| Research Article | ||
Open Vet. J.. 2025; 15(2): 668-679 Open Veterinary Journal, (2025), Vol. 15(2): 668-679 Research Article Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promotersBodhi Agustono1,2,*, Maya Nurwartanti Yunita2, Widya Paramita Lokapirnasari3, Sunaryo Hadi Warsito3, Tabita Dameria Marbun4, and Sarasati Windria51Doctoral Program of Veterinary Science, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 2Faculty of Health, Medicine and Life Sciences, Universitas Airlangga, Surabaya, Indonesia 3Division of Animal Husbandry, Department of Veterinary Science, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 4Animal Science Laboratory, Kyungpook National University, Sangju, Korea 5Department of Biomedical Science, Faculty of Medicine, Universitas Padjajaran, Bandung, Indonesia *Corresponding Author: Bodhi Agustono. Doctoral Program of Veterinary Science, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. bodhiagustono [at] fkh.unair.ac.id Submitted: 16/10/2024 Accepted: 03/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
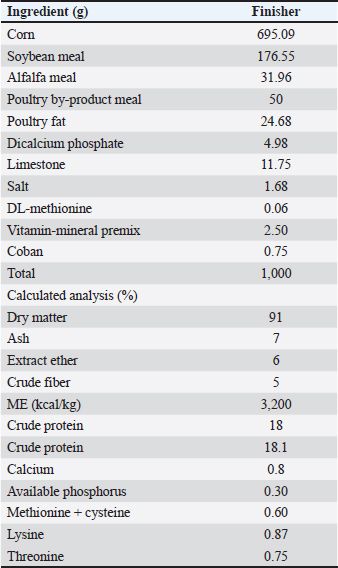
AbstractBackground: The rising global concern over antibiotic resistance has heightened scrutiny of antibiotic growth promoters (AGPs) in poultry farming, prompting a shift toward alternative feed additives to ensure sustainable and safe poultry production. This trend aligns with the increasing demand for free-range and naturally raised chicken meat in various regions, including Indonesia. In response, Indonesian breeders have turned to medium-sized male layer chickens (MLCs) as substitutes for traditional free-range chickens. This practice, coupled with the need to replace AGPs, highlights the critical importance of exploring innovative and natural solutions to enhance poultry growth and health. Aim: This study investigated the effects of probiotics as an alternative to AGPs on the growth performance, carcass traits, and immune organs of male ISA Brown layer chickens. Methods: The 180-day-old male ISA Brown layer chickens were used for the study. The intervention included six treatments. T1 basal feed, T2 2.5 g AGP/kg feed, T3 1 ml probiotic/kg feed, T4 3 ml probiotic/kg feed, T5 4 ml probiotic/kg feed, and T6 5 ml probiotic/kg feed. Probiotics used were Lactobacillus acidophilus, Bifidobacterium sp., and Lactobacillus plantarum at a concentration of 1.2 × 109 CFU/ml. The feeding trial lasted for 21 days for chickens aged 21–42 days, assessing growth performance [body weight, feed consumption, digestibility, and feed conversion ratio (FCR)], carcass traits, non-edible organs, and immune organs. Results: The findings demonstrate that probiotic supplementation significantly outperformed the AGP-treated group (T2) in enhancing growth performance, carcass weight, pectoral weight development, FCR, internal and immune organ weights, nutrient intake, and digestibility. While AGPs showed improvements over the control (T1), probiotic- supplemented groups, particularly T6, achieved superior results across all parameters, indicating that probiotics are not only a viable alternative to AGPs but also a more effective and sustainable approach for poultry production. Conclusion: The probiotics used in the study at 4 and 5 ml/kg of feed significantly enhanced the performance, immune organ development, and carcass attributes of MLCs, demonstrating their effectiveness as a viable alternative to AGPs. These findings highlight the potential of probiotics to improve poultry production sustainability by reducing reliance on antibiotics, enhancing growth and health outcomes, and promoting animal welfare through natural and efficient dietary interventions. Keywords: Probiotics, Good health, Growth performance, Antibiotic alternatives, Male layer chicken IntroductionThe global poultry industry faces increasing pressure to identify effective alternatives to antibiotic growth promoters (AGPs) due to rising concerns about antibiotic resistance and consumer demand for sustainable and natural meat production (Broom, 2021). In Indonesia, chicken meat, particularly from broilers and local free-range chickens, remains highly popular. However, the low productivity of free-range chickens has led breeders to use medium-sized male layer chicken (MLC) as substitutes, highlighting the need for innovative solutions to enhance productivity without AGPs (Rahmat et al., 2015; Ramadhanti et al., 2021). The qualitative qualities of MLC are better than those of male broilers; however, this is dependent on the raising system and slaughter age (Gerken et al., 2003). The flesh of medium-MLC is chosen by meat producers because its texture is comparable to that of free-range chicken; thus, the general public prefers medium-MLC (Gerken et al., 2003; Lichovníková et al., 2009). The excessive use of AGPs as feed additives in livestock to enhance production and efficiency poses significant risks, including the development of resistance to pathogenic bacteria in poultry, which can impact human health (Lokapirnasari et al., 2019; Agustono et al., 2022). Antimicrobial resistance (AMR) can propagate through the food chain via direct or indirect interactions among various stakeholders and the environment, which are also recognized pathways for the transmission of zoonotic diseases, including those originating from livestock products (Abreu et al., 2023). Amid the current AMR crisis, the Indonesian Government has formally banned the use of AGPs as feed additives (Marshall and Levy, 2011). This prohibition underscores the need for alternative strategies to enhance production while safeguarding animal health by preventing the accumulation of harmful residues on their surfaces (Cook, 2004). As a sustainable and health-conscious alternative, probiotics have emerged as a promising solution, offering comparable benefits in promoting growth and feed efficiency without the associated risks of antibiotic resistance (Kalia et al., 2022). Probiotics serve as a feed additive and offer an alternative to antibiotics for incorporation into animal feed. The benefits of probiotics include improved performance and production in poultry. Probiotics can improve nutrient absorption and function as an antibiotic to stop the growth of harmful bacteria in the digestive system (Krysiak et al., 2021). Moreover, probiotics can help chickens with their health and ability to reproduce. Probiotics contain microbiota such as Lactobacillus spp. and Bifidobacterium spp. that can be used to raise the beneficial bacteria