| Research Article | ||
Open Vet. J.. 2025; 15(2): 680-689 Open Veterinary Journal, (2025), Vol. 15(2): 680-689 Research Article Prevalence of multidrug-resistant Vibrion species in fish in a reduction trial using lemon juice and sesame oilAlaa Eldin M.A. Morshdy1, Ghada M. Abd El Galil2, Rasha M. El Bayomi1, Amer Al Ali3, Rehab E. Mohamed4, Amany M. Shosha5 and Wageh Sobhy Darwish1*1Department of Food Hygiene, Safety, and Technology, Faculty of Veterinary Medicine, Zagazig University, Zagazig city, Egypt 2Department of Food Hygiene, Safety, and Technology, Faculty of Veterinary Medicine, Arish University, Arish city, Egypt 3Department of Medical Laboratory Sciences, College of Applied Medical Sciences, University of Bisha, Bisha, Saudi Arabia. 4Department of Zoonoses, Faculty of Veterinary Medicine, Zagazig University, Zagazig city, Egypt 55Bacterioloy, Mycology, Immunology Department, Faculty of Veterinary, Medicine, Mansoura University, Mansoura, Egypt. *Correspondence to: Wageh Sobhy Darwish. Department of Food Hygiene, Safety, and Technology, Faculty of Veterinary Medicine, Zagazig University, Zagazig city, Egypt. wagehdarwish [at] yahoo.ca Submitted: 11/10/2024 Accepted: 03/02/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
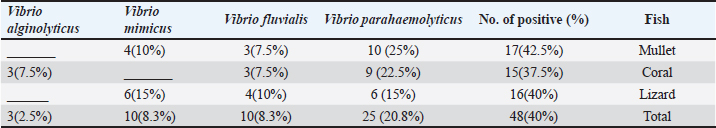
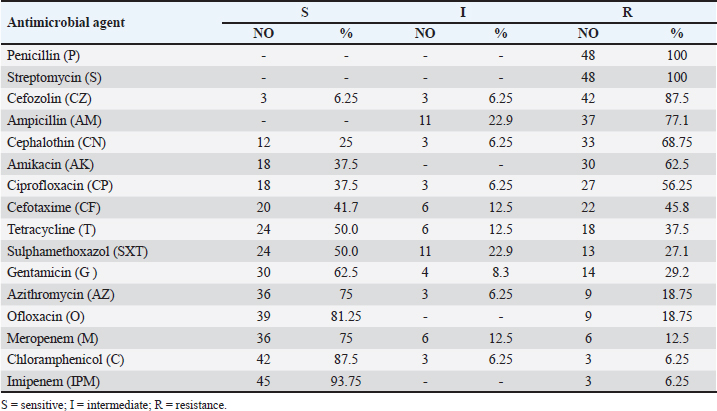
AbstractBackground: Because of its high nutritional value, excellent taste, and ease of digestion, fish is one of the most popular foods. However, it also serves as a vehicle for various pathogenic microorganisms, particularly Vibrio species, which pose a risk to public health. Aim: This study targeted studying the prevalence of multidrug-resistant Vibrio species in three marine fish retailed in Egypt. In addition, the antimicrobial activities of lemon juice and sesame oil against Vibrio parahaemolyticus were screened. Methods: For these reasons, this study assessed the prevalence of Vibrio species in marine fish (mullet, brush tooth lizard, and coral fish) that are sold in the local market of Zagazig, Egypt. Additionally, certain virulence factors were detected in the recovered V. parahaemolyticus isolates. The antimicrobial susceptibility of the recovered V. parahaemolyticus isolates was also screened. The antivibrio activities of lemon juice and sesame oil (1% and 2%) were additionally tested. Results: The acquired data showed that the prevalence of Vibrio species was 40% among the tested fish samples. V. parahaemolyticus was isolated at 20.8%. Vibrio fluvialis (8.3%), Vibrio Mimicus (8.3%), and Vibrio alginolyticus (2.5%). The recovered isolates were highly resistant to cefazolin (87.5%), ampicillin (77.1%), cephalothin (68.75%), penicillin, and streptomycin (100%, each). Lemon juice and sesame oil had marked antibacterial activities against V. parahaemolyticus. Conclusion: Fish dipping in lemon juice and sesame oil was an effective way to lower the load of V. parahaemolyticus in fish. In addition, sanitary measures must be implemented to prevent microbial contamination in fish markets and aquatic environments. Keywords: Fish, Vibrio parahaemolyticus, Antibiotic sensitivity, Virulence genes, Lemon juice, Sesame oil IntroductionThe nutritional significance of fish has led to a simultaneous increase in consumption during the past ten years (Morshdy et al., 2022a). Fish is a perishable, high-protein food that contains essential amino acids, vitamins, minerals, and Omega-3 fatty acids, all of which are vital to human life (Morshdy et al., 2023). According to Ibrahim et al. (2018), polyunsaturated fatty acids found in fish protect consumers from coronary heart disease by lowering the risk of thrombosis, arrhythmias, fatal heart attacks, and sudden death. Given that fish have lower feed conversion rates, shorter production cycles, and lower rearing and feeding costs than other animal proteins, they may be able to contribute to solving the animal protein shortage issue (Liu et al., 2018). Marine fish, particularly the mullet, brush-tooth lizard, and coral fish, can easily harbor pathogenic microorganisms because of their filter-feeding nature and the microbial overcrowded habitat, particularly of the Vibrio species (Austin and Austin, 2012; Caburlotto et al., 2016). Regarding human diseases, the three most significant Vibrio species are cholerae, parahaemolyticus, and vulnificus. The clinical symptoms of Vibrio species vary according to the species in question (Tsai et al., 2013). An important human disease caused by seafood is Vibrio parahaemolyticus. Headache, nausea, vomiting, abdominal cramps, diarrhea, and low temperature are symptoms of acute gastroenteritis, which can be caused by raw or undercooked seafood contaminated with V. Parahaemolyticus (Chao et al., 2010). The overuse of antibiotics in medicine and agriculture has led to an increase in bacteria resistant to these drugs (Moore et al., 2014). Molecular methods such as polymerase chain reaction (PCR) assays are widely used to identify the harmful V. parahaemolyticus strain in environmental and dietary samples (Zaafrane et al., 2022). From a food safety perspective, it is essential to monitor microbiological contamination of seafood continuously. Food shelf-life, safety, and marketability can all be increased by using natural antibacterial substances to preserve the product’s fresh appearance (Jun et al., 2012). Researchers and consumers alike have become interested in natural bacteriostatic agents as concerns over artificial food preservation additive safety and consumer health have grown (Yassin et al., 2022). As secondary metabolites, plants produce phenolic chemicals, and certain plant phenolics or phenolic-rich extracts have been shown to remarkably inhibit a wide spectrum of bacteria (Darwish et al., 2020). Phenolic compounds can be used as seasonings to enhance product flavor and microbial quality (Mahmoud et al., 