| Review Article | ||
Open Vet. J.. 2025; 15(2): 556-564 Open Veterinary Journal, (2025), Vol. 15(2): 556-564 Review Article Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?Curtis Wells Dewey*Elemental Pet Vets, PLLC, Freeville, NY *Corresponding Author: Curtis Wells Dewey. Elemental Pet Vets, PLLC, Freeville, NY. Email: elementalpetvets [at] outlook.com Submitted: 15/10/2024 Accepted: 20/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
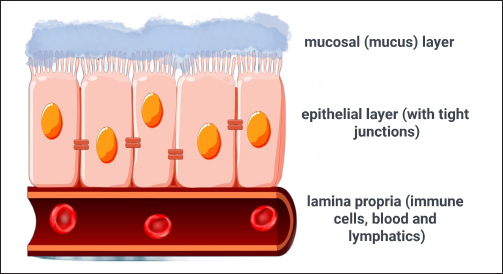
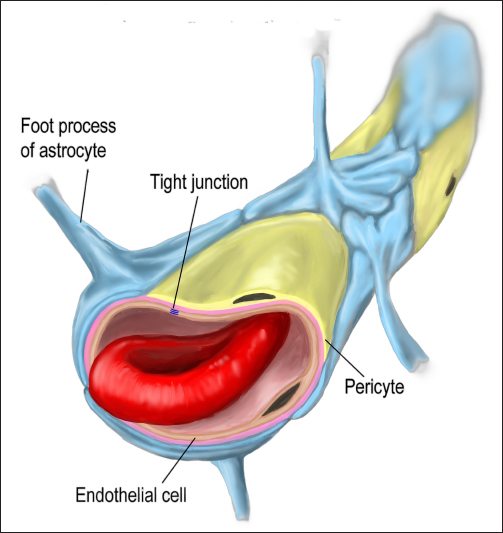
AbstractCanine cognitive dysfunction (CCD) is the dog version of human Alzheimer’s disease (AD), and it has strikingly similar pathological features to those of this neurodegenerative disorder. The gastrointestinal system is in constant communication with the brain via several conduits collectively termed the gut–brain axis. The microbial population of the gut, referred to as the microbiota, has a profound effect on the interactions that occur along this communication route. Recent evidence suggests that dysbiosis, an abnormal gastrointestinal microbial population, is linked to cognitive impairment in rodent AD models and human AD. There is also evidence from rodent AD models that correcting dysbiosis by transferring fecal material from healthy donors to the gastrointestinal tracts of cognitively impaired recipients [fecal microbiome transplantation (FMT)] reverses AD-associated brain pathology and improves cognitive function. Although limited, some clinical reports have described the improvement of cognitive function with FMT in human AD. The goals of this review article are to provide an overview of the mechanisms involved in dysbiosis- associated cognitive decline and the role of FMT in therapy for such decline. The potential role of FMT in CCD is also discussed. Keywords: Canine, Cognitive, dysfunction, Alzheimer’s, Gut–brain axis, Microbiota, Fecal, Transplantation IntroductionIn recent years, there has been considerable interest in the role that the gut microbiota plays in health and disease. The microbiota refers to the microorganisms that colonize the gastrointestinal tract, whereas the microbiome refers to these organisms, their genetic composition (metagenomics), and their biochemical products (Alessandri et al., 2019; Allam-Ndoul et al., 2020; Doifode et al., 2021). Bacterial species comprise the bulk of the gut microbiota, with smaller populations of viruses, fungi, and protozoa (Alessandri et al., 2019; Allam-Ndoul et al., 2020). Dysbiosis describes a condition in which the normal gastrointestinal microbiome is disturbed and an abnormal percentage of pathogenic bacterial species is present (Ambrosini et al., 2019; Alkhalifa et al., 2023). The pathogenesis of dysbiosis is not clearly defined, but potential contributors to this imbalance include genetics, drugs, and diet (Alessandri et al., 2019; Liu et al., 2022; Chui et al., 2024). Not surprisingly, dysbiosis has been implicated in the pathogenesis of several gastrointestinal disorders (e.g., inflammatory bowel disease, Clostridium difficile-associated enteritis) (Brandt and Aroniadis, 2013; Vendrik et al., 2020; Chaitman et al., 2021; Niina et al., 2021; Chandra et al., 2023; Matheson and Holsinger, 2023). Dysbiosis has also been linked to disorders of the central nervous system (CNS), most notably Alzheimer’s disease (AD) (Ambrosini et al., 2019; Borsom et al., 2020; Doifode et al., 2021; Angoorani et al., 2022; Alkhalifa et al., 2023; Chandra et al., 2023; Kuzniar et al., 2024; Zhao et al., 2024). CCD is the dog version of human AD and shares many pathophysiological features, such as compromised brain vasculature, deposition of toxic β-amyloid proteins around neurons and blood vessels, neuronal mitochondrial dysfunction, neuroinflammation, and oxidative and excitotoxic neuronal damage (Landsberg et al., 2012; Chapagain et al., 2018; Dewey et al., 2019). Like human AD, CCD is characterized by a slowly progressive cognitive decline that affects geriatric patients (typically 9 years or older) (Landsberg et al., 2012; Chapagain et al., 2018; Dewey et al., 2019). The prevalence of CCD in the canine population has been estimated to be between 14% and 35%, and it is thought that this range may be an underestimate (Nielson et al., 2001; Azkona et al., 2009; Salvin et al., 2010; Landsberg et al., 2012; Chapagain et al., 2018). In a recent questionnaire-based study (Kim et al., 2023), the prevalence of CCD in dogs aged >9 years was 25.9%. As with human AD, the prevalence of CCD appears to increase as dogs age, with percentages as high as 68% in dogs 15–16 years old (Nielson et al., 2001; Azkona et al., 2009; Salvin et al., 2010; Landsberg et al., 2012; Schutt et al., 2015; Chapagain et al., 2018; Kim et al., 2023). The gastrointestinal system has its own nervous system, the enteric nervous system (ENS). The gut is normally in constant communication with the CNS via nerves connecting the ENS and the brain (primarily the vagus nerve, as well as sympathetic pathways), but also via humoral routes (blood and lymphatics). This bidirectional pathway is called the gut–brain axis (GBA) (Ambrosini et al., 2019; Borsom et al., 2020; Doifode et al., 2021; Angoorani et al., 2022; Lai et al., 2022; Kuzniar et al., 2024; Zhao et al., 2024). There are two main anatomical barriers on either side of the GBA: the intestinal barrier (IB) and the blood–brain barrier (BBB); these barriers preclude the entry of potentially damaging molecules and/or organisms