectively. The isolates PP033602, PP033603, PP033606, and PP033607 showed 98.96%, 98.96%, 98.45%, and 98.45% identity, respectively, depending on NCBI- BLAST Homology sequence identity (Table 5). This results detected according to the gene that was used in the study and comparison in addition to parasite species, while another study may detect another parasite species and use different genes for sequencing.
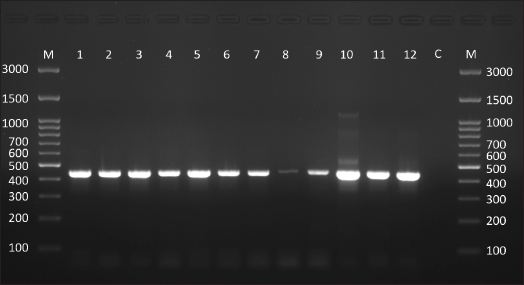
Fig. 1. Agarose gel electrophoresis (1.5% agarose) shows the positive product of Leishmania spp. targeting a partial region within the small subunit ribosomal RNA gene (size = 445 bp). M: molecular weight marker (Genedirex, South Korea). C: negative control, deionized-distilled H2O was added as a template.
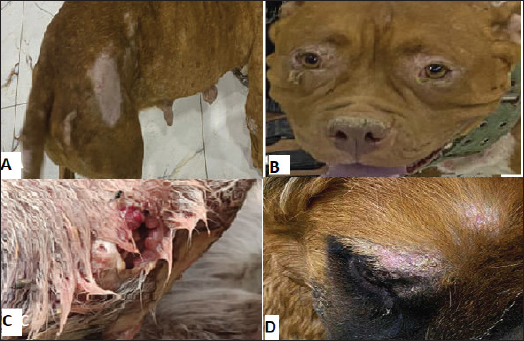
Fig. 2. Representative clinical signs of the study population with leishmaniasis. (A) Presence of skin lesions, loss of hair, and hyperkeratinization in flank, hip, tail, and ankle. (B) Periorbital skin lesions. (C) Wounds and inflammation in the skin polarized to insects. (D) Supraorbital skin lesions with scales. DiscussionCanine leishmaniasis is a zoonotic chronic disease; the vector is mostly infected sand flies and can be potentially fatal to humans and dogs and their diagnosis and laboratory aspects are very difficult, especially due to the lack of more effective drugs and vaccines (Ribeiro et al., 2018). Difficult diagnosis may be due to asymptomatic dogs that are predominantly infected without symptoms. Leishmania infantum is widely distributed throughout the canine population, and the dog is a reservoir host that acts as a focus for transmission of the infection; the percentage of infection was 48% that was agreed to Al Obaidii and Almashhadany (2023), who recorded the highest risk positive results in buffy coat samples compared to skin biopsy using the ITS1 gene. Nested PCR showed that the overall prevalence of leishmaniasis in dogs was 88.33% based on the use of kDNA gene primers that belonged to L. tropica in the middle Euphrates and Al Diwanyia city (AlSeady and Al-Dabbagh, 2024). Table 1. Number of infected dogs according to age.
Table 2.Number of infected dogs according to sex.
Table 3.Number of infected dogs according to the presence of clinical signs.
Table 4.Number of infected dogs according to location collected from.
Fig. 3. Phylogenetic tree of L. infantum based on 18S gene sequences. The tree was built using the maximum likelihood method and was drawn to scale, with branch lengths measured as the number of substitutions per site (below the branches). There were 386 positions in the final dataset. Evolutionary analyses were conducted using MEGA10 software. The scale bar indicates the distance between these sequences. The prevalence of infection in younger animals (<1 year) was 20%, and the percentage recorded in older animals (≤1 year old) was 28%; there was no significant difference at p-value and this result was similar to that recorded by Lima et al. (2019) and Al Obaidii and Almashhadany (2023). The reason is the chance that older dogs have more environmental contact than younger dogs during the suckling period, as the contact with their moms and the risk of infection increase with age (Lima et al., 2019). Another study by Rombolá et al., (2021) and Salant et al., (2021) documented that older dogs mainly had higher positivity rates than younger strays, and rural dogs had higher infectivity rates than owned and urban dogs. The possibility of vertical transmission from mothers to puppies in the cases of a small-age dog infection is one of the transmission methods (Rosypal, 2005; Al Obaidii, 2019; Lima et al., 2019; Salant et al., 2021). The infectivity rate in males was 28% and in females was 20%; there was no significant difference at p-value, and these results were similar to those of Al Obaidii and Almashhadany (2023). The reason was that males were affected by fighting between them on dominion the herd which led to exposure of several dogs to skin wounds; wounds may be attractive for insects that transmit some diseases, including Leishmania. Table 5.The NCBI-BLAST homology sequence identity (%) of local L. infantum numbers (PP033598, PP033599, PP033600, PP033601, PP033602, PP033603, PP033604, PP033605, PP033606, and PP033607), which were compared with other global sequences.