2021). According to Yasin et al. (2022) and Takó et al. (2020), phenolic compounds can alter the shape of bacteria, weaken bacterial cell walls, and affect biofilm formation. Sesamol is a phenolic compound that is found in sesame oil, meal, and seeds. It has been shown to have antioxidant, immunomodulatory, anticancer, antiangiogenic, and microbiological properties (Xie et al., 2021; Zhou et al., 2021). The principal organic acid in lemon juice, citric acid, is used to flavor and preserve food. Antioxidant and antibacterial properties make it a natural meat preservative (Nawi et al., 2017). The antibacterial activity of sesame oil against Vibrio species has not been investigated. Thus, this study assessed the prevalence of Vibrio species in fish samples collected from markets in the Sharkia Governorate, Egypt. The study involved biochemical identification, virulence gene detection, and antibiotic resistance profiling in Vibrio isolates. The antibacterial properties of sesame oil and lemon juice against V. parahaemolyticus were also investigated. Material and MethodsCollection and preparation of samplesA total of 120 fish samples were collected at random from different fish stores in Zagazig City, Sharkia Governorate, Egypt. The samples included 40 mullets (Mugil cephalus) (average weight 325 ± 40 g), brush- tooth lizards (Saurida undosquamis) (average weight 125 ± 30 g), and Coral fish (Pagrus major) (average weight 230 ± 40 g) (forty of each). The samples were promptly marked and forwarded to the Faculty of Veterinary Medicine, Laboratory of Meat Hygiene, Zagazig University, Egypt, for bacterial isolation and identification upon collection. The inedible fins were removed, the surface of the fish was sterilized with a hot spatula, and muscle samples were collected from each examined fish. Isolation of Vibrio speciesFollowing guidelines set out by the Food and Drug Administration (FDA, 2004), Vibrio species was successfully isolated. After meticulously measuring out 90 ml of sterile alkaline peptone water (APW, Micro Master, India) and 10 g of fish meat, the two were mixed together. Subsequently, according to the International Standardization Organization (ISO/TS, 21872-1, 2007), the mixture was kept at a temperature of 35 ± 2°C 2°C for 24–48 hours. A loopful of each APW broth was placed on thiosulfate citrate bile salt sucrose agar plates (TCBS, Hi-Media, India), and the plates were left to sit at 37°C for an entire day. After being picked, washed, and biochemically identified (arginine dihydrolase, lysine decarboxylase, ornithine decarboxylase), citrate, and D-glucosamine utilization, Voges-Proskauer tests and growth in 8% NaCl were performed), colonies having a yellowish-green or greenish-blue hue were analyzed according to ISO/ TS, 21872-1 (2007) and ISO/TS, 21872-2 (2007). The detection of somatic (O) and capsular (K) antigens was performed using V. parahaemolyticus antiserum (Hardy Diagnostics Co., USA) in accordance with the methods described by Ansaruzzaman et al. (2005). Antimicrobial susceptibility testingIn accordance with Adeleye et al. (2008), the Kirby- Bauer disc diffusion method was used to assess the antimicrobial susceptibility of 48 Vibrio isolates to 16 different antibiotics: penicillin (P), Streptomycin (S), Cefozolin (CZ), Ampicillin (AM), Cephalothin (CN), ceprofloxacin (CP), Cefotaxime (CF), Tetracycline (T), Sulphamethoxazol (SXT), Gentamicin (G), Azithromycin (AZ), Ofloxacin (O), meropenem (M), and Chloramphenicol (C). Following the National Committee for Clinical Laboratory Standards (NCCLS’s) guides (NCCLS, 2001), inhibitory zones were located. Molecular characterization of V. parahaemolyticus isolatesFor bacterial DNA extraction, a QIAamp DNA Mini kit (Catalog no. 51304) was used by QIAGEN (GmbH, Hilden, Germany). The thermolabile hemolysin (tlh), thermostable direct hemolysin (tdh), and thermostable related hemolysin (trh) genes amplification was performed using primers supplied by Metabion (Germany). The primer preparation, PCR cycling conditions, and electrophoresis were according to (Bej et al., 1999). Lemon juice and sesame oil as natural decontaminantsIn the experimental trial, 25 mullet fish of the same size were used. Each fish was inoculated in the back muscle with 1 ml of V. parahaemolyticus broth, which was adjusted to 0.5 McFarland (6 log 10 cfu/ml). The fish that inoculated was maintained at room temperature (25°C) for half an hour. The inoculated fish were then split into groups and dipped in solutions made with two concentrations (1% and 2%) of lemon juice and sesame oil (Nawi et al., 2017; Morshdy et al., 2022a). The negative control was dipped in sterile distilled water. Statistical analysisThe reduction counts were converted into log 10 relevant values. Values represent means ± SD. On- wayy analyses of variance followed by the posthock Tukey’s Kramer HSD test were performed using JMP software provided by SAS institute, USA, with p<0.05 being deemed significant. ResultsThe prevalence of Vibrio species in the analyzed fish species, including mullets, brush-tooth lizards, and coral fish, is presented in Table 1. The bacteriological analysis of the fish samples indicated that Vibrio species were found in 40% of all the samples analyzed. V. parahaemolyticus was found in 25 of the 120 samples tested (20.8%), Vibrio fluvialis was found in 10 samples (8.3%), Vibrio mimicus was found in 10 samples (8.3%), and Vibrio alginolyticus was found in 3 samples (2.5%). The highest isolation rate was observed in mullets (42.5%), whereas the lowest was recorded in coral fish (37.5%). Table 2 shows that all of the Vibrio isolates tested were completely resistant to penicillin and streptomycin (100%). They are also resistant to cefazolin (87.5%), ampicillin (77.1%), and cephalothin (68.75%). Table 1. Prevalence of Vibrio species isolated from fish samples (N = 40 of each).