across the intestinal epithelium and the brain vasculature, respectively (Zenaro et al., 2017; Cai et al., 2018; Allam-Ndoul et al., 2020; Di Tommaso et al., 2021; Kurz et al., 2021; Alkhalifa et al., 2023). In health, the microbiota produces multiple chemical mediators that both influence communication across the GBA and protect the integrity of the IB and BBB (Doifode et al., 2021; Angoorani et al., 2022; Liu et al., 2022; Chandra et al., 2023). In dysbiosis, the IB is disrupted, allowing pro-inflammatory pathogens and mediators to breach the intestinal barrier and affect the brain via the GBA; these pro-inflammatory mediators then damage the BBB, leading to direct and indirect neuroinflammation. Neuroinflammation resulting from dysbiosis and the brain’s response to it (e.g., deposition of toxic beta-amyloid proteins) are thought to be pivotal factors in the pathogenesis of AD (Ambrosini et al., 2019; Borsom et al., 2020; Allam-Ndoul et al., 2020; Doifode et al., 2021; Alkhalifa et al., 2023; Chandra et al., 2023; Jin et al., 2023). Fecal microbiome transplantation (FMT) is the transfer of fecal material from a healthy donor (with a normal microbiome) to a patient with a dysbiosis-associated disorder (Brandt and Aroniadis, 2013; Chaitman et al., 2021; Kielbik and Witkowska-Pilaszewicz, 2024). FMT is performed by transferring fresh or freeze-dried donor fecal material to the recipient via oral capsules, endoscopy, or enema (Brandt and Aroniadis, 2013; Chaitman et al., 2021). FMT has been shown to be a safe and successful treatment option for humans and dogs with chronic gastrointestinal conditions (Vendrik et al., 2020; Niina et al., 2021; Alves et al., 2023; Lee et al., 2024; Levine and Hoffman, 2023; Toresson et al., 2023; Rojas et al., 2024). FMT has also been shown to improve cognition and reverse brain changes in rodent AD models (Sun et al., 2019; Kim et al., 2020; Elangovan et al., 2023; Matheson and Holsinger, 2023). In addition, there are several clinical reports of FMT improving cognitive function in human AD patients (Hazan, 2020; Park et al., 2021; Park et al., 2022; Kim et al., 2024). The intestinal barrier, the blood–brain barrier, and the GBAThe IB has several physical and chemical components designed to prohibit the passage of potentially damaging chemicals and/or organisms from the gut lumen across the intestinal epithelium and potentially into the bloodstream and/or lymphatics (Fig. 1); the layers of this barrier include the mucosal (mucus) layer, the epithelial layer, and the lamina propria (containing immune cells) (Allam-Ndoul et al., 2020; Doifode et al., 2021; Di Tommaso et al., 2021; Liu et al., 2022). The mucus or mucosal layer is composed of water, glycoproteins (mucin), and several antimicrobial proteins such as immunoglobulin (from plasma cells) and α-defensin (from Paneth cells) (Allam-Ndoul et al., 2020; Di Tommaso et al., 2021). Cells of the epithelial layer are linked together by several proteins, collectively called junctional complexes; tight junctions (TJs) are the main type of junctional complex connecting epithelial cells. TJs preclude the entry of pathogens or pathogenic material through intercellular spaces (Doifode et al., 2021; Di Tommaso et al., 2021; Liu et al., 2022). The final layer or the IB is the lamina propria, a loose connective tissue layer containing immune cells (e.g., plasma cells, macrophages); once a harmful substance or organism breaches this last layer, such materials can then reach the brain via several routes (Allam-Ndoul et al., 2020; Di Tommaso et al., 2021). Similar to the IB, the BBB is designed to prevent entry of pathogens and toxins into the brain from the exterior environment, in this case the bloodstream (Fig. 2). The three main components of the BBB are as follows: the TJs of capillary endothelial cells, astrocytic foot processes, and perivascular cells called pericytes (Zenaro et al., 2017; Cai et al., 2018; Brown et al., 2019; Allam-Ndoul et al., 2020; Kurz et al., 2021; Alkhalifa et al., 2023). Endothelial cell TJs form the main component of the BBB, with astrocytic foot processes contributing to this barrier by covering most of the capillary endothelial surface area. Pericytes and microglia contribute to this secondary barrier and provide additional protection. Pericytes are multifunctional cells located along the walls of brain capillaries that can regulate regional blood flow, affect local astrocyte function, maintain the integrity of endothelial TJs, and actively transport pathogenic materials out of the brain parenchyma (e.g., β-amyloid) (Zenaro et al., 2017; Cai et al., 2018; Brown et al., 2019; Allam-Ndoul et al., 2020; Kurz et al., 2021; Alkhalifa et al., 2023). Microglia are the CNS version of macrophages. In health, the main functions of microglia are surveillance and protection; these cells can recognize and phagocytize foreign material that breaches the BBB (Zenaro et al., 2017; Doifode et al., 2021). There are two main types of activated microglia: M1 and M2. Normally, the M2 type is predominant; these microglia are neuroprotective. M2 microglia release anti-inflammatory cytokines and promote the release of neurotrophic factors (Tang and Le, 2016; Soriano et al., 2022). In chronic inflammatory states, such as AD, the microglial population shifts to the M1 type; M1 microglia produce pro-inflammatory cytokines (e.g., IL-1β, IL-6, TNFα), which can damage the BBB and brain parenchyma (Tang and Le, 2016; Soriano et al., 2022).