Furthermore, the most neglected clinical signs were skin lesions, hyperkeratinization onychogryphosis, and skin wounds in different parts of the body, especially the face, ears, nose, and skin around the eyes; skin lesions were indicated for infective dogs, there was a significant difference, and the results were similar to those noticed by Rombolá et al. (2021). Garbi et al. (2015) noticed that there were skin ulcers on the face and quarters and diarrhea, vomiting, and paw pad fissure, and the other animals did not show any findings; there was thickening in the paws (Alwan and Jassim, 2021) that differ from Garbi et al. (2015) who said that there were 11.54% of animals are asymptomatic and 88.46% were symptomatic for canine leishmaniasis. The infection rate was higher in Sanyia depending on the collected samples from this district compared to other districts; there was a significant difference and disagreement with Al-Maeahi and Marhoon (2018) and Alwan and Jassim (2021), who said Sanyia had the lowest percentage than Al Hamza Al Sharqi district when they collected blood from infected children with VL; the reason may be due to favorable environmental conditions for sand flies breeding in these regions, the increased exposure of dogs to sand fly bites and do not involved of Al Hamza Al Sharqi in the current study it was being higher prevalence rate in canine leishmaniasis. The sequence results and phylogenetic tree analysis showed that all sent isolates were positive for L. infantum and resemble global isolates such as Brazilian (Marcili et al., 2020; Rogerio et al., 2023), French (Medkour et al., 2019), and American isolates (Peacock et al., 2007). These results may be caused by the recent breeding of animals and the mating of local canine breeds with foreign breeds; thus, puppies can be infected with leishmaniasis during pregnancy (Rosypal, 2005). The weak quarantine of the Iraqi border, especially with Iran and Turkey, aids to introduce different foreign breeds that may carry the infection asymptomatically attributed to the similarity between recently recorded strains and the latest Brazilian, French, and American strains. Nucleotide sequence analysis indicated that all sequences have at least 99.07% of identity with other Leishmania (Leishmania) infantum strains and isolates were close genetically with a very low sequence variability among isolates and L. infantum using internal transcribed spacer genes; identity ranged from 99.63% up to 100% of identity (Al Obaidii and Almashhadany, 2023), whereas Al-Ardi (2022) detected Leishmania spp. using nanoparticles with an overall relative sensitivity of 90% using nanoparticles of the Ist1 gene. Few studies in Iraq, especially those on canines, and wide spectrum of Leishmania spp. and the current study specialized on L. infantum only attributed to recorded this results from people habit to open mind on the world and attributed many families to borrowing and breeding the puppies while imported or domestic animals trading with animals and matting to domestic breeds so the dogs can transmit the infection that aids to the presence of Leishmania within high percent; in addition to the dogs is a reservoir hosts and asymptomatic reservoir host that act as focus for transmission of the disease. This is the first study in Iraq to report a canine L. infantum in Al Qadisiya province using the sequence analysis of the 18S gene, while Al-Ardi (2022); Ferreira et al. (2022); Al Obaidii and Almashhadany (2023) reported the presence of Leishmania DNA in blood samples when they detected the ITS1 gene of Leishmania. A wide range of the