Table 2 Antimicrobial susceptibility of the Vibrio species (N = 48).
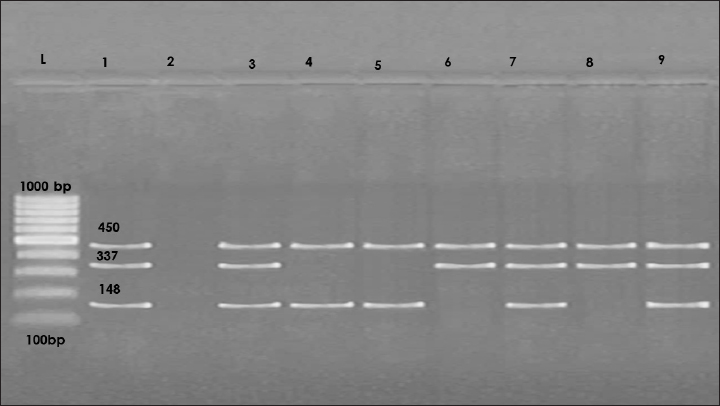
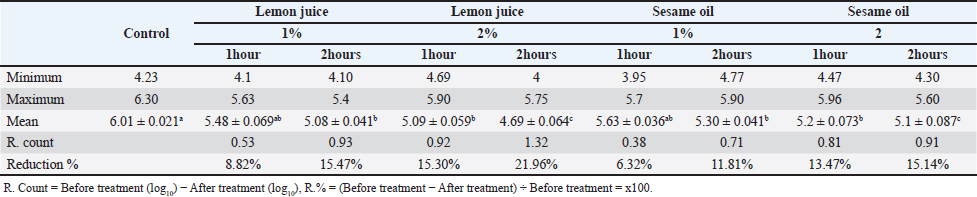
Concurrently, Vibrio species exhibited greater sensitivity to Imipenem (93.75%), Chloramphenicol (87.5%), and Meropenem (75%). tlh was identified in all examined V. parahaemolyticus isolates. The th and trh virulence genes were identified in 71.5% of the analyzed isolates (Fig. 1). This study evaluated the antibacterial efficacy of 1% and 2% lemon juice and sesame oil against V. parahaemolyticus. The findings indicate that the reduction counts of V. parahaemolyticus were 0.53, 0.93, 0.92, and 1.32, with reduction percentages of 8.82%, 15.47%, 15.30%, and 21.96%, respectively, following immersion in fresh lemon juice extract 1% and 2% for 1 and 2 hours. In addition, this study investigated how well sesame oil at 1% and 2% concentrations reduced bacterial counts as a possible method of decontaminating raw fish infected with V. parahaemolyticus. The results indicated that the reduction counts of V. parahaemolyticus were 0.38, 0.71, 0.81, and 0.91 with reduction percentages of 6.32, 11.81, 13.4, and 15.14 following immersion in fresh sesame oil 1% and 2% for 1 and 2 hours, respectively (Table 3). The results showed a positive correlation between the number of reductions and the time of exposure. In addition, after immersion in either 2% sesame oil or lemon juice for 2 hours, V. parahaemolyticus exhibited a significant decrease (p < 0.05). Results showed that fish treated with a 2-hours solution of lemon juice and sesame oil had a much lower prevalence of V. parahaemolyticus. Lemon juice had a much stronger reduction effect against V. parahaemolyticus. DiscussionThe risk of health problems increases when eating seafood contaminated with Vibrio spp., an aquatic pathogen (Morshdy et al., 2023). Two main factors make marine fish susceptible to contamination by different microorganisms: (1) exposure to contaminated water; and (2) post-harvest contamination caused by improper handling, transportation, marketing, and storage. This kind of contamination can make fish very dangerous to eat or even unfit for human consumption (Sugumar et al., 2008). Water, soil, and people who handle the fish are some of the many potential causes of microbial contamination. According to Mahmoud et al. (2022), improper handling of fish during fishing, transportation, and shipping greatly increases the risk of cross-contamination and acts as a stressor, making it easier for germs to move from the fish’s stomach to its muscles. Contamination of the tested fish likely occurred during transportation and sale, as evidenced by the isolation of Vibrio species from the samples. Vibrio species thrive in environments where fish are maintained at room temperature. The prevalence rates of Vibrio species in the current investigation were consistent with studies done in other countries. While 72.7% of the fish samples examined in Turkey tested positive for Vibrio species, only 59% of the fish samples in Malaysia tested positive (Radu et al., 2003; Yu et al., 2016). V. parahaemolyticus, V. fluvialis, and V. mimicus were the three most studied species of Vibrio species in fish-related studies (Morshdy et al., 2022b). One hundred sixty-six (44%) of the fish samples tested positive for Vibrio species in Morshdy et al. (2021b). Vibrio cholerae was found in just one sample (0.7%), while V. parahaemolyticus was present in 28 samples (18.6%). Similarly, El-Sharaby et al. (2018) recorded an isolation rate of 49% for the Vibrio species in Egypt’s freshwater fish. Vibrio species were found in 52% of the fish samples sold in Egypt, according to Morshdy et al. (2022a), among which 42.3% were V. parahaemolyticus, 26.92% were V. mimicus, 9.62% were R. Count = Before treatment (log10) –After treatment (log10), R.% = (Before treatment –After treatment) ÷ Before treatment = x100. V. alginolyticus, and 1.92% were V. cholera. According to Suresh et al. (2018), the majority of freshwater fish tested positive for V. parahaemolyticus, whereas a small percentage tested positive for V. vulnificus, 4.6% for V. alginolyticus, and 3.45% for V. cholera. Crustaceans sold in Egypt’s Sharkia Governorate tested positive for 36 Vibrio isolates, representing 16% of the total. Among them, V. parahaemolyticus accounted for 15.1% and V. cholerae for 0.9% (Ahmed et al., 2018). Freshwater fish in China were found to contain 10.33% V. cholerae, 3.89% V. parahaemolyticus, 1.24% V. alginolyticus, and 0.76% V. vulnificus, as reported by Yan et al. (2019). According to Jakšić et al. (2002), shrimp samples from Croatia had lower prevalence rates of V. parahaemolyticus (7.14%), V. alginolyticus (3.57%), and V. vulnificus (10.71%). The highest prevalence of V. parahaemolyticus in Selangor shrimp samples, Malaysia, was reported by Tan et al. (2020) at 88.57%. In Thailand, 38% of farmed shrimp samples tested positive for V. parahaemolyticus, whereas 26.67% of Chinese seafood and shrimp tested positive, according to research by Yano et al. (2014) and Jiang et al. (2019), respectively. In addition, Yücel and Balci (2010) discovered that V. parahaemolyticus, V. vulnificus, and V. mimicus were present in marine fish from Turkey at proportions of 40%, 18%, and 6%, respectively. Salinity, geographical location, seasonal fluctuations, and isolation procedures can affect the prevalence of different Vibrio species (Morshdy et al., 2023). Correlations of Vibrio species with environmental factors like temperature and salinity may shed light on why these organisms are so common. The prevalence of V. parahaemolyticus increased during the warmer months, which can be attributed to the higher temperatures (Morshdy et al., 2022a). In addition, microbes may enter a latent state in the bottom sediments and then reemerge into the water column when high temperatures resume (Pfeffer et al., 2003).