Fig. 1. Schematic illustration of the IB. The first barrier is the mucosal (mucus) layer composed of water, glycoproteins, and antimicrobial proteins. The second or epithelial layer contains TJs between cells to prohibit the passage of potentially harmful substances and/or pathogens. The final layer is the lamina propria which contains immune cells such as plasma cells and macrophages.
Fig. 2. Illustration depicting the major components of the BBB. These components include endothelial cell TJs, astrocyte foot processes, and multifunctional cells called pericytes. The GBA describes a bidirectional communication pathway between the gut and brain (Fig. 3). This communication pathway has several contributors, including neural (primarily via the vagus nerve, but also sympathetic pathways), humoral (the bloodstream and lymphatics) and the hypothalamic–pituitary–adrenal axis (HPA) (Ambrosini et al., 2019; Borsom et al., 2020; Doifode et al., 2021; Angoorani et al., 2022; Lai et al., 2022; Rusch et al., 2023; Kuzniar et al., 2024; Zhao et al., 2024). Various chemical mediators from the gut microbiota normally traverse the GBA, influencing brain function; similarly, the brain can affect gastrointestinal function as well as the gut microbiota via these pathways (Ambrosini et al., 2019; Borsom et al., 2020; Doifode et al., 2021; Angoorani et al., 2022; Lai et al., 2022; Rusch et al., 2023; Kuzniar et al., 2024; Zhao et al., 2024). When the IB and BBB are intact, the passage of substances along the GBA is tightly regulated. The microbiota, dysbiosis, and cognitive declineThe healthy gastrointestinal microbiota contains a wide diversity of different bacterial phyla, with most of these organisms providing health benefits to the host; when this balance shifts in favor of more pathogenic bacteria, the state of the microbiome is referred to as dysbiosis (Ambrosini et al., 2019; Alkhalifa et al., 2023). Gut bacteria normally communicate with the brain via the GBA by producing an array of chemical mediators. These mediators include neurotransmitters (e.g., acetylcholine, gamma-aminobutyric acid, serotonin, and dopamine), hormones (e.g., glucocorticoids), histamine, tryptophan derivatives (e.g., indoles), and short-chain fatty acids (SCFAs). In the normal state, these chemical mediators act together to maintain brain health (Ambrosini et al., 2019; Borsom et al., 2020; Doifode et al., 2021; Angoorani et al., 2022; Chandra et al., 2023). In particular, SCFAs exhibit multiple beneficial neuroprotective effects. SFCAs are produced almost exclusively from gut bacteria fermenting indigestible dietary fibers; these fatty acids are less than six carbon atoms in length and include acetic acid (acetate), butyric acid (butyrate), and propionic acid (propionate) (Doifode et al., 2021; Lai et al., 2022; Liu et al., 2022; Chandra et al., 2023; Seo and Holtzman, 2023). SCFAs play a pivotal role in maintaining the integrity of both the IB and the BBB (via various mechanisms, including regulating TJ proteins of both the IB and BBB, stimulating mucus production in the gut), enhance the expression of brain-derived neurotrophic factor, modulate the development and function of brain astrocytes and microglia, inhibit the production of pro-inflammatory cytokines, and inhibit the production/stimulate elimination of toxic Aβ in the brain (Sun et al., 2019; Doifode et al., 2021; Lai et al., 2022; Liu et al., 2022; Chandra et al., 2023; Seo and Holtzman, 2023).