population in the intestine (Lokapirnasari et al., 2017). A lactic acid bacterium known as the microbiota belongs to the group of microbes that grow and benefit livestock’s digestive systems. For more effective outcomes, probiotics can be administered by blending them into feed or drinking water (Lokapirnasari et al., 2017; Agustono et al., 2022). The administration of probiotics to poultry can improve nutritional digestion, decrease pathogen growth, modulate immune response, and increase antioxidant capacity while lowering the feed conversion ratio (FCR) (Al-Khalaifah, 2018; Krysiak et al., 2021). The symbiotic benefits of combining three or more probiotics have been studied in several studies. Probiotics have been used in a variety of doses, yet it is impossible to pinpoint the precise dosage (Abd El-Hack et al., 2020). This study aimed to assess the effects of probiotic supplementation, specifically Lactobacillus acidophilus, Lactobacillus plantarum, and Bifidobacterium spp., as alternatives to AGPs on the growth performance, organ development, nutrient intake, and digestibility in MLC during the finisher period. These probiotic strains were chosen for their proven roles in enhancing gut health, nutrient absorption, and immune function, making them highly relevant for optimizing poultry production outcomes. Materials and MethodsEthical approvalEthical approval was obtained from the Ethical Committee of Airlangga University, Indonesia (Approval Number: 518/HRECC.FODM/IX/2021) before the commencement of the experimental trial. Study period and locationThe research was conducted between August and October of 2021. The investigation was carried out within the research animal enclosures of the Animal Feed Laboratory, Faculty of Veterinary Medicine, Airlangga University. Feed analysis was also performed at this institution. Experimental designThis research used a completely randomized factorial design pattern. In total, 180 males of the ISA Brown strain were allocated into six treatment groups with five replications, and each replication consisted of six individuals for this study, so that each treatment group consisted of 30 animals. ISA Brown laying hen cages were arranged individually and then fed to the experimental group twice a day in the morning and evening. Drinking water and feed are provided ad libitum. This study employed the following treatments: (T1) 100% basal feed (BF), (T2) BF plus 2.5 g of AGP (Virginiamycin) per kg of feed, (T3) BF plus 1 ml of probiotic per kg of feed, (T4) BF plus 3 ml of probiotic per kg of feed, (T5) BF plus 4 ml of probiotic per kg of feed, and (T6) BF plus 5 ml of probiotic per kg of feed. Probiotic mixing is done by spraying it evenly on the basal feed. Rearing systemThe nutrient composition of the starting phase is shown in Table 1. Within the control group, neither AGP nor probiotics were introduced to the meal. Group T2 was fed AGP by putting 2.5 g of AGP added to diet (kg) of diet and then sprayed with the probiotic solution 1.2 × 109 CFU/ml of the probiotic strains L. acidophilus, Bifidobacterium sp., and L. plantarum. The ISA Brown stud feed of the experimental group was sprayed with the probiotic solution by the treatment dosages (T3, T4, T5, and T6) and then air-dried for 5–10 minutes of probiotics per kilogram of feed. The treatment stage was carried out in the finisher phases from 21 to 42 days old. Temperature and humidity in the research cage were 27.18°C ± 0.623°C and 64.6% ± 2.73%. Table 1. Ingredients and calculated analysis of basal diet.
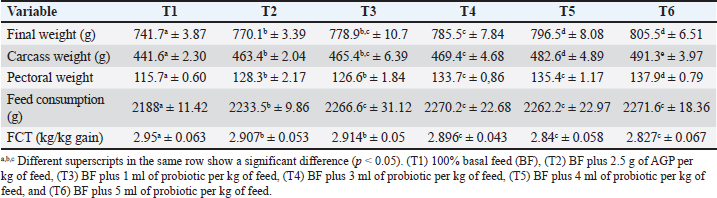
Observed parametersFCR = Feed consumption divided by body weight at 42 days of maintenance; Feed efficiency = (Body weight gain divided by feed consumption) per centimeter; Feed consumption (g) = Number of feeds administered (g) –number of unconsumed feeds. Intake of crude protein (g) = (Amount of feed given (g) × % dry matter of feed × % crude protein of feed) –(Amount × of unconsumed feed × % dry matter of unconsumed feed × % crude protein of unconsumed feed); Intake of crude fiber (g) = (Amount of feed given (g) × % dry matter of feed × % crude fiber of feed) –(Amount of unconsumed feed × % dry matter of unconsumed feed × % crude fiber of unconsumed feed). Intake of organic matter (g) = (Amount of feed given (g) × % dry matter of feed × % organic matter of feed) –(Amount of unconsumed feed × % dry matter of unconsumed feed × % organic matter of unconsumed feed). Intake of inorganic matter (g) = (Amount of feed given (g) × % dry matter of feed × % inorganic matter of feed) –(Amount of unconsumed feed × % dry matter of unconsumed feed × % inorganic matter of unconsumed feed). All MLC were slaughtered after the treatments, and their organs were extracted to determine the internal organs (liver, lungs, kidney, and heart), non-edible organs (head, leg, and wing), and immune organs (spleen, bursa fabricius, and thymus). Statistical examinationThe statistical analysis was performed using SPSS Version 26.0. for windows. Data was first tested for normality with the Kolmogorov-Smirnov test, then if the data were normally distributed continued with one- way ANOVA used to determine differences between treatment groups at a significance level of p < 0.05. If significant differences were found, Duncan’s multiple range test was applied at a significance level of 5%. ResultsGrowth performanceThe growth performance of MLC was significantly enhanced by the supplementation of probiotics as a replacement for AGPs during the finisher period (21 days old rearing) which can be seen in Table 2. The final weights of chickens across different treatment groups showed a clear trend of improvement with increasing probiotic supplementation. Specifically, the final weights were 741.7 ± 3.87 g (T1, control), 770.1 ± 3.39 g (T2, AGP), 778.9 ± 10.7 g (T3), 785.5 ± 7.84 g (T4), 796.5 ± 8.08 g (T5), and 805.5 ± 6.51 g (T6), with significant differences (p < 0.05) observed among the groups. The T6 group showed the highest final weight. Carcass weight was also positively affected by probiotic supplementation, with recorded carcass weights of 441.6 ± 2.30 g (T1), 463.4 ± 2.04 g (T2), 465.4 ± 6.39 g (T3), 469.4 ± 4.68 g (T4), 482.6 ± 4.89 g (T5), and 491.3 ± 3.97 g (T6), showing significant differences (p < 0.05) and T6 having the highest carcass weight. Table 2. Performances of MLC were enhanced by the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) as a replacement for AGPs during the finisher period (42 days old rearing).