Fig. 1. Representative image for multiplex polymerase chain reaction for 1.5% agarose gel for V. parahaemolyticus characterization; trh (148 bp), tdh (337 bp) and tlh (450 bp). M: A ladder of 100 base pairs, 1: Positive control; 2: Negative control, 3, 7, 9: positive strains for trh, tdh and tlh genes, 4 & 5: positive strain for trh and tlh genes, 6 & 8: positive strains for tdh, and tlh, genes. Table 3. Effects of lemon juice and Sesame oil on Vibrio parahaemolyticus counts log10cfu/g in fish after varying exposure periods.
Antibiotic residues in aquaculture products are associated with serious public health risks. Consistent with previous research, this investigation demonstrated that V. parahaemolyticus is resistant to many medications in Malaysia (Letchumanan et al., 2015). A majority of these antibiotics were ampicillin (82%), followed by amikacin (51%), cefotaxime (37%), gentamicin (11%), ceftazidime (15%), levofloxacin (9%), and imipenem (2%). A large portion of the seafood imported into China was discovered to be resistant to the following antibiotics: ampicillin (95.46%), amikacin (30%), tetracycline (17.78%), sulphamethoxazole/trimethoprim (17.78%), and ciprofloxacin (2.22%), according to Jiang et al. (2019). Tan et al. (2020) reported that V. parahaemolyticus strains in Selangor, Malaysia, were not sensitive to ampicillin (84.17%), cephalothin (54.17%), amikacin (37.50%), ciprofloxacin (13.33%), gentamicin (6.67%), cefotaxime (5%), ceftazidime (5%), or levofloxacin (1.67%). A study was conducted in the region. In the same way, Jiang et al. (2019) found that imipenem and meropenem worked well to treat all V. parahaemolyticus isolates obtained from different types of Chinese seafood. According to Tan et al. (2020), V. parahaemolyticus is resistant to tetracycline (94.17%), imipenem and meropenem (98.33%), levofloxacin (73.33%), ceftazidime (70.83%), gentamicin (64.17%), and levofloxacin (73.33%). The results demonstrate the importance of regularly testing different types of Vibrio found in nature against a large number of antimicrobial drugs and treatments that are used in medicine. In this way, useful baseline data can be collected in the laboratory, which can be used in real-life therapy (Baker-Austin et al., 2008). Several studies from around the world have found a link between V. parahaemolyticus isolates that have tdh and/or trh genes and their ability to cause disease (Nordstrom et al., 2007; Ahmed et al., 2018). According to Morshdy et al. (2023), the vast majority of V. parahaemolyticus strains found in the environment do not cause disease. However, when it comes to clinical isolates, 99% are harmful because they include the tdh and/or trh genes. Although a small percentage of environmental isolates carrying the tdh and/or trh genes are considered hazardous, this number is still rather low (Letchumanan et al., 2014). For several reasons, it is important to identify tdh and/or trh V. parahaemolyticus in freshwater and marine fish samples. These infectious microbes may initially cause gastrointestinal illness (Jun et al., 2012). Additionally, according to Thongjun et al. (2013), pathogenic V. parahaemolyticus causes significant economic losses in the aquaculture sector due to disease propagation and contamination of seafood. Unlike a previous study by Letchumanan et al. (2015), which found the presence of trh and tdh in V. parahaemolyticus isolates from Malaysian shrimp sales 10% of the time, the present investigation revealed a higher number of virulent genes (tdh and trh) in V. parahaemolyticus strains. Although Tan et al. (2020) reported that V. parahaemolyticus strains found in seafood samples from Selangor, Malaysia, did not have the harmful tdh and trh genes, Jiang et al. (2019) also detected virulence-associated genes in the recovered V. parahaemolyticus isolates from Chinese seafood, with a frequency of 1.11% for tdh and 5.56% for trh. Among 165 V. parahaemolyticus isolates from marine and freshwater fish in Selangor, Malaysia, in 2018, only 4 (2.4%) exhibited evidence of the trh gene. They were all negative for tdh. One in fourteen V. parahaemolyticus isolates from Egyptian crustaceans harbored the tdh and/or trh genes (Ahmed et al., 2018). Among the V. parahaemolyticus strains detected in Chinese seafood, 28.4% carried the trh gene and 2.1% carried the tdh gene. Xie et al. (2017) reported that no isolate possessed both genes simultaneously. In China, researchers conducted two separate experiments. Xie et al. (2015) reported that among all the V. parahaemolyticus isolates that tested positive for the tdh gene, 45.9% possessed the trh gene. Some genes, like OmpU, hlyA, and rtxA-D, make V. cholerae more harmful (Rivera et al., 2001; Ruenchit et al., 2017; Morrishdy et al., 2023). An infection with ambient V. cholerae can cause mild gastroenteritis if the hlyA gene is present; however, most of these isolates are not lethal (Saravanan et al., 2007). Two samples of V. cholerae isolated from Egyptian crustaceans included the tdh and hlyA genes. According to Ahmed et al. (2018), these samples did not originate from the O1 or O139. The rtxC and hylA virulence genes