Fig. 3. Schematic illustration of the GBA. This communication system is a bidirectional circuit composed of both humoral (blood and lymphatic vessels) and neural (primarily vagus nerve, with some contribution from sympathetic pathways) components. A third component (not pictured) is the HPA. Dysbiosis is linked to cognitive decline in both rodent AD models and human AD; in addition, the development of dysbiosis precedes cognitive decline in both scenarios (Ambrosini et al., 2019; Jung et al., 2022; Chen et al., 2023; Ferreiro et al., 2023; Jemimah et al., 2023; Matheson and Holsinger, 2023; Zhao et al., 2024). In human AD, a shift to a less diverse, more pathogenic population of bacteria phyla has been demonstrated prior to the onset of mild cognitive impairment (Chen et al., 2023; Ferreiro et al., 2023; Jemimah et al., 2023). In short, the state of the gut microbiota can be predictive of the likelihood of AD development (Jung et al., 2022; Chen et al., 2023; Zhao et al., 2024). Several bacterial phyla have been associated with cognitive decline in human AD; conversely, a number of bacterial phyla have been associated with cognitive health (Cammann et al., 2023; Chandra et al., 2023; Elangovan et al., 2023; Zhao et al., 2024). In general, pathogenic bacteria are pro-inflammatory, whereas the “healthy” bacteria are anti-inflammatory. Some “healthy” bacteria that have been shown to be decreased in human AD patients’ microbiome includes Firmicutes, Bifidobacteria, and Lactobacillus; more pathogenic bacteria that tend to be more prominent in patients with AD include Actinobacteria and Proteobacteria (Angoorani et al., 2022; Chandra et al., 2023; Chen et al., 2023; Elangovan et al., 2023; Ferreiro et al., 2023; Seo and Holtzman, 2023). In general, the “healthy” bacteria are the main source of SCFAs, whereas the pathogenic bacteria release inflammatory mediators, such as lipopolysaccharide (LPS-endotoxin) and bacterial Aβ; in dysbiosis, these inflammatory mediators disrupt the IB, promote the production of pro-inflammatory cytokines, and subsequently disrupt the BBB via inducing neuroinflammation via the GBA (Doifode et al., 2021; Cammann et al., 2023; Chandra et al., 2023). There is considerable evidence that Aβ is produced in the brain in response to pathogenic microbes and/or their products; Aβ is known to be an antimicrobial protein (Chandra et al., 2023; Giridharan et al., 2023; Jin et al., 2023). Although Aβ plays a major role in AD pathophysiology as a neurotoxic protein, its overproduction may be a secondary response to the neuroinflammation induced by dysbiosis (Ambrosini et al., 2019; Vendrik et al., 2020; Doifode et al., 2021; Jemimah et al., 2023; Jin et al., 2023). The microbiome/microbiota of dogs is very similar to that of humans, presumably because dogs and humans have shared the same environment and diet over thousands of years (Coelho et al., 2018; Ambrosini et al., 2019; Kubinyi et al., 2020). CCD is a naturally occurring animal model of human AD (Landsberg et al., 2012; Chapagain et al., 2018; Dewey et al., 2019). Although there are limited data in dogs linking dysbiosis to CCD (ref), there is evidence that the canine microbiota affects memory in dogs; higher microbiota levels of Bifidobacteria and lower levels of Actinobacteria have been associated with higher memory test performance in dogs (Kubinyi et al., 2020; Ma et al., 2024). FMT as a potential therapy for CCD/ADFMT entails the delivery of fecal material from a healthy individual with a normal microbiome to the gastrointestinal tract of a patient with a dysbiosis- associated disorder (e.g., AD) to shift the patient’s microbiota toward a healthier population (Brandt and Aroniadis, 2013; Chaitman et al., 2021; Doifode et al., 2021; Matheson and Holsinger, 2023). Most of the information regarding the efficacy of FMT for AD is from rodent models of AD (Elangovan et al., 2023; Levine and Hoffman, 2023). Based on these data, FMT is successful in improving cognitive function and reversing pathologic changes in the brain (Sun et al., 2019; Kim et al., 2020; Elangovan et al., 2023). Specifically, FMT resulted in decreased deposition and accelerated elimination of brain Aβ deposits, decreased phosphorylation of tau protein and formation of neurofibrillary tangles in the brain, restoration of a more normal microbiome, elevated levels of SCFA in the gut lumen, and decreased intestinal and systemic inflammation (Sun et al., 2019; Kim et al., 2020; Elangovan et al., 2023). Conversely, it has been shown in rodents that transferring fecal contents from a cognitively impaired donor to a non-impaired recipient