seriously of L. infantum lies in infectious diseases between humans and animals and black fever disease that infects children, which is mainly known as VL. Many people breed dogs to guard and protect their livestock, houses, and wide fields, or for ornament and entertainment, which causes a serious problem in many areas. Leishmaniasis is a major health problem for pets and domestic canines in tropical and subtropical areas that have been linked to poverty and limited access to health care; control is difficult because the vectors are present (Marcili et al., 2020; Rogerio et al., 2023). ConclusionLeishmaniasis is an endemic disease in Iraq; dogs are the main reservoir host of the disease, which is infected without clinical signs; a high percentage of infectivity rate forebode is a risk to human beings and animals in Al Diwanyia province, especially. AcknowledgmentThe authors would like to thank the College of Veterinary Medicine at the University of Al-Qadisyiah for providing support for this work, the Veterinary teaching hospital for their valuable assistance, and Dr. Zoo clinic for providing animal samples. Conflict of interestThe authors declare no conflict of interest in the manuscript. ReferencesAl Obaidii, W.A. 2019. The serological diagnosis of canine Leishmaniasis by using ELISA in Nineveh province. Iraqi J. Vet. Sci. 33(2), 111–114; doi:10.33899/ijvs.2019.163194. Al Obaidii, W.A. and Al Mashhadany, D.A. 2023. Molecular detection of Leishmania in cutaneous scrapping and blood samples of dogs in Mosul city. Iraqi J. Vet. Sci. 37(Supplement I), 15–19; doi:10.33899/ijvs.2023.1386030.2815. Al-Ardi, M.H. 2022. Rapid diagnosis of Leishmania spp. in blood samples from dogs using gold nanoparticles. Iraqi J. Vet. Sci. 36(3), 587–590; doi:10.33899/ijvs.2021.130985.1906. Albuquerque, A., Campino, L., Cardoso, L. and Cortes, S. 2017. Evaluation of four molecular methods to detect Leishmania infection in dogs. Parasit. Vectors 10(57), 1–5; doi:10.1186%2Fs13071-017-2002-2. Al-Maeahi, A.M. and Marhoon, I.A. 2018. Parasitological survey of visceral leishmaniasis (Kala-azar) in Al-Diwanyiah province, Iraq. J. Pharm. Sci. Res. 10(12), 3146–3148. Available via https://www.researchgate.net/publication/339336304. AlSamarai, A.M. and AlObaidi, H.S. 2009. Cutaneous leishmaniasis in Iraq. J. Infect. Dev. Ctries. 3(2), 123–129; doi:10.3855/jidc.59. Alseady, H.H. and Al-Dabbagh, S.M. 2024. Isolation and molecular identification of cutaneous leishmaniasis in humans and dogs in middle Euphrates, Iraq. Iraqi J. Vet. Sci. 38(2), 427–435; doi:10.33899/ijvs.2023.143821.3259. Alwan, M.S. and Jassim, G.A. 2021. Epidemiological and genotypic study of cutaneous leishmaniasis in Al-Diwanyiah Province, Iraq. Plant Archives 21(1), 473–476; doi:10.51470/PLANTARCHIVES.2021.v21.S1.073. Costa, C.H.N., Chang, K.P., Costa, D.L. and Cunha, F.V.M. 2023. From infection to death: an overview of the pathogenesis of visceral Leishmaniasis. Pathogens 12(7), 1–25; doi:10.3390/pathogens12070969. da Silva, R.E., Sampaio, B.M., Tonhosolo, R., da Costa, A.P., da Silva Costa, L.E., Nieri-Bastos, F.A., Sperança, M.A. and Marcili, A. 2019. Exploring Leishmania infantum cathepsin as a new molecular marker for phylogenetic relationship and visceral leishmaniasis diagnosis. BMC Infect. Dis. 19, 859; doi:10.1186/s12879-019-4463-8. Eshetu, E., Awekew, A. and Bassa, T. 2016. The public health significance of Leishmaniasis: an overview. J. Nat. Sci. Res. 6(5), 48–57. Ferreira, B.A., Martins, T.F.C., Coser, E.M., Oliveira, V.L., Yamashshiro-Kanashiro, E.H., Rocha, M.C., Pinto, M.M., Cotrim, P.C. and Coelho, A.C. 2022. Isolation, typing, and drug susceptibility of Leishmania (Leishmania) infantum isolates from dogs of the municipality of Embo das Artes, and endemic region for canine leishmaniasis in Brazil. Parasitol. Res. 121:2683–2695; doi:10.1007/s00436-022-07594-5. Gani, Z.H., Hassan, M.K. and Jassim, AM 2010. Sero-epidemiological study of visceral leishmaniasis in Basrah, Southern Iraq. J. Pak. Med. Assoc. 