of V. cholerae were detected in abundance in freshwater fish from Shanghai, China, according to Xu et al. (2019). Both were present at 95.8% and 87.8% of the time. Lemon juice demonstrated promising results for lowering V. parahaemolyticus counts in the experiment. Lemon Juice reduced V. parahaemolyticus counts at 8.82%, 15.47%, 15.30%, and 21.96%, respectively, following immersion in 1% and 2% juice for 1 and 2 hours, respectively. In the same way, fish infected with V. parahaemolyticus were immersed in 5% lemon juice for 0.5, 1, and 2 hours, respectively. This led to much higher decrease rates of 45.08%, 51.23%, and 62.08% (Morshdy et al., 2022). At the same time, Sushmita (2022) and Tsai et al. (2021) proved that lemon juice may treat V. cholerae, while Nawi et al. (2017) proved that lemon juice was effective against V. parahaemolyticus, making it a natural decontaminant for seafood. In addition, when tilapia filets were marinated in lime juice at 25°C for 30 minutes and 120 minutes, Ibrahim et al. (2018) and Kato et al. (2018) discovered that the counts of V. parahaemolyticus were dramatically reduced. The effects of 1% lemon essential oil were studied by Morshdy et al. (2021b), who reported its notable antibacterial activities. The main reasons lemthat lemonice is so populare its low cost, ease of availability, and lack of negative adverse effects. The high levels of bioflavonoids, limonene, pectin, calcium, magnesium, and vitamins in lemon juice improve its antiviral, antibacterial, and immune-boosting properties (Alsaraf et al., 2016). Sesame oil was used in this investigation because sesame oil is inexpensive and has inherent antioxidant properties. Using different amounts of vegetable oil, the antioxidant and oxidation activities were evaluated over several hours. Adding more vegetable oil boosts antioxidant activity, which makes fish less oxidatively stable and allows for longer storage times. The obtained results indicated that the reduction rates of V. parahaemolyticus were 6.32%, 11.81%, 13.4%, and 15.14% following immersion in fresh sesame oil 1% and 2% for 1 and 2 hours, respectively. The sesame oil was likely to have a clear antimicrobial effect in several reports. On days 4, 7, and 15, Anwer et al. (2021) found values (log 10 cfu/g) of 4.19 ± 0.85, 4.89 ± 0.75, and 5.69 ± 0.53, respectively. For 1, 2, and 5 days, the values recorded by Vanitha et al. (2015) were 3.91, 4.67, and 6.1, respectively. The results of this investigation are consistent with those of Yerlikaya et al. (2005) and Yazgan (2013), who observed that plant oils can effectively improve the microbiological, sensory, and chemical qualities of fish. Future studies are needed to investigate the antimicrobial activities of other plant oils and the mechanisms underlying these effects. ConclusionThe present study revealed that the marketed mullet, brush-tooth lizard, and coral fish might act as potential sources of Vibrio species. In particular, V. parahaemolyticus was found at a higher prevalence rate of 20.8%. Using lemon juice and sesame oil could reduce the load of V. parahaemolyticus, particularly at 2%. Efficient sanitary measures are highly recommended during fish handling, starting from catching, storage, and marketing. AcknowledgmentsThe authors are thankful to the Deanship of Graduate Studies and Scientific Research at the University of Bisha for supporting this work through the Fast-Track Research Support Program. Conflict of interestNone. Authors’ contributionsAll authors made an equal contribution. ReferencesAdeleye, A., Vivian, E., Rita, N., Stella, S. and Emamanuel, O. 2008. Antimicrobial susceptibility of potentially pathogenic halophilic Vibrio species isolated from seafoods in Lagos, Nigeria. Afri. J. Biotechnol. 7(20), 3791–4. Ahmed, H.A., El Bayomi, R.M., Hussein, M.A., Khedr, M.H., Remela, E.M.A. and El-Ashram, A.M. 2018. Molecular characterization, antibiotic resistance pattern and biofilm formation of Vibrio parahaemolyticus and V. cholerae isolated from crustaceans and humans. Int. J. Food Microbiol. 274, 31–7. Alsaraf, K.M., Abd, S.T., Hussein, N.S. and Jabor, A.A. 2016. In vitro evaluation the effect of natural products against Vibrio cholerae. Int. J. Adv. Res. 3, 271–5. Ansaruzzaman, M., Lucas, M., Deen, J.L., Bhuiyan, N.A., Wang, X.Y., Safa, A., Sultana, M., Chowdhury, A., Nair, G.B., Sack, D.A. and Von Seidlein, L. 2005. Pandemic serovars (O3: K6 and O4: K68) of Vibrio parahaemolyticus associated with diarrhea in Mozambique: spread of the pandemic into the African continent. J. Clin. Microbiol. 