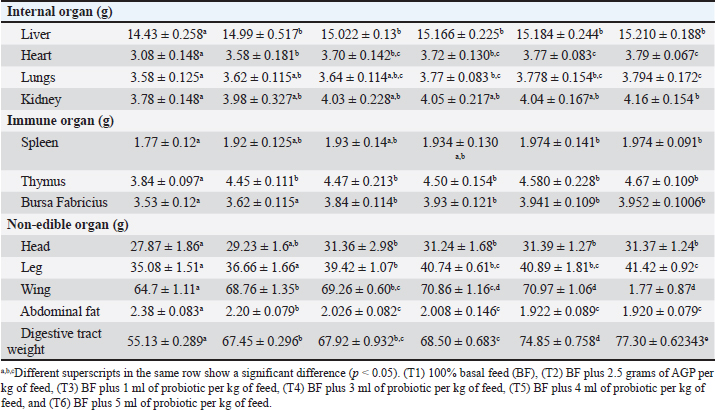
The FCR showed significant improvements in the groups supplemented with probiotics: 2.95 ± 0.063 (T1), 2.907 ± 0.053 (T2), 2.914 ± 0.05 (T3), 2.896 ± 0.043 (T4), 2.84 ± 0.058 (T5), and 2.827 ± 0.067 (T6), with significant differences (p < 0.05) and T6 having the best FCR. The study found that the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) significantly improved the growth performance, carcass weight, pectoral weight development, feed consumption, and FCR of MLC during the finisher period compared to both the basal diet and AGPs. The improvements were more pronounced with higher doses of probiotics, suggesting a dose-dependent effect. Pectoral weight (g)Pectoral weight exhibited significant improvements with probiotic supplementation as can be observed in Table 2: 115.7 ± 0.60 g (T1), 128.3 ± 2.17 g (T2), 126.6 ± 1.84 g (T3), 133.7 ± 0.86 g (T4), 135.4 ± 1.17 g (T5), and 137.9 ± 0.79 g (T6), with significant differences (p < 0.05) observed and the highest pectoral weight in T6. Feed consumption increased with probiotic supplementation: 2188 ± 11.42 g (T1), 2233.5 ± 9.86 g (T2), 2266.6 ± 31.12 g (T3), 2270.2 ± 22.68 g (T4), 2262.2 ± 22.97 g (T5), and 2271.6 ± 18.36 g (T6), with significant differences (p < 0.05), and T6 showing the highest feed consumption. Internal organsThe supplementation of probiotics significantly influenced the internal organ weights of MLC which can be seen in Table 3. The liver weights were 14.43 ± 0.258 g (T1), 14.99 ± 0.517 g (T2), 15.022 ± 0.13 g (T3), 15.166 ± 0.225 g (T4), 15.184 ± 0.244 g (T5), and 15.210 ± 0.188 g (T6), with significant differences (p < 0.05) observed and T6 having the highest liver weight. The heart weights showed a similar trend, with significant differences (p < 0.05) among the groups: 3.08 ± 0.148 g (T1), 3.58 ± 0.181 g (T2), 3.70 ± 0.142 g (T3), 3.72 ± 0.130 g (T4), 3.77 ± 0.083 g (T5), and 3.79 ± 0.067 g (T6), with T6 showing the highest heart weight. Lung weights also increased with probiotic supplementation, with weights of 3.58 ± 0.125 g (T1), 3.62 ± 0.115 g (T2), 3.64 ± 0.114 g (T3), 3.77 ± 0.083 g (T4), 3.778 ± 0.154 g (T5), and 3.794 ± 0.172 g (T6), showing significant differences (p < 0.05) and T6 having the highest lung weight. Kidney weights followed the same pattern: 3.78 ± 0.148 g (T1), 3.98 ± 0.327 g (T2), 4.03 ± 0.228 g (T3), 4.05 ± 0.217 g (T4), 4.04 ± 0.167 g (T5), and 4.16 ± 0.154 g (T6), with significant differences (p < 0.05) and T6 showing the highest kidney weight. Table 3. Internal, immune, and non-edible organs of MLC were enhanced by the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) as a replacement for AGPs during the finisher period (42 days old rearing).