can induce cognitive deficits in the recipient (Soriano et al., 2022; Grabrucker et al., 2023). Albeit sparse, some clinical literature supports the role of FMT in human AD. At the time of submission of this manuscript, there were two single case reports, a case series of five patients, and a small (10 FMT patients, 10 controls) prospective controlled study evaluating FMT’s effect on cognitive impairment in patients with AD being treated for Clostridium difficile-associated enteritis; cognitive scores improved in all cases, and this improvement was significant in the one controlled study (Hazan, 2020; Park et al., 2021; Park et al., 2022; Kim et al., 2024). Except for one case in which it was not investigated, the fecal microbiota population was altered by FMT treatment (Park et al., 2021; Park et al., 2022; Kim et al., 2024). FMT has been successfully used in dogs to treat several gastrointestinal disorders, including chronic vomiting, Clostridium perfringens-associated diarrhea, acute hemorrhagic diarrhea, Parvovirus-associated diarrhea, and large bowel diarrhea (Brandt and Aroniadis, 2013; Gal et al., 2021; Niina et al., 2021; Alves et al., 2023; Torreson et al., 2023; Kielbik and Witkowska- Pilaszewicz, 2024; Rojas et al., 2024). Increased levels of SCFA-producing bacteria have been documented in the microbiota of dogs after FMT for gastrointestinal disease (Rojas et al., 2024). To the author’s knowledge, there are no reports of FMT for the treatment of CCD. Considering the available data from rodent AD models and limited human AD reports, FMT for treating cognitive decline may be a treatment avenue worth investigating in dogs. ConclusionManipulating the microbiome with FMT may represent another treatment option for CCD, the dog version of human AD. Abnormal microbiome populations, termed dysbiosis, have been documented in rodent AD models and human AD patients. Dysbiosis is believed to precede cognitive decline in AD and may play a causative role. Dysbiosis leads to loss of IB integrity, regional and systemic inflammation, subsequent breakdown of the BBB, and ultimately neuroinflammation and cognitive impairment. By repopulating a cognitively impaired recipient’s microbiome with feces from a normal donor, it may be possible to re-establish a healthy microbiota, thereby reversing some of the damage to the IB, the BBB, and the brain. Because of the similarities between human AD and CCD, as well as their similar microbiome compositions, investigation into FMT for CCD may provide insights and potential benefits for dogs and people with cognitive decline. AcknowledgmentsThere are no acknowledgments for this article. FundingThis article received no funding. Conflict of interestThere are no conflicts of interest with this article. Authors’ contributionsDr. Dewey is the sole author. Data availabilityThere are no data available, as this is a review article. ReferencesAlessandri, G., Milani, C., Mancabelli, L., Mangifesta, M., Lugli, G.A., Viappiani, A., Duranti, S., Turroni, F., Ossiprandi, M.C., van Sinderen, D. and Ventura, M. 2019. Metagenomic dissection of the canine gut microbiota: insights into taxonomic, metabolic and nutritional features. Env. Microbiol. 21, 1331–1343. Allam-Ndoul, B., Castonguay-Paradis, S. and Veilleux, A. 2020. Gut microbiota and intestinal trans-epithelial permeability. Int. J. Mol. Sci. 21(17), 6402; doi:10.3390/ijms21176402. Alkhalifa, A.E., Al-Ghraiybah, N.F., Odum, J., Shunnarah, J.G., Austin, N. and Kaddoumi, A. 2023. Blood-brain barrier breakdown in Alzheimer’s disease: mechanisms and targeted strategies. Int. J. Mol. Sci. 24(22), 16288; doi:10.3390/ijms242216288. Alves, J.C., Santos, A., Jorge, P. and Pitaes, A. 2023. Faecal microbiome transplantation improves clinical signs of chronic idiopathic large bowel diarrhoea in working dogs. Vet. Rec. 193(10), e3052; doi:10.1002/vetr.3052. Ambrosini, Y.M., Borcherding, D., Kanthasamy, A., Kim, H.J., Wilette, A.A., Jergens, A., Allenspach, K. and Mochel, J.P. 2019. The gut-brain axis in neurodegenerative diseases and relevance of the canine model: a review. Front. Aging. Neurosci. 11, 130; doi:10.3389/fnagi.2019.00130 Angoorani, P., Ejtahed, H-S., Siadat, S.D., Sharifi, F. and Larijani, B. 2022. Is there any link between cognitive impairment and gut microbiota? A systematic review. Geront. 68. 1201–1213. Azkona, G., Garcia-Belenguer, S.G., Chacon, G., Fosado, B., Leon, M. and Palacio, J. 2009. Prevalence and risk factors of behavioural changes associated with age-related cognitive impairment in geriatric dogs. J. Small. Anim. Pract. 50. 