43(6), 2559–62. Anwer, I., Abbas, F., Hafeez-ur-Rehman, M., Nadeem, M., Khalil, M., Butt, M. and Inayat, M., 2021. Nanoemulsion of Virgin Rapeseed and Sesame Oil improve Oxidation Stability and Sensoric acceptability of Vacuum Packed Fish Fillets. Research Square. https://doi.org/10.21203/rs.3.rs-152180/v1 Austin, B. and Austin, D.A. 2012. Bacterial fish pathogens, disease of farmed and wild fish, 4th ed. Godalming, UK: Springer Praxis. Baker-Austin, C., McArthur, J.V., Tuckfield, R.C., Najarro, M., Lindell, A.H., Gooch, J.A.N. and Stepanauskas, R. 2008. Antibiotic resistance in the shellfish pathogen Vibrio parahaemolyticus isolated from the coastal water and sediment of Georgia and South Carolina, USA. J. Food Prot. 71, 2552–8. Bej, A.K., Patterson, D.P., Brasher, C.W., Vickery, M.C., Jones, D.D. and Kaysner, C.A. 1999. Detection of total and hemolysin-producing Vibrio parahaemolyticus in shellfish using multiplex PCR amplification of tl, tdh, and trh. J. Microbiol. Methods 36, 215–25. Caburlotto, G., Suffredini, E., Toson, M., Fasolato, L., Antonetti, P., Zambon, M. and Manfrin, A. 2016. Occurrence and molecular characterization of Vibrio parahaemolyticus in crustaceans commercialized in Venice area, Italy. Int. J. Food Microbiol. 220, 39–49. Chao, G.X., Jiao, X.N., Zhou, X.H., Wang, F., Yang, Z.Q. and Huang, J.L. 2010. Distribution of genes encoding four pathogenicity islands VPaIs., T6SS, Biofilm, and type I Pilus in food and clinical strains of Vibrio parahaemolyticus in China. Foodborne Pathog. Dis. 76, 649e658. Darwish, A., Ahmed, S. and Aboel-Ainin, M., 2020. Nutritional properties and antioxidant activity of seven sweet potato cultivars and clones (Ipomoea batatas L.). Scient. J. Agri. Sci., 2(2), 123–136. El-Sharaby, S.M.A., Abd-Elgaber, M., Tarabees, R., Khalil, R.H., Ali, M.N. and El-Ballal, S. 2018. Bacteriological and histopathological studies on Vibrio species isolated from naturally infected freshwater fish in delta region. Egypt. Adv. Anim. Vet. Sci. 6, 17–26. FDA’s Bacteriological Analytical Manual. 2004. Bacteriological analytical manual food and drug administration. BAM chapter 9: Vibrio. Available via https://www.fda.gov/food/laboratory-methods-food/bam-chapter-9-Vibrio Ibrahim, H.M., Amin, R.A. and Ghanaym, H.R. 2018. Effect of marination on Vibrio parahaemolyticus in tilapia fillets. Benha Vet. Med. J. 34, 234–45. ISO/TS, 21872-1. 2007. Specifies a horizontal method for the detection of the two main pathogenic Vibrio species causing intestinal illness in humans: V. parahaemolyticus and V. cholerae. Available via https://www.isoorg/standard/38278.html ISO/TS, 21872-2. 2007. Specifies a horizontal method for detection of the enteropathogenic Vibrio species, causing illness in or via the intestinal tract, other than Vibrio parahaemolyticus and Vibrio cholerae. Include Vibrio fluvialis, Vibrio mimicus, and Vibrio vulnificus. Available via https://www.iso.org/standard/38279.html Jakšić, S., Uhitil, S., Petrak, T., Bažulić, D. and Karolyi, L.G. 2002. Occurrence of Vibrio spp. in sea fish, shrimps and bivalve molluscs harvested from Adriatic sea. Food Control 13, 491–3. Jiang, Y., Chu, Y., Xie, G., Li, F., Wang, L., Huang, J., Zhai, Y. and Yao, L. 2019. Antimicrobial resistance, virulence and genetic relationship of Vibrio parahaemolyticus in seafood from coasts of Bohai Sea and Yellow Sea, China. Int. J. Food Microbiol. 290, 116–24. Jun, J.W., Kim, J.H., Choresca, Jr., C.H., Shin, S.P., Han, J.E., Han, S.Y., Chai, J.Y. and Park, S.C. 2012. Isolation, molecular characterization, and antibiotic susceptibility of Vibrio parahaemolyticus in Korean seafood. Foodborne Pathog. Dis. 9, 224–31. Kato, Y., Otsubo, K., Furuya, R., Suemichi, Y. and Nagayama, C. 2018. Bacteriostatic effect of lemon fruit juice: it’s potential as an oral rinsing agent. J. Oral Hyg. Health 6, 2332-0702. Letchumanan, V., Chan, K.G. and Lee, L.H. 2014. Vibrio parahaemolyticus: a review on the pathogenesis, prevalence, and advance molecular identification techniques. Front. Microbiol. 5, 705–18. Letchumanan, V., Yin, W.F., Lee, L.H. and Chan, K.G. 2015. Prevalence and antimicrobial susceptibility of Vibrio parahaemolyticus isolated from retail shrimps in Malaysia. Front. Microbiol. 6, 33–44. Liu, X., Lu, S., Guo, W., Xi, B. and Wang, W. 2018. Antibiotics in the aquatic environments: a review of lakes, China. Sci. Total Environ. 627, 1195–208. Mahmoud, A.F.A., Elshopary, N., El-Naby, G.H. and El Bayomi, R. 2021. Reduction of biogenic amines production in chilled minced meat using antimicrobial seasonings. J. Microbial. Biotechnol. Food Sci. 10(6), e3663. Mahmoud, A.F., Hafez, A.E., Seadawy, H.G., Mohamed, E.F., Abdallah, K.M., 2022. Quality Assessment and Impact of Gamma Irradiation on Histamine Content in Some Fish Consumed in Sharkia Province, Egypt. J. Adv. Vet. Res., 12, 760-767. Moore, S., Thomson, N., Mutreja, A. and Piarroux, R. 2014. Widespread epidemic cholera caused by a restricted subset of Vibrio cholerae clones. Clin. Microbiol. Infect. 20, 373–9. Morshdy, A.E., Abdelhameed, N.S., Abdallah, K., El-tahlawy, A.S. and El Bayomi, R.M. 2023. Antibiotic resistance profile and molecular characterization of Vibrio parahaemolyticus and Vibrio cholerae isolated from fish. J. Adv. Vet. Res. 13, 3. Morshdy, A.E.M., El-Ghandour, A.R., Hussein, M.A. and El Bayomi, R.M. 2022a. Prevalence of antibiotic-resistant Vibrio isolated from some marketed fish in egypt with decontamination trial by lemon juice. J. Adv. Vet. Res. 12, 353–7. Morshdy, A.E.M., Hussein, M.A., Mohamed, M.A.A., Hamed, E., El-Murr, A.E. and Darwish, W.S. 2022b. Tetracycline residues in tilapia and catfish tissue and the effect of different cooking methods on oxytetracycline and doxycycline residues. J. Cons. Prot. Food Saf. 17, 387–93. Morshdy, A.E.M.A., Nahla, B.M., Shafik, S. and Hussein, M.A. 2021b. Antimicrobial effect of essential oils on multidrug-resistant Salmonella typhimurium in chicken fillets. Pak. Vet. J. 41, 545–51. Nawi, S.F.A.M., Zain, Z.M., Zahari, M.Z., Hamid, A.A.A., Afandi, N.F.A., Fadzilah, N.Z.H. and Azmi, S.N.F. 2017. The inhibitory effect of lemon juice (Citrus limon) on Vibrio parahaemolyticus in Raw Oyster (Crassostrea virginica). J. Clin. Health Sci. 2, 31–3. NCCLS (National Committee for Clinical Laboratory Standards). 