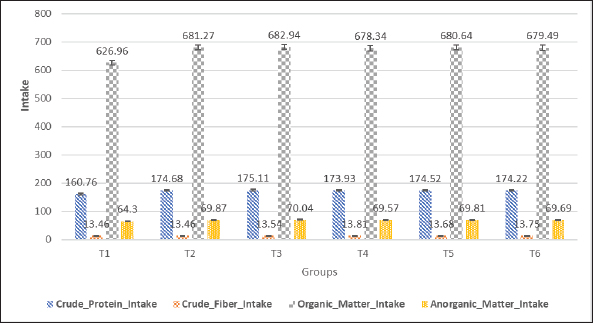
Immune organsThe weight of immune organs is also positively affected by probiotic supplementation in MLC, which can be seen in Table 3. Spleen weights were 1.77 ± 0.12 g (T1), 1.92 ± 0.125 g (T2), 1.93 ± 0.14 g (T3), 1.934 ± 0.130 g (T4), 1.974 ± 0.141 g (T5), and 1.974 ± 0.091 g (T6), with significant differences (p < 0.05) and T6 having the highest spleen weight. Thymus weights were significantly higher (p < 0.05) in the probiotic- supplemented groups: 3.84 ± 0.097 g (T1), 4.45 ± 0.111 g (T2), 4.47 ± 0.213 g (T3), 4.50 ± 0.154 g (T4), 4.580 ± 0.228 g (T5), and 4.67 ± 0.109 g (T6). The bursa of Fabricius weights also increased with supplementation: 3.53 ± 0.12 g (T1), 3.62 ± 0.115 g (T2), 3.84 ± 0.114 g (T3), 3.93 ± 0.121 g (T4), 3.941 ± 0.109 g (T5), and 3.952 ± 0.1006 g (T6), with significant differences (p < 0.05) and T6 having the highest weight. Non-edible organsNon-edible organ weights showed significant improvements with probiotic supplementation as can be observed in Table 3. The head weights were 27.87 ± 1.86 g (T1), 29.23 ± 1.6 g (T2), 31.36 ± 2.98 g (T3), 31.24 ± 1.68 g (T4), 31.39 ± 1.27 g (T5), and 31.37 ± 1.24 g (T6), with significant differences (p < 0.05) and the highest weight in T3. Leg weights were as follows: 35.08 ± 1.51 g (T1), 36.66 ± 1.66 g (T2), 39.42 ± 1.07 g (T3), 40.74 ± 0.61 g (T4), 40.89 ± 1.81 g (T5), and 41.42 ± 0.92 g (T6), showing significant differences (p < 0.05), with T6 having the highest leg weight. Wing weights followed the same pattern: 64.7 ± 1.11 g (T1), 68.76 ± 1.35 g (T2), 69.26 ± 0.60 g (T3), 70.86 ± 1.16 g (T4), 70.97 ± 1.06 g (T5), and 71.77 ± 0.87 g (T6), with significant differences (p < 0.05) and T6 showing the highest wing weight. Abdominal fat weights decreased with higher probiotic supplementation: 2.38 ± 0.083 g (T1), 2.20 ± 0.079 g (T2), 2.026 ± 0.082 g (T3), 2.008 ± 0.146 g (T4), 1.922 ± 0.089 g (T5), and 1.920 ± 0.079 g (T6), with significant differences (p < 0.05) and the lowest weight in T6. The digestive tract weights were significantly higher (p < 0.05) in the probiotic-supplemented groups: 55.13 ± 0.289 g (T1), 67.45 ± 0.296 g (T2), 67.92 ± 0.932 g (T3), 68.50 ± 0.683 g (T4), 74.85 ± 0.758 g (T5), and 77.30 ± 0.623 g (T6). In summary, the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) significantly improved the weights of internal, immune, and non-edible organs in MLC during the finisher period compared to both the basal diet and AGPs. Intake and digestibility of nutrientsNutrient intakeThe supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) as a replacement for AGPs significantly affected the nutrient intake of MLCs during the finisher period. Crude protein intake increased significantly with the addition of probiotics. The control group (T1) had the lowest intake at 160.76 ± 0.603 g, while the groups receiving probiotics showed higher intakes as follows: T2 (174.68 ± 0.659 g), T3 (173.93 ± 0.954 g), T4 (175.11 ± 0.661 g), T5 (175.32 ± 0.807 g), and T6 (175.22 ± 0.88 g). The highest crude protein intake was observed in T5, indicating that higher levels of probiotic supplementation lead to an increase in protein intake. Crude fiber intake did not show significant differences among the treatments, indicating that probiotic supplementation did not affect this variable. The values were as follows: T1 (13.46 ± 0.177 g), T2 (13.46 ± 0.174 g), T3 (13.54 ± 1.183 g), T4 (13.81 ± 0.546 g), T5 (13.68 ± 0.865 g), and T6 (13.75 ± 0.705 g). Organic matter intake increased significantly with probiotic supplementation. The control group (T1) had the lowest intake at 626.96 ± 2.371 g. The probiotic-supplemented groups showed higher intakes: T2 (678.34 ± 3.722 g), T3 (681.27 ± 2.571 g), T4 (682.94 ± 2.58 g), T5 (682.64 ± 3.151 g), and T6 (682.49 ± 3.437 g). The highest organic matter intake was observed in T4. Inorganic matter intake also increased significantly with the addition of probiotics. The control group (T1) had the lowest intake at 64.3 ± 0.243 g, while the probiotic-supplemented groups showed higher intakes: T2 (69.87 ± 0.263 g), T3 (69.57 ± 0.381 g), T4 (70.04 ± 0.264 grams), T5 (70.11 ± 0.323 g), and T6 (70.08 ± 0.352 g). The highest inorganic matter intake was observed in T5. In conclusion, the data indicate that the supplementation of probiotics significantly enhanced the nutrient intake of MLC during the finisher period. Specifically, there was a notable increase in crude protein, organic matter, and inorganic matter intake in the probiotic-supplemented groups, while crude fiber intake remained unaffected (Fig. 1).
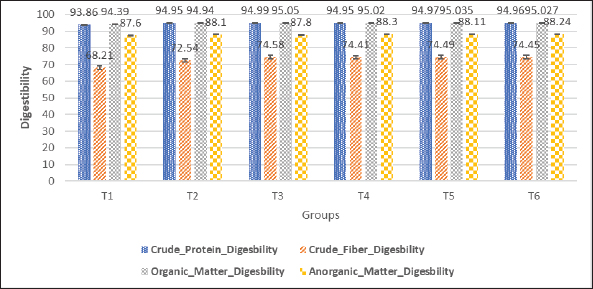
Fig. 1. Nutrient intake (g) of MLC were enhanced by the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) as a replacement for AGPs during the finisher period (42 days old rearing). Nutrient digestibilityThe nutrient digestibility of MLC was significantly enhanced by the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) as a replacement for AGPs during the finisher period as can be observed in Table 5. The control group (T1) exhibited the lowest crude protein digestibility at 93.86% ± 0.0031%. All probiotic-supplemented groups (T2–T6) demonstrated significantly higher digestibility values: T2 (94.95% ± 0.0034%), T3 (94.99% ± 0.0015%), T4 (94.95% ± 0.0022%), T5 (94.97% ± 0.0018%), and T6 (94.96% ± 0.0020%). Similarly, the crude fiber digestibility was lowest in the control group (T1) at 68.21% ± 0.0421%, while the groups receiving probiotics showed significant improvements as follows: T2 (72.54% ± 0.0328%), T3 (74.58% ± 0.022%), T4 (74.41% ± 0.0111%), T5 (74.49% ± 0.0165%), and T6 (74.45% ± 0.0138%). The control group (T1) also had the lowest organic matter digestibility at 94.39% ± 0.0021%, but supplementation with probiotics resulted in significantly higher digestibility in all groups as follows: T2 (94.94% ± 0.0025%), T3 (95.05% ± 0.0019%), T4 (95.02% ± 0.0032%), T5 (95.035% ± 0.0026%), and T6 (95.027% ± 0.0029%). Table 4. Nutrient intake (gram) of MLC were enhanced by the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) as a replacement for AGPs during the finisher period (42 days old rearing).