87–91. Borsom, E.M., Lee, K. and Cope, E.K. 2020. Do the bugs in your gut eat your memories? Relationship between gut microbiota and Alzheimer’s disease. Brain. Sci. 10(11), 814; doi:10.3390/brainsci10110814. Brandt, L.J. and Aroniadis, O.C. 2013. An overview of fecal microbiota transplantation: techniques, indications, and outcomes. Gastr. Endosc. 78(2), 240–249; doi:10.1016/j.gie.2013.03.1329. Brown, L.S., Foster, C.G., Courtney, J-M., King, N.E., Howells, D.W. and Sutherland, B.A. 2019. Pericytes and neurovascular function in the healthy and diseased brain. Front. Cell. Neurosci. 13, 282; doi:10.3389/fncel.2019.00282. Cai, Z., Qiao, P-F., Wan, C-Q., Cai, M., Zhou, N-K. and Li, Q. 2018. Role of the blood-brain barrier in Alzheimer’s disease. J. Alz. Dis. 63. 1223–1234. Cammann, D., Lu, Y., Cummings, M.J., Zhang, M.L., Cue, J.M., Do, J., Ebersole, J., Chen, X., Oh, E.C., Cummings, J.L. and Chen, J. 2023. Genetic correlations between Alzheimer’s disease and gut microbiome genera. Sci. Reports 13, 5258; doi:10.1038/s41598-023-31730-5. Chaitman, J. and Gaschen, F. 2021. Fecal microbiota transplantation in dogs. Vet. Clin. Small. Anim. 51. 219–233. Chandra, S., Sisodia, S.S. and Vassar, R.J. 2023. The gut microbiome in Alzheimer’s disease: what we know and what remains to be explored. Molec. Neurodeg. 18, 9; doi:10.1186/s13024-023-00595-7. Chapagain D, Range F, Huber L, Viranyi Z. 2018. Cognitive aging in dogs. Gerontology 64(2): 165–171. doi:10.1159/000481621. Chen, G., Zhou, X., Zhu, Y., Shi, W. and Kong, L. 2023. Gut microbiome characteristics in subjective cognitive decline, mild cognitive impairment and Alzheimer’s disease: a systematic review and meta-analysis. Eur. J. Neurol. 30, 3568–3580. Chui, Z.S.W., Chan, L.M.L., Zhang, E.W.H., Liang, S., Choi, E.P.H., Lok, K.Y.L., Tun, H.M. and Kwok, J.Y.Y. 2024. Effects of microbiome-based interventions on neurodegenerative diseases: a systematic review and meta-analysis. Sci. Reports. 14(1), 9558; doi:10.1038/s41598-024-59250-w. Coelho, L.P., Kultima, J.R., Costea, P.I., Fournier, C., Pan, Y., Czarnecki-Maulden, G., Hayward, M.R., Forslund, S.K., Schmidt, T.S.B., Descombes, P., Jackson, J.R., Li, Q. and Bork, P. 2018. Similarity of the dog and human gut microbiomes in gene content and response to diet. Microbiome 6, 72; doi:10.1186/s40168-018-0450-3. Dewey CW, Davies ES, Xie H, Wakshlag JJ. 2019. Canine cognitive dysfunction: pathophysiology, diagnosis, and treatment. Vet Clin Small Anim 49:477–499. doi:10.1016/j.cvsm.2019.01.013. Di Tomasso, N., Gasbarrini, A. and Ponziani, F.R. 2021. Intestinal barrier in human health and disease. Int. J. Environ. Res. Public Health. 18, 12836; doi:10.3390/ijerph182312836. Doifode, T., Giridharan, V.V., Generoso, J.S., Bhatti, G., Collodel, A., Schulz, P.E., Forlenza, O.V. and Barichello, T. 2021. The impact of the microbiota-gut-brain axis on Alzheimer’s disease pathophysiology. Pharm. Res. 164, 105314; doi:10.1016/j.phrs.2020.105314. Elangovan, S., Barody, T.J. and Holsinger, R.M.D. 2023. Fecal microbiota transplantation reduces pathology and improves cognition in a mouse model of Alzheimer’s disease. Cells 12, 119; doi:10.3390/cells12010119. Ferreiro, A.L., Choi, J., Ryou, J., Newcomber, E.P., Thompson, R., Bollinger, R.M., Hall-Moore, C., Ndao, I.M., Sax, L., Benzinger, T.L.S., Stark, S.L., Holtzman, D.M., Fagan, A.M., Schindler, S.E., Cruchaga, C., Butt, O.H., Morris, J.C., Tarr, P.I., Ances, B.M. and Dantas, G. 2023. Gut microbiome composition may be an indicator of preclinical Alzheimer’s disease. Sci. Transl. Med. 15(700), eabo2984; doi: 10.1126/scitranslmed.abo2984. Gal, A., Barko, P.C., Biggs, P.J., Gedye, K.R., Midwinter, A.C., Williams, D.A., Burchell, R.K. and Pazzi, P. 2021. One dog’s waste is another dog’s wealth: a pilot study of fecal microbiota transplantation in dogs with acute hemorrhagic diarrhea syndrome. PLos One 16, e0250344; doi:10.1371/journal.pone.0250344. Giridharan, V.V., Catumbela, C.S.G., Catalao, C.H.R., Lee, J., Ganesh, B.P., Petronilho, F., Dal-Pissol, F., Morales, R. and Barichello, T. 2023. Sepsis exacerbates Alzheimer’s disease pathophysiology, modulates the gut microbiome, increases neuroinflammation and amyloid burden. Mol. Psych. 28, 4463–4473. Grabrucker, S., Marizzono, M., Silajdzic, E., Lopizzo, N., Mombelli, E., Nicolas, S., Dohm-Hansen, S., Scassellati, C., Moretti, D.V., Rosa, M., Hoffmann, K., Cryan, J.K., O’Leary, O.F., English, J.A., Lavelle, A., O’Neill, C., Thuret, S., Cattaneo, A. and Nolan, Y.M. 2023. Microbiota from Alzheimer’s patients induce deficits in cognition and hippocampal neurogenesis. Brain. 