60(6), 464–469. Garbi, M., Mhadhbi, M., Rejeb, A., Jaouadi, K., Rouatbi, M. and Darghouth, M.A. 2015. Leishmaniasis (Leishmania infantum infection) in dogs. Rev. Sci. Tech. 34(2), 613–626; doi:10.20506/rst.34.2.2384. Giraldo-Martínz, L.A., Petano-Duque, J.M., Uribe-García, H.F., Chacón-Novoa, R.A., Guzmán-Barragán, B.L. and Rondón-Barragán, I. 2022. High prevalence of Leishmania spp. in dogs from Central Wast Colombia. Vet. Ital. 58(4), 379–389. Lima, C., Colella, V., Latrofa, M.S., Cardoso, L., Otranto, D. and Ahlo, A.M. 2019. Molecular detection of Leishmania spp. in dogs and a cat from Doha, Qatar. Parasit. Vectors. 12(1), 125–128; doi:10.1186/s13071-019-3394-y. Marcili, A., da Silva, R.E., de Costa, V.P., Nieri-Bostos, F.A., Azevedo, R.C., Filho, J.M. and Tonhosolo, R. 2020. Canine Visceral Leishmaniasis in Sao Paulo, Brazil, the most populous city of South America: isolation, molecular diagnosis, and phylogenetic inferences. Vector Borne Zoonotic Dis. 20(10), 768–772; doi:10.1089/vbz.2020.2638. Medkour, H., Davoust, B., Dulieu, F., Maurizi, L., Lamour, T., Marie, J.L. and Mediannikov, O. 2019. Potential animal reservoirs (dogs and bats) of human visceral leishmaniasis due to Leishmania infantum in French Guiana. PLoS Negl. Trop. Dis. 19, 13(6), e0007456; doi:10.1371/journal.pntd.0007456. Najafi, L., Omidian, M., Rezaei, Z., Shahabi, S., Ghorbani, F., Arefkhah, N., Mohebali, M., Zaraei, Z. and Sarkari, B. 2021. Molecular and serological evaluation of zoonotic visceral leishmaniasis in dogs in a rural area of Fars province, southern Iran, as a source of Leishmania infantum infection. Vet. Med. Sci. 7(4), 1082–1089; doi:10.1002/vms3.432. Osorio-Peralta, D.C., Petano-Duque, J.M., and Rondón-Barragán, I.S. 2024. Leishmania spp. diagnosis and therabutic management in a cat from urban area in Ibagué (Colombia). Vet. Parasitol. Reg. Stud. Reports. 47, 100980; doi:10.1016/j.vprsr.2023.100980. Peacock, C.S., Seeger, K., Harris, D., Murphy, L., Ruiz, J.C., Quail, M.A., Peters, N., Adlem, E., Tivey, A., Aslett, M., Kerhornou, A., Ivens, A., Fraser, A., Rajandream, M.A., Carver, T., Norbertczak, H., Chillingworth, T., Hance, Z., Jagels, K., Moule, S., Ormond, D., Rutter, S., Squares, R., Whitehead, S., Rabbinowitsch, E., Arrowsmith, C., White, B., Thurston, S., Bringaud, F., Baldauf, S.L., Faulconbridge, A., Jeffares, D., Depledge, D.P., Oyola, S.O., Hilly, J.D., Brito, L.O., Tosi, L.R.O., Barrell, B., Cruz, A.K., Mottaram, J.C., Smith, D.F. and Berriman, M. 2007. Comparative genomic analysis of three Leishmania species that cause diverse human disease. Nat. Genet. 39(7), 839–847; doi: 10.1038%2Fng2053. Ribeiro, R.R., Marques Michalick, M.S., da Silva, M.E., dos Santos, C.C.P., Frezard, F.J.G. and da Silva, S.M. 2018. Canine leishmaniasis: an overview of the current status and strategies for control. Biomed. Res. Int. 2018, 3296893; doi:10.1155/2018/3296893. Rogerio, L.A., Takahashi, T.Y., Gardoso, L., Takamiya, N.T., Melo, E.V., Jesus, A.R., de Oliveira, F.A., Forrester, S., Jeffares, D.C., de Silva, J.S., Ribeiro, J.M., Almeida, R.P. and Maruyama, S.R. 2023. Co-infection of Leishmania infantum and Crithidia – related species in a case of refractory relapsed visceral leishmaniasis with non-ulcerated