Table 5. Nutrient digestibility of MLC was enhanced by the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) as a replacement for AGPs during the finisher period (42 days old rearing).
Lastly, the inorganic matter digestibility was lowest in the control group (T1) at 87.6% ± 0.0081%, with probiotic-supplemented groups displaying significant improvements as follows: T2 (88.1% ± 0.0083%), T3 (87.8% ± 0.0046%), T4 (88.3% ± 0.0053%), T5 (88.11% ± 0.0049%), and T6 (88.24% ± 0.0051%). These data indicate that the supplementation of probiotics significantly enhanced the digestibility of crude protein, crude fiber, organic matter, and inorganic matter in MLC during the finisher period. These improvements were consistent across all probiotic- supplemented groups, demonstrating the efficacy of probiotics as a replacement for AGPs (Fig. 2).
Fig. 2. Nutrient digestibility of MLC was enhanced by the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) as a replacement for AGPs during the finisher period (42 days old rearing). DiscussionThis study evaluated the effects of supplementation of L. acidophilus, L. plantarum, and Bifidobacterium spp. as alternatives to AGPs on the growth performance, organ development, nutrient intake, and digestibility in MLC during the finisher period. Key findings revealed significant improvements in growth metrics, organ weights, and nutrient digestibility, highlighting probiotics’ role in enhancing intestinal health by increasing villus height and optimizing nutrient absorption, aligning with prior research on their physiological benefits (Mahdavi, 2005; Bogucka et al., 2019). In this study, supplementation with L. acidophilus, L. plantarum, and Bifidobacterium spp. resulted in a significant increase (p < 0.05) in the intake of protein, lipid, fiber, and organic matter. This finding aligns with previous research by Kabir (2009) and Yulianto et al. (2010), which stated that microbiota affects the physiological processes of the digestive system, including digestion, assimilation, and propulsion. Probiotics improve colonization resistance and exert direct inhibitory effects against pathogens, potentially reducing the incidence and duration of certain diseases (Kabir, 2009). Probiotic supplementation maintains normal gastrointestinal microbiota, establishing the first natural barrier against pathogenic microorganisms through competitive exclusion and antagonism (Fijan, 2014; Bhogoju and Nahashon, 2022). This study showed that the supplementation of L. acidophilus, L. plantarum, and Bifidobacterium spp. significantly increased the absorption of protein, lipid, fiber, and organic matter which is thought to increase the activity of digestive enzymes in the intestine. Probiotics are known to enhance gut health by balancing the intestinal microbiota, which positively impacts the absorption of various nutrients. Probiotics enhance animal performance and body weight by generating various enzymes that aid in fiber digestion and optimize nutrient absorption in livestock (Khalid et al., 2011). Lactic acid bacteria as probiotic can break down carbohydrates into simpler molecules such as glucose, supplying energy to the. Additionally, Lactobacillus enhances muscle growth and meat quality by modulating various mechanistic pathways involved in muscle development (Tsao et al., 2021; Zhang et al., 2022). For instance, supplementation with probiotics such as L. acidophilus and Bifidobacterium spp. has been reported to increase the bioavailability of minerals such as calcium, phosphorus, and magnesium, which are essential for bone growth and metabolic functions (Bielik and Kolisek, 2021; Suliburska et al., 2021). Additionally, probiotics can modulate lipid metabolism by reducing plasma cholesterol levels through the deconjugation of bile salts, which enhances cholesterol (Kumar et al., 2012; Song et al., 2023). The effects of probiotics on vitamin metabolism are also gaining attention, particularly B-complex vitamins such as B12, which are synthesized by certain gut microbes and play a vital role in energy metabolism (Morowitz et al., 2011; Bermúdez-Humarán et al., 2024). Additionally, this study demonstrated a consistent trend of increased immune organ weights in MLC supplemented with probiotics, indicating their potential to enhance immune system development and function. For instance, the spleen weight was significantly higher (p < 0.05) in probiotic-treated groups, with the T6 group showing the greatest weight (1.974 ± 0.091 g) compared to the control group (1.77 ± 0.12 g), suggesting that probiotics promote lymphoid tissue development, thereby increasing the spleen’s capacity to filter pathogens and produce immune cells. Similarly, thymus weights were markedly higher in probiotic-supplemented groups, with T6 achieving 4.67 ± 0.109 g compared to 3.84 ± 0.097 g in the control group, highlighting the role of probiotics in supporting T-cell maturation and adaptive immunity. The bursa of Fabricius, crucial for humoral immunity in birds, also showed significant weight increases in probiotic-treated groups, with T6 recording the highest weight (3.952 ± 0.1006 g) compared to the control group (3.53 ± 0.12 g). This finding aligns with prior research indicating that probiotics, such as L. plantarum and Bifidobacterium spp., enhance the production and function of B cells, thereby improving antibody-mediated defense against pathogens (Borda-Molina et al., 2018; Agustono et al., 2019; Bhogoju and Nahashon, 2022). Probiotics exert their effects through multiple mechanisms, including maintaining a balanced gut microbiota, which is crucial for immune homeostasis. They promote the growth of beneficial bacteria, competitively exclude pathogens, and reduce the production of harmful substances such as ammonia and enterotoxins, thereby protecting the gut lining and enhancing systemic immune function. Furthermore, probiotics stimulate both cellular and humoral immunity by modulating cytokine production, enhancing macrophage activity, and promoting lymphocyte proliferation. For example, they increase anti-inflammatory cytokines such as IL-10 while suppressing pro-inflammatory cytokines such as IL-6 and TNF-α, maintaining immune balance (Fijan, 2014; Borda-Molina et al., 2018). Additionally, probiotics influence the development of Peyer’s patches and specialized lymphoid tissues in the gut, enhancing antigen response and contributing to systemic immunity. These findings corroborate studies by Mahdavi (2005) and Raheem et al (2021), which reported that probiotics increase lymphoid tissue size and support both innate and adaptive immunity, although strain-specific differences and dosages remain key variables warranting further investigation. Probiotic microorganisms, generally recognized as safe, are essential components of the natural gastrointestinal microbiota, making them a practical and safe alternative to AGPs. Studies have demonstrated that even a single dose of probiotic bacteria can significantly enhance feed efficiency, FCR, and body weight gain in poultry, supporting their application in modern poultry farming (Fathima et al., 2022). The use of probiotics not only reduces the reliance on AGPs, mitigating the risks of AMR, but also promotes poultry welfare by fostering gut health and resilience to diseases. Moreover, improved productivity and nutrient utilization through probiotics align with the industry’s need for sustainable and ethical farming practices, offering a dual advantage of economic benefits and enhanced animal welfare (Alkhalf et al., 2010). Microbiota in the gastrointestinal tract aids in the production of digestive enzymes (Pan and Yu, 2013; Baümler and Sperandio, 2016). This study supports the conclusion that a single isolate of Bifidobacterium spp. can enhance the feed intake, nutrient intake, FCR, and feed efficacy of MLC (Fig. 3). Intestinal microbiota synthesizes probiotic strains such as Bifidobacterium spp., generating numerous metabolites, including short-chain fatty acids (SCFAs) (Louis et al., 2007; Markowiak-Kopeć and Śliżewska, 2020).