146, 4916–4934. Hazan, S. 2020. Rapid improvement in Alzheimer’s disease symptoms following fecal microbiota transplantation: a case report. J. Int. Med. Res. 48, 1–6. Jemimah, S., Chabib, C.M.M.C., Hadjileontiadis, L. and Alshehhi, A. 2023. Gut microbiome dysbiosis in Alzheimer’s disease and mild cognitive impairment: a systematic review and meta-analysis. PLos One 18(5), e0285346; doi:10.1371/journal.pone.0285346. Jin, J., Xu, Z., Zhang, L., Zhang, C., Zhao, X., Mao, Y., Zhang, H., Liang, X., Wu, J, Yang, Y. and Zhang, J. 2023. Gut derived β-amyloid: likely a centerpiece of the gut-brain axis contributing to Alzheimer’s pathogenesis. Gut. Microbes. 15, 2167172; doi:10.1080/19490976.2023.2167172. Jung, J.H., Kin, G., Byun, M.S., Lee, J.H., Yi, D., Park, H. and Lee, D.Y. 2022. Gut microbiome alterations in preclinical Alzheimer’s disease. PLos One 17(11), e0278276; doi:10.1371/journal.pone.0278276. Kielbik, P. and Witkowska-Pilaszewicz, O. 2024. The relationship between canine behavioral disorders and gut microbiome and future therapeutic perspectives. Animals 14(14), 2048; doi:10.3390/ani14142048. Kim, S-S., Choi, D., Yu, H., Ju, J., Hong, S., Shin, J., Won, S., Gwag, B.J., Youn, H-Y. and Lee, J.H. 2023. Prevalence and risk factors of canine cognitive dysfunction syndrome in South Korea. App. Anim. Behav. Sci. 268, 106066; doi:10.1016/j.applanim.2023.106066. Kim, J-S., Park, H., Lee, J-H, Shin, J., Cha, B., Kwon, K.S., Shin, Y.W., Kim, Y., Kim, Y.J., Bae, J.S., Lee, J-H., Choi, S-J., Kim, T.J., Ko, S-B. and Park, S-H. 2024. Effect of altered gene expression in lipid metabolism on cognitive improvement in patients with Alzheimer’s dementia following fecal microbiota transplantation: a preliminary study. Ther. Adv. Neurol. Disord. 17, 1–14. Kim, M-S., Kim, Y., Choi, H., Kim, W., Park, S., Lee, D., Kim, D.K., Kim, H.J., Choi, H., Hyun, D-W., Lee, J-Y., Choi, E.Y., Lee, D-S, Bae, J-W. and Mook-Jung, I. 2020. Transfer of a healthy microbiota reduces amyloid and tau pathology in an Alzheimer’s disease animal model. Gut 69, 283–294. Kubinyi, E., Bel Rhali, S., Sandor, S., Szabo, A. and Felfoldi, T. 2020. Gut microbiome composition is associated with age and memory performance in pet dogs. Animals 10, 1488; doi:10.3390/ani10091488. Kurz, C., Walker, L., Rauchmann, B-S. and Perneczky, R. 2022. Dysfunction of the blood-brain barrier in Alzheimer’s disease: evidence from human studies. Neuropathol. Appl. Neurobiol. 48(3), e12782; doi:10.1111/nan.12782. Kuzniar, J., Kozubek, P., Czaja, M. and Leszek, J. 2024. Correlation between Alzheimer’s disease and gastrointestinal tract disorders. Nutrients 16, 2366; doi:10.3390/nu16142366. Lai, Y. 2022. Toward elucidating the human gut microbiota-brain axis: molecules, biochemistry, and implications for health and diseases. Biochemistry 61, 2806–2821. Landsberg GM, Nichol J, Araujo JA. 2012. Cognitive dysfunction syndrome: a disease of canine and feline brain aging. Vet Clin Small Anim 42:749–768. Lee, M.A., Questa, M., Wanakumjorn, P., Koi, A., McLaughlin, B., Weimer, B.C., Buono, A., Suchodolski, J.S. and Marsilio, S. 2024. Safety profile and effects on the peripheral immune response of fecal microbiota transplantation in clinically healthy dogs. J. Vet. Intern. Med. 38, 1425–1436. Levine, B.H. and Hoffman, J.M. 2023. Gut microbiome transplants and their health impacts across species. Microorganisms 11, 1488; doi:10.3390/microorganisms11061488. Liu, L., Shang, L., Jin, D., Wu, X. and Long, B. 2022. General anesthesia bullies the gut: a toxic relationship with dysbiosis and cognitive dysfunction. Psychopharmacology 239, 709–728. Ma, X., Lazarowski, L., Zhang, Y., Krichbaum, S., Smith, J.G., Zheng, J., Cao, W., Haney, P.S., Wilborn, R.R., Price, S.B., Singletary, M., Waggoner, P. and Wang, X. 2024. Associations between memory performance and Bifidobacterium pseudolongum abundance in the canine gut microbiome. iScience 27, 109611; doi:10.1016/j.isci.2024.109611. Matheson, J-A.T. and Holsinger, R.M.D. 2023. The role of fecal microbiota transplantation in the treatment of neurodegenerative diseases: a review. Int. J. Mol. Sci. 24(2), 1001; doi:10.3390/ijms24021001. Nielson, J.C., Hart, B.L., Cliff, K.D. and Ruehl, W.W. 2001. Prevalence of behavioral changes associated with age-related cognitive impairment in dogs. J. Am. Vet. Med. Assoc. 