cutaneous manifestation in Brazil. Int. J. Infect. Dis. 133, 85–88; doi:10.1016/j.ijid.2023.05.012. Rombolà, P., Barlozzari, G., Carvelli, A., Scarpulla, M., Iacoponi, F. and Macrì, G. 2021. Seroprevalence and risk factors associated with exposure to Leishmania infantum in dogs, in an endemic Mediterranean region. PLoS One 16, e0244923; doi:10.1371/journal.pone.0244923. Rosypal, A.C. 2005. Characterization of Canine Leishmaniasis in the United States: pathogenesis, immunological responses, and transmission of an American isolate of Leishmania infantum. [Ph.D. dissertation]. Blacksburg, Virginia, North America. pp 125–131. Available via http://hdl.handle.net/10919/26873 Salant, H., Nachum-Biala, Y., Feinmesser, B., Perelmutter, M. and Baneth, G. 2021. Early onset of clinical leishmaniosis in a litter of pups with evidence of in utero transmission. Parasi. Vectors 14(1), 1–9; doi:10.1186/s13071-021-04824-0. Solano-Gallego, L., Cardoso, L., Pennisi, M.G., Petersen, C., Bourdeau, P., Oliva, G., Miro, G., Ferrer, L. and Baneth, G. 2017. Diagnostic challenges in the era of Canine Leishmania infantum Vaccines. Trends Parasitol. 33(9), 706–717; doi:10.1016/j.pt.2017.06.004. SPSS. 2006. 15.0 Command Syntax Reference, SPSS Inc., Chicago, Illinois, USA. Stecher, G., Tamura, K. and Kumar, S. 2020. Molecular evolutionary genetic analysis (MEGA) for macOS. Mol. Biol. Evol. 37(4), 1237–1239; doi:10.1093/molbev/msz312. Sundar, S. and Rai, M. 2002. Laboratory diagnosis of visceral leishmaniasis. Clin. Diagn. Lab. Immunol. 9(5), 951–958; doi:10.1128%2FCDLI.9.5.951-958.2002. Tafuri, W.L., De Oliveira, M.R., Melo, M.N. and Tafuri W.L. 2001. Canine visceral leishmaniosis: a remarkable histopathological picture of one case reported from Brazil. Vet. Parasitology. 96(3), 203–212; doi:10.1016/s0304-4017(00)00436-2. | ||
| How to Cite this Article |
| Pubmed Style Fadhil SA, Ali MJ. Molecular characterization of Leishmania infection in canine. Open Vet. J.. 2025; 15(2): 660-667. doi:10.5455/OVJ.2025.v15.i2.14 Web Style Fadhil SA, Ali MJ. Molecular characterization of Leishmania infection in canine. https://www.openveterinaryjournal.com/?mno=223635 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.14 AMA (American Medical Association) Style Fadhil SA, Ali MJ. Molecular characterization of Leishmania infection in canine. Open Vet. J.. 2025; 15(2): 660-667. doi:10.5455/OVJ.2025.v15.i2.14 Vancouver/ICMJE Style Fadhil SA, Ali MJ. Molecular characterization of Leishmania infection in canine. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 660-667. doi:10.5455/OVJ.2025.v15.i2.14 Harvard Style Fadhil, S. A. & Ali, . M. J. (2025) Molecular characterization of Leishmania infection in canine. Open Vet. J., 15 (2), 660-667. doi:10.5455/OVJ.2025.v15.i2.14 Turabian Style Fadhil, Samar Abbas, and Mansour Jadaan Ali. 2025. Molecular characterization of Leishmania infection in canine. Open Veterinary Journal, 15 (2), 660-667. doi:10.5455/OVJ.2025.v15.i2.14 Chicago Style Fadhil, Samar Abbas, and Mansour Jadaan Ali. "Molecular characterization of Leishmania infection in canine." Open Veterinary Journal 15 (2025), 660-667. doi:10.5455/OVJ.2025.v15.i2.14 MLA (The Modern Language Association) Style Fadhil, Samar Abbas, and Mansour Jadaan Ali. "Molecular characterization of Leishmania infection in canine." Open Veterinary Journal 15.2 (2025), 660-667. Print. doi:10.5455/OVJ.2025.v15.i2.14 APA (American Psychological Association) Style Fadhil, S. A. & Ali, . M. J. (2025) Molecular characterization of Leishmania infection in canine. Open Veterinary Journal, 15 (2), 660-667. doi:10.5455/OVJ.2025.v15.i2.14 |