Fig. 3. FCR of MLC was enhanced by the supplementation of probiotics (L. acidophilus, L. plantarum, and Bifidobacterium spp.) as a replacement for AGPs during the finisher period (42 days old rearing). This study highlights the effectiveness of L. acidophilus, L. plantarum, and Bifidobacterium spp. in enhancing feed intake, nutrient utilization, FCR, and overall feed efficiency in MLCs, showcasing their potential as valuable probiotics. The intestinal microbiota synthesizes Bifidobacterium spp., producing bioactive metabolites, including SCFAs, which are essential for gut health and nutrient metabolism (Louis et al., 2007; Markowiak-Kopeć and Śliżewska, 2020). Likewise, L. plantarum robustly produces SCFAs that enhance nutrient absorption and lower gut pH, creating an unfavorable environment for pathogens (Zhao et al., 2024). Beyond these, L. acidophilus improves gut barrier integrity, modulates immune responses, and enhances mucosal IgA secretion, reducing intestinal permeability (Chandrasekaran et al., 2024). Enterococcus faecium also promotes nutrient uptake and feed efficiency by stabilizing the microbiome and inhibiting harmful bacteria such as Clostridium perfringens (Markowiak and Śliżewska, 2017). These probiotics also contribute to the synthesis of bacteriocins and vitamins, further supporting poultry health (Markowiak-Kopeć and Śliżewska, 2020). Notably, multi-strain formulations, combining strains such as Bifidobacterium spp. and Lactobacillus spp., demonstrate synergistic benefits by improving microbiota diversity and resilience against gastrointestinal challenges (Zhou et al., 2014; Wu et al., 2021). These findings underscore the versatile and potent role of probiotics in enhancing poultry productivity and health. SCFAs, primarily consisting of acetate, propionate, and butyrate, play a crucial role in regulating the intestinal health of poultry. They are predominantly absorbed from the intestinal tract and serve as a key substrate for energy production by enterocytes (Liu et al., 2021). Beneficial bacteria, such as Bifidobacterium and Lactobacillus, proliferate in poultry in the presence of SCFAs. Butyrate contributes to a positive feedback loop by increasing the populations of butyrate-producing bacteria such as Lactobacillus, Christensenellaceae, and Blautia (Mollica et al., 2017). This compound mitigates liver inflammation and fat accumulation associated with high-fat diets by promoting intestinal mucosal repair, fortifying tight junctions, and limiting the translocation of intestinal endotoxins to the liver. Butyrate and vitamin D have anti-inflammatory and antibacterial effects on Salmonella colitis through VDR. Combining the vitamins may help autoimmune and infectious colitis (Liu et al., 2023). SCFAs not only enhance gut microbe barrier function and contribute to gut homeostasis but also lower luminal pH and inhibit pathogen colonization. Several factors, including antibacterial peptides and immunoglobulins, regulate the dysbiosis of gut microbiota, which can lead to various digestive disorders such as intestinal inflammation (Panda A. K. et al., 2009; Couto et al., 2020). ConclusionThis study highlights the significant benefits of probiotic supplementation with L. acidophilus, L. plantarum, and Bifidobacterium spp. in improving nutrient intake, digestibility, and growth performance in MLC during the finisher period, emphasizing their potential as sustainable alternatives to AGPs. Enhanced nutrient utilization and absorption underscore the role of probiotics in promoting healthier poultry production systems. However, limitations such as the study’s focus on specific probiotic strains and a short timeframe suggest the need for further research. Future studies should explore long-term effects, optimal dosages, and strain-specific benefits, while also assessing probiotics’ influence on gut microbiota, immune responses, and interactions with diverse dietary compositions to fully understand their potential in advancing poultry health, welfare, and sustainability. AcknowledgmentsThe authors are grateful to the Faculty of Health, Medicine, and Life Sciences for providing animal enclosures for this study. The authors did not receive any funds for this study. Conflict of interestThe authors declare that they have no competing interests. ReferencesAbd El-Hack, M.E., El-Saadony, M.T., Shafi, M.E., Qattan, S.Y.A., Batiha, G.E., Khafaga, A.F., Abdel-Moneim, A.E. and Alagawany, M. 2020. Probiotics in poultry feed: a comprehensive review. J. Anim. Physiol. Anim. Nutr. 104(6), 1835–1850; doi:10.1111/jpn.13454. Abreu, R., Semedo-Lemsaddek, T., Cunha, E., Tavares, L. and Oliveira, M. 2023. Antimicrobial drug resistance in poultry production: current status and innovative strategies for bacterial control. Microorganisms 11(4), 953; doi:10.3390/microorganisms11040953. Agustono, B., Al Arif, M.A., Yunita, M.N., Purnama, M.T.E. and Ulkhaq, F. 2019. Bioactivity of digestive enzymes and histological descriptions of jejunum of broilers supplemented with sunflower seed flour (Helianthus annuus L). Indian Vet. J. 96(8), 12–15. Agustono, B., Lokapirnasari, W.P., Yunita, M.N., Kinanti, R.N., Cesa, A.E. and Windria, S. 2022. Efficacy of dietary supplementary probiotics as