2001. Performance standards for antimicrobial susceptibility testing. Supplement M100-S11. Wayne, Pa: Clinical and Laboratory Standards Institute (CLSI). Nordstrom, J.L., Vickery, M.C., Blackstone, G.M., Murray, S.L. and DePaola, A. 2007. Development of a multiplex real-time PCR assay with an internal amplification control for the detection of total and pathogenic Vibrio parahaemolyticus bacteria in oysters. Appl. Environ. Microbiol. 73, 5840–7. Pfeffer, C.S., Hite, M.F. and Oliver, J.D. 2003. Ecology of Vibrio vulnificus in estuarine waters of eastern North Carolina. Appl. Environ. Microbiol. 69, 3526–31. Radu, S., Ahmad, N., Ling, F.H. and Reezal, A. 2003. Prevalence and resistance to antibiotics for Aeromonas spp. from retail fish in Malaysia. Int. J. Food Microbiol. 81, 261–6. Rivera, I.N., Chun, J., Huq, A., Sack, R.B. and Colwell, R.R. 2001. Genotypes associated with virulence in environmental isolates of Vibrio cholerae. Appl. Environ. Microbiol. 67, 2421–9. Ruenchit, P., Reamtong, O., Siripanichgon, K., Chaicumpa, W. and Diraphat, P. 2017. New facet of non-O1/non-O139 Vibrio cholerae hemolysin A: a competitive factor in the ecological niche. FEMS Microbiol. Ecol. 93, 1–12. Saravanan, V., Sanath Kumar, H., Karunasagar, I. and Karunasagar, I. 2007. Putative virulence genes of Vibrio cholerae from seafoods and the coastal environment of Southwest India. Int. J. Food Microbiol. 119, 329–33. Sugumar, G., Chrisolite, B., Velayutham, P., Selvan, A. and Ramesh, U. 2008. Occurrence and seasonal variation of bacterial indicators of faecal pollution along Thoothukudi coast, Tamil Nadu. J. Environ. Biol. 29(3), 387–91. Suresh, Y., Subhashini, N., Kiranmayi, C.B., Srinivas, K., Ram, V.P., Chaitanya, G. and Rao, T.S. 2018. Isolation, molecular characterization and antimicrobial resistance patterns of four different Vibrio species isolated from fresh water fishes. Int. J. Curr. Microbiol. Appl. Sci. 7, 3080–8. Sushmita, D. 2022. Vibrio cholerae Food Poisoning- Cholera toxin (CT). Microbe Notes. Available via https://microbenotes.com/Vibrio-cholerae-food-poisoning/ Takó, M., Kerekes, E.B., Zambrano, C., Kotogán, A., Papp, T., Krisch, J. and Vágvölgyi, C. 2020. Plant phenolics and phenolic-enriched extracts as antimicrobial agents against food-contaminating microorganisms. Antioxidants 9(2), 165. Tan, C.W., Rukayadi, Y., Hasan, H., Thung, T.Y., Lee, E., Rollon, W.D., Hara, H., Kayali, A.Y., Nishibuchi, M. and Radu, S. 2020. Prevalence and antibiotic resistance patterns of Vibrio parahaemolyticus isolated from different types of seafood in Selangor, Malaysia. Saudi J. Biol. Sci. 27, 1602–8. Thongjun, J., Mittraparp-Arthorn, P., Yingkajorn, M., Kongreung, J., Nishibuchi, M. and Vuddhakul, V. 2013. The trend of Vibrio parahaemolyticus infections in Southern Thailand from 2006 to 2010. Trop. Med. Health 41, 151–6. Tsai, S.E., Jong, K.J., Tey, Y.H., Yu, W.T., Chiou, C.S., Lee, Y.S. and Wong, H.C. 2013. Molecular characterization of clinical and environmental Vibrio parahaemolyticus isolates in Taiwan. Int. J. Food Microbiol. 165, 18–26. Tsai, C.C., Lin, L.Y., Lai, T.M. and Chou, L.C. 2021. To evaluate the effects of lactic acid bacteria fermented lemon juice from Limon and Eureka varieties of Taiwan on antipathogenic bacteria and anti-allergy. J. Food Nutr. Res. 9, 382–8. Vanitha, M., Dhanapal, K. and Reddy, G.V.S. 2015. Quality changes in fish burger from Catla (Catla Catla) during refrigerated storage. J. Food Sci. Technol. 52(3), 1766–71. Xie, Y., Liu, J., Shi, Y., Wang, B., Wang, X., Wang, W. and Cheng, L. 2021. The combination of sesamol and clofibric acid moieties leads to a novel potent hypolipidemic agent with antioxidant, anti-inflammatory and hepatoprotective activity. Bioorg. Med. Chem. Lett. 44, 128121. Xie, T., Wu, Q., Xu, X., Zhang, J. and Guo, W. 2015. Prevalence and population analysis of Vibrio parahaemolyticus in aquatic products from South China markets. FEMS Microbiol. Lett. 362, 1–7. Xie, T., Wu, Q., Zhang, J., Xu, X. and Cheng, J. 2017. Comparison of Vibrio parahaemolyticus isolates from aquatic products and clinical by antibiotic susceptibility, virulence, and molecular characterization. Food Control 71, 315–21. Xu, M., Wu, J. and Chen, L. 2019. Virulence, antimicrobial and heavy metal tolerance, and genetic diversity of Vibrio cholerae recovered from commonly consumed freshwater fish. Environ. Sci. Pollut. Res. 26, 27338–52. Yan, L., Pei, X., Zhang, X., Guan, W., Chui, H., Jia, H. and Yang, D. 2019. Occurrence of four pathogenic Vibrios in Chinese freshwater fish farms in 2016. Food Control 95, 85–9 Yano, Y., Hamano, K., Satomi, M., Tsutsui, I., Ban, M. and Aue-Umneoy, D. 2014. Prevalence and antimicrobial susceptibility of Vibrio species related to food safety isolated from shrimp cultured at inland ponds in Thailand. Food Control 38, 30–6. Yasin, G., Jasim, S.A., Mahmudiono, T., Al-Shawi, S.G., Shichiyakh, R.A., Shoukat, S., Kadhim, A.J., Iswanto, A.H., Saleh, M.M. and Fenjan, M., 2022. Investigating the effect of garlic (Allium sativum) essential oil on foodborne pathogenic microorganisms. Food Sci. Technol., 42, p.e03822. Yassin, M.T., Mostafa, A.A.F., Al-Askar, A.A. and Sayed, S.R. 2022. In vitro antimicrobial activity of Thymus vulgaris extracts against some nosocomial and food poisoning bacterial strains. Process Biochem. 