218, 1787–1791. Niina, A., Kibe, R., Suzuki, R., Yuchi, Y., Teshima, T., Matsumoto, H., Kataoka, Y. and Koyama, H. 2021. Fecal microbiota transplantation as a new treatment for canine inflammatory bowel disease. Biosci. Microb. Food. Health 40, 98–104. Park, S-H., Lee, J.H., Shin, J., Kim, J-S., Cha, B., Lee, S., Kwon, K.S., Shin, Y.W. and Choi, S.H. 2021. Cognitive function improvement after fecal microbiota transplantation in Alzheimer’s dementia patient: a case report. Curr. Opin. Med. Res. Opin. 37, 1739–1744. Park, S-H., Lee, J-H., Kim, J-S, Kim, T.J., Shin, J., Im, J.H., Cha, B., Lee, S., Kwon, K.S., Shin, Y.W., Ko, S-B. and Choi, S.H. 2022. Fecal microbiota transplantation can improve cognition in patients with cognitive decline and Clostridioides difficile infection. Aging 14, 6449–6446. Rojas, C.A., Entrolezo, Z., Jarett, J.K., Jospin, G., Martin, A. and Ganz, H. 2024. Microbiome responses to oral fecal microbiota transplantation in a cohort of domestic dogs. Vet. Sci. 11(1), 42; doi:10.3390/vetsci11010042. Rusch, J.A., Layden, B.T. and Dugas, L.R. 2023. Signalling cognition: the gut microbiota and hypothalamic-pituitary-adrenal axis. Front. Endocr. 14, 1130689; doi: 10.3389/fendo.2023.1130689. Salvin, H.E., McGreevy, P.D., Sachdev, P.S., Perminder, S. and Valenzuela, M.J. 2010. Under diagnosis of canine cognitive dysfunction: a cross-sectional survey of older companion dogs. Vet. J. 184, 277–281. Schutt, T., Toft, N. and Berendt, M. 2015. Cognitive dysfunction, progression of age-related behavioral changes, biomarkers, and survival in dogs more than 8 years old. J. Vet. Intern. Med. 29, 1569–1577. Seo, S-O. and Holtzman, D.M. 2024. Current understanding of the Alzheimer’s disease-associated microbiome and therapeutic strategies. Exp. Molec. Med. 56, 86–94. Soriano, S., Curry, K., Wang, Q., Chow, E., Treangen, T.J. and Villapol, S. 2022. Fecal microbiota transplantation derived from Alzheimer’s disease mice worsens brain trauma outcomes in wild-type controls. Int. J. Mol. Sci. 23(9), 4476; doi:10.3390/ijms23094476. Sun, J., Xu, J., Ling, Y., Wang, G., Gong, T., Yang, C., Ye, S., Ye, K., Wei, D., Song, Z., Chen, D. and Liu, J. 2019. Fecal microbiota transplantation alleviated Alzheimer’s disease-like pathogenesis in APP/PS1 transgenic mice. Transl. Psychiatr. 9, 189; doi:10.1038/s41398-019-0525-3. Tang, Y. and Le, W. 2016. Differential roles of M1 and M2 microglia in neurodegenerative diseases. Mol Neurobiol. 53:1181–1194. Torresson, L., Spillmann, T., Pilla, R., Ludvigsson, U., Hellgren, J., Olmedal, G. and Suchodolski, J.S. 2023. Clinical effects of faecal microbiota transplantation as adjunctive therapy in dogs with chronic enteropathies-a retrospective case series of 41 dogs. Vet. Sci. 10, 271; doi:10.3390/vetsci10040271. Vendrik, K.E.W., Ooijevaar, R.E., de Jong, P.R.C., Laman, J.D., van Oosten, B.W., van Hilten, J.J., Ducarmon, Q.R., Keller, J.J., Kuijper, E.J. and Contrarino, M.F. 2020. Fecal microbiota transplantation in neurologic disorders. Front. Cell. Inf. Microbiol. 10, 98; doi: 10.3389/fcimb.2020.00098. Zenaro, E., Piacentino, G. and Constantin, G. 2017. The blood-brain barrier in Alzheimer’s disease. Neurobiol. Dis. 107, 41–56. Zhao, Q., Baranova, A. and Zhang, F. 2024. Evaluating causal effects of gut microbiome on Alzheimer’s disease. J. Prev. Alz. Dis. 11(6), 1843–1848; doi:10.14283/jpad.2024.113. | ||
| How to Cite this Article |
| Pubmed Style Curtis Wells Dewey. Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?. Open Vet. J.. 2025; 15(2): 556-564. doi:10.5455/OVJ.2025.v15.i2.6 Web Style Curtis Wells Dewey. Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?. https://www.openveterinaryjournal.com/?mno=224731 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.6 AMA (American Medical Association) Style Curtis Wells Dewey. Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?. Open Vet. J.. 2025; 15(2): 556-564. doi:10.5455/OVJ.2025.v15.i2.6 Vancouver/ICMJE Style Curtis Wells Dewey. Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 556-564. doi:10.5455/OVJ.2025.v15.i2.6 Harvard Style Curtis Wells Dewey (2025) Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?. Open Vet. J., 15 (2), 556-564. doi:10.5455/OVJ.2025.v15.i2.6 Turabian Style Curtis Wells Dewey. 2025. Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?. Open Veterinary Journal, 15 (2), 556-564. doi:10.5455/OVJ.2025.v15.i2.6 Chicago Style Curtis Wells Dewey. "Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?." Open Veterinary Journal 15 (2025), 556-564. doi:10.5455/OVJ.2025.v15.i2.6 MLA (The Modern Language Association) Style Curtis Wells Dewey. "Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?." Open Veterinary Journal 15.2 (2025), 556-564. Print. doi:10.5455/OVJ.2025.v15.i2.6 APA (American Psychological Association) Style Curtis Wells Dewey (2025) Poop for thought: Can fecal microbiome transplantation improve cognitive function in aging dogs?. Open Veterinary Journal, 15 (2), 556-564. doi:10.5455/OVJ.2025.v15.i2.6 |