substitutes for antibiotic growth promoters during the starter period on growth performances, carcass traits, and immune organs of male layer chicken. Vet. World 15(2), 324–330; doi:10.14202/vetworld.2022.324-330. Al-Khalaifah, H.S. 2018. Benefits of probiotics and/or prebiotics for antibiotic-reduced poultry. Poult. Sci. 97(11), 3807–3815; doi:10.3382/ps/pey160. Alkhalf, A., Alhaj, M. and Al-Homidan, I. 2010. Influence of probiotic supplementation on immune response of broiler chicks. Egypt. Poult. Sci. 30(30), 271–280. Baümler, A.J. and Sperandio, V. 2016. Interactions between the microbiota and pathogenic bacteria in the gut. Nature 535(7610), 85–93; doi:10.1038/nature18849. Bermúdez-Humarán, L.G., Chassaing, B. and Langella, P. 2024. Exploring the interaction and impact of probiotic and commensal bacteria on vitamins, minerals and short chain fatty acids metabolism. Microb. Cell Fact. 23(1), 172; doi:10.1186/s12934-024-02449-3. Bhogoju, S. and Nahashon, S. 2022. Recent advances in probiotic application in animal health and nutrition: a review. Agriculture 12(2), 304; doi:10.3390/agriculture12020304. Bielik V, and Kolisek M. 2021. Bioaccessibility and Bioavailability of Minerals in Relation to a Healthy Gut Microbiome. Int J Mol Sci. 22(13):6803. doi: 10.3390/ijms22136803 Bogucka, J., Ribeiro, D.M., Bogusławska-Tryk, M., Dankowiakowska, A., da Costa, R.P.R. and Bednarczyk, M. 2019. Microstructure of the small intestine in broiler chickens fed a diet with probiotic or synbiotic supplementation. J. Anim. Physiol. Anim. Nutr. 103(6), 1785–1791; doi:10.1111/jpn.13182. Borda-Molina, D., Seifert, J. and Camarinha-Silva, A. 2018. Current perspectives of the chicken gastrointestinal tract and its microbiome. Comput. Struct. Biotechnol. J. 16, 131–139; doi:10.1016/j.csbj.2018.03.002. Broom, D. 2021. Farm animal welfare: a key component of the sustainability of farming systems. Veterinarski Glasnik, 75(2), 145–151. https://doi.org/10.2298/VETGL210514007B Chandrasekaran, P., Weiskirchen, S. and Weiskirchen, R. 2024. Effects of probiotics on gut microbiota: an overview. Int. J. Mol. Sci. 25(11), 1–20; doi:10.3390/ijms25116022. Cook, M.E. 2004. Antibodies : alternatives to antibiotics in improving growth and feed efficiency. J. Appl. Poult. Res. 13, 106–119. Couto, M.R., Gonçalves, P., Magro, F. and Martel, F. 2020. Microbiota-derived butyrate regulates intestinal inflammation: focus on inflammatory bowel disease. Pharmacol. Res. 159, 104947; doi:10.1016/j.phrs.2020.104947. Fathima, S., Shanmugasundaram, R., Adams, D. and Selvaraj, R.K. 2022. Gastrointestinal microbiota and their manipulation for improved growth and performance in chickens. Foods 11(10), 1401; doi:10.3390/foods11101401. Fijan, S. 2014. Microorganisms with claimed probiotic properties: an overview of recent literature. Int. J. Environ. Res. Public Health 11(5), 4745–4767; doi:10.3390/ijerph110504745. Gerken, M., Jaenecke, D. and Kreuzer, M. 2003. Growth, behaviour and carcass characteristics of egg-type cockerels compared to male broilers. World’s Poult. Sci. J. 59, 46–49. Kabir, S.M.L. 2009. The role of probiotics in the poultry industry. Int. J. Mol. Sci. 10(8), 3531–3546; doi:10.3390/ijms10083531. Kalia, V.C., Shim, W.Y., Patel, S.K.S., Gong, C. and J.-K. Lee. 2022. Recent developments in antimicrobial growth promoters in chicken health: opportunities and challenges. Sci. Total Environ. 834, 155300; doi:10.1016/j.scitotenv.2022.155300. Khalid, F.M., Shahzad, A.M., Sarwar, M., Rehman, A.U., Sharif, M. and Mukhtar, N. 2011. Probiotics and lamb performance: a review. African J. Agric. Res. 6(23), 5198–5203; doi:10.5897/AJAR11.1134. Krysiak, K., Konkol, D. and Korczyński, M. 2021. Review overview of the use of probiotics in poultry production. Animals 11(6), 1620; doi:10.3390/ani11061620. Kumar, M., Nagpal, R., Kumar, R., Hemalatha, R., Verma, V., Kumar, A., Chakraborty, C., Singh, B., Marotta, F., Jain, S. and Yadav, H. 2012. Cholesterol-lowering probiotics as potential biotherapeutics for metabolic diseases. Exp. Diabetes. Res. 2012, 902917; doi:10.1155/2012/902917. Lichovníková, M., Jandásek, J., Jürzl, M. and Dračková, E. 2009. The meat quality of layer males from free range in comparison with fast growing chickens. Czech J. Anim. Sci. 54(11), 490–497; doi:10.17221/3/2009-cjas. Liu, L., Li, Q., Yang, Y. and Guo, A. 2021. Biological function of short-chain fatty acids and its regulation on intestinal health of poultry. Front. Vet. Sci. 8, 736739; doi:10.3389/fvets.2021.736739. Liu, T., Sun, Z., Yang, Z. and Qiao, X. 2023. Microbiota-derived short-chain fatty acids and modulation of host-derived peptides formation: Focused on host defense peptides. Biomed. Pharmacother. 162, 114586; doi:10.1016/j.biopha.2023.114586. Lokapirnasari, W.P., Dewi, A.R., Fathinah, A., Hidanah, S., Harijani, N. Soeharsono, Karimah, B. and Andriani, A.D. 2017. Effect of probiotic supplementation on organic feed to alternative antibiotic growth promoter on production performance and economics analysis of quail. Vet. World 10(12), 1508–1514; doi:10.14202/vetworld.2017.1508-1514. Lokapirnasari, W.P., Pribadi, T.B., Al Arif, A., Soeharsono, S., Hidanah, S., Harijani, N., Najwan, R., Huda, K., Wardhani, H.C.P., Rahman, N.F.N. and Yulianto, A.B. 2019. Potency of probiotics Bifidobacterium spp. and Lactobacillus casei to improve growth performance and business analysis in organic laying hens. Vet. World 12(6), 860–867; doi:10.14202/vetworld.2019.860-867. Louis, P., Scott, K.P., Duncan, S.H. and Flint, H.J. 2007. Understanding the effects of diet on bacterial metabolism in the large intestine. J. Appl. Microbiol. 