115, 152–9. Yazgan, H. 2013. Effects of nanoemulsion based on sunflower oil on sensory, chemical and microbiological quality of sea bass (Dicentrarchus labrax) and sea bream (Sparu saurata) stored at chilled temperature (2 ± 2 °C). Ph.D. Thesis, Department of Fisheries and Processing Technology, Institute of Natural and Applied Science, Cukurova University, Adana, Turkey. Yen, P.T.H., Linh, N.Q. and Tram, N.D.Q. 2021. The identification and determination of toxin genes of Vibrio strains causing hemorrhagic disease on red drum (Sciaenops ocellatus) using PCR. AMB Express 11, 1–8. Yerlikaya, P., Gokoglu, N. and Uran, H. 2005. Quality changes of fish patties produced from anchovy during refrigerated storage. Eur. Food Res. Technol. 220(3–4), 287–91. Yücel, N. and Balci, Ş. 2010. Prevalence of Listeria, Aeromonas, and Vibrio species in fish used for human consumption in Turkey. J. Food Prot. 73, 380–4. Yu, Q., Niu, M., Yu, M., Liu, Y., Wang, D. and Shi, X. 2016. Prevalence and antimicrobial susceptibility of Vibrio parahaemolyticus isolated from retail shellfish in Shanghai. Food Control 60, 263–8. Zaafrane, S., Maatouk, K., Alibi, S. and Ben Mansour, H. 2022. Occurrence and antibiotic resistance of Vibrio parahaemolyticus isolated from the Tunisian coastal seawater. J. Water Health 20, 369–84. Zhou, S., Zou, H., Huang, G. and Chen, G. 2021. Preparations and antioxidant activities of sesamol and its derivatives. Bioorgan. Med. Chem. Lett. 31, 127716. | ||
| How to Cite this Article |
| Pubmed Style Morshdy AEM, Galil GMAE, Bayomi RME, Ali AA, Mohamed RE, Shosha AM, Darwish WS. Prevalence of multidrug-resistant Vibrio species in fish in a reduction trial using lemon juice and sesame oil. Open Vet. J.. 2025; 15(2): 680-689. doi:10.5455/OVJ.2025.v15.i2.16 Web Style Morshdy AEM, Galil GMAE, Bayomi RME, Ali AA, Mohamed RE, Shosha AM, Darwish WS. Prevalence of multidrug-resistant Vibrio species in fish in a reduction trial using lemon juice and sesame oil. https://www.openveterinaryjournal.com/?mno=224295 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.16 AMA (American Medical Association) Style Morshdy AEM, Galil GMAE, Bayomi RME, Ali AA, Mohamed RE, Shosha AM, Darwish WS. Prevalence of multidrug-resistant Vibrio species in fish in a reduction trial using lemon juice and sesame oil. Open Vet. J.. 2025; 15(2): 680-689. doi:10.5455/OVJ.2025.v15.i2.16 Vancouver/ICMJE Style Morshdy AEM, Galil GMAE, Bayomi RME, Ali AA, Mohamed RE, Shosha AM, Darwish WS. Prevalence of multidrug-resistant Vibrio species in fish in a reduction trial using lemon juice and sesame oil. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 680-689. doi:10.5455/OVJ.2025.v15.i2.16 Harvard Style Morshdy, A. E. M., Galil, . G. M. A. E., Bayomi, . R. M. E., Ali, . A. A., Mohamed, . R. E., Shosha, . A. M. & Darwish, . W. S. (2025) Prevalence of multidrug-resistant Vibrio species in fish in a reduction trial using lemon juice and sesame oil. Open Vet. J., 15 (2), 680-689. doi:10.5455/OVJ.2025.v15.i2.16 Turabian Style Morshdy, Alaa Eldin M.a., Ghada M. Abd El Galil, Rasha M. El Bayomi, Amer Al Ali, Rehab E. Mohamed, Amany M. Shosha, and Wageh Sobhy Darwish. 2025. Prevalence of multidrug-resistant Vibrio species in fish in a reduction trial using lemon juice and sesame oil. Open Veterinary Journal, 15 (2), 680-689. doi:10.5455/OVJ.2025.v15.i2.16 Chicago Style Morshdy, Alaa Eldin M.a., Ghada M. Abd El Galil, Rasha M. El Bayomi, Amer Al Ali, Rehab E. Mohamed, Amany M. Shosha, and Wageh Sobhy Darwish. "Prevalence of multidrug-resistant Vibrio species in fish in a reduction trial using lemon juice and sesame oil." Open Veterinary Journal 15 (2025), 680-689. doi:10.5455/OVJ.2025.v15.i2.16 MLA (The Modern Language Association) Style Morshdy, Alaa Eldin M.a., Ghada M. Abd El Galil, Rasha M. El Bayomi, Amer Al Ali, Rehab E. Mohamed, Amany M. Shosha, and Wageh Sobhy Darwish. "Prevalence of multidrug-resistant Vibrio species in fish in a reduction trial using lemon juice and sesame oil." Open Veterinary Journal 15.2 (2025), 680-689. Print. doi:10.5455/OVJ.2025.v15.i2.16 APA (American Psychological Association) Style Morshdy, A. E. M., Galil, . G. M. A. E., Bayomi, . R. M. E., Ali, . A. A., Mohamed, . R. E., Shosha, . A. M. & Darwish, . W. S. (2025) Prevalence of multidrug-resistant Vibrio species in fish in a reduction trial using lemon juice and sesame oil. Open Veterinary Journal, 15 (2), 680-689. doi:10.5455/OVJ.2025.v15.i2.16 |