102, 1197–1208. Mahdavi, A.H., Rahmani, H. and Pourreza, J. 2005. Effect of probiotic supplements on egg quality and laying Hen’s performance. Int. J. Poult. Sci. 4, 488–492. Markowiak-Kopeć, P. and Śliżewska, K. 2020. The effect of probiotics on the production of short-chain fatty acids by human intestinal microbiome. Nutrients 12(4); doi:10.3390/nu12041107. Markowiak, P. and Śliżewska, K. 2017. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients 9(9), 1021; doi:10.3390/nu9091021. Marshall, B.M. and Levy, S.B. 2011. Food animals and antimicrobials : impacts on human health. 24(4), 718–733; doi:10.1128/CMR.00002-11. Mollica, M.P., Mattace Raso, G., Cavaliere, G., Trinchese, G., De Filippo, C., Aceto, S., Prisco, M., Pirozzi, C., Di Guida, F., Lama, A., Crispino, M., Tronino, D., Di Vaio, P., Berni Canani, R., Calignano, A. and Meli, R. 2017. Butyrate regulates liver mitochondrial function, efficiency, and dynamics in insulin-resistant obese mice. Diabetes 66(5), 1405–1418; doi:10.2337/db16-0924. Morowitz, M.J., Carlisle, E.M. and Alverdy, J.C. 2011. Contributions of intestinal bacteria to nutrition and metabolism in the critically ill. Surg. Clin. North Am. 91(4), 771–85, viii; doi:10.1016/j.suc.2011.05.001. Pan, D. and Yu, Z. 2013. Intestinal microbiome of poultry and its interaction with host and diet. Gut Microbes 5(1), 108–119; doi:10.4161/gmic.26945. Panda, A.K., Rama Rao, S.V., Raju, M.V. and Shyam Sunder, G. 2009. Effect of butyric acid on performance, gastrointestinal tract health and carcass characteristics in broiler chickens. Asian-Aust. J. Anim. Sci. 22(7), 1026–1031. Raheem, A., Liang, L., Zhang, G. and Cui, S. 2021. Modulatory effects of probiotics during pathogenic infections with emphasis on immune regulation. Front. Immunol. 12(April), 1–32; doi:10.3389/fimmu.2021.616713. Rahmat, N., Rudy, S. and Khaira, N. 2015. Pengaruh ransum dengan persentase serat kasar yang berbeda terhadap performa ayam jantan tipe medium umur 3-8 minggu. J. Ilm. Peternak. Terpadu. 3(2), 12–19. Ramadhanti, A.R., Puspita, N.O.J., Refalta, C.F., Kurnianto, H. and Saragih, H.T.S. 2021. Performance of male layer fed ration containing green algae (Spirogyra jaoensis) extract. Trop. Anim. Sci. J. 44(1), 100–107; doi:10.5398/tasj.2021.44.1.100. Song, X., Liu, Y., Zhang, X., Weng, P., Zhang, R. and Wu, Z. 2023. Role of intestinal probiotics in the modulation of lipid metabolism : implications for therapeutic treatments. Food Sci. Hum. Wellness 12(5), 1439–1449; doi:10.1016/j.fshw.2023.02.005. Suliburska J, Harahap IA, Skrypnik K, Bogdański P. 2021. The Impact of Multispecies Probiotics on Calcium and Magnesium Status in Healthy Male Rats. Nutrients. 13(10):3513. doi: 10.3390/nu13103513. Tsao, S.P., B.A. Nurrahma, R. Kumar, C.H. Wu, T.H. Yeh, et al. 2021. Probiotic enhancement of antioxidant capacity and alterations of gut microbiota composition in 6-hydroxydopamin-induced Parkinson’s disease rats. Antioxidants 10(11), 1823; doi:10.3390/antiox10111823. Wu, L., Tang, Z., Chen, H., Ren, Z., Ding, Q., Liang, K. and Sun, Z. 2021. Mutual interaction between gut microbiota and protein/amino acid metabolism for host mucosal immunity and health. Anim. Nutr. 7(1), 11–16; doi:10.1016/j.aninu.2020.11.003. Yulianto, B.Y., Al Arif, A. and Lokapirnasari, W.P. 2010. The potency of bifidobacterium spp. as an alternative to antibiotic growth promoters on feed conversion ratio, feed efficiency, and nutrient intake in laying hens. J. Southwest Jiaotong Univ. 56(6), 42–46. Zhang, M., Guo, Y., Su, R., Corazzin, M., Hou, R., Xie, J., Zhang, Y., Zhao, L., Su, L. and Jin, Y. 2022. Transcriptome analysis reveals the molecular regulatory network of muscle development and meat quality in Sunit lamb supplemented with dietary probiotic. Meat Sci. 194, 108996; doi:10.1016/j.meatsci.2022.108996. Zhao, X., Liu, S., Li, S., Jiang, W., Wang, J., Xiao, J., Chen, T., Ma, J., Khan, M.Z., Wang, W. and Li, M. 2024. Unlocking the power of postbiotics: a revolutionary approach to nutrition for humans and animals. Cell Metab. 36(4), 725–744; doi:10.1016/j.cmet.2024.03.004. Zhou, P., Tan, Y.Q., Zhang, L., Zhou, Y.M., Gao, F. and Zhou, G.H. 2014. Effects of dietary supplementation with the combination of zeolite and attapulgite on growth performance, nutrient digestibility, secretion of digestive enzymes and intestinal health in broiler chickens. Asian-Australasian J. Anim. Sci. 27(9), 1311–1318; doi:10.5713/ajas.2014.14241. | ||
| How to Cite this Article |
| Pubmed Style Agustono B, Yunita MN, Lokapirnasari WP, Warsito SH, Marbun TD, Windria S. Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promoters. Open Vet. J.. 2025; 15(2): 668-679. doi:10.5455/OVJ.2025.v15.i2.15 Web Style Agustono B, Yunita MN, Lokapirnasari WP, Warsito SH, Marbun TD, Windria S. Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promoters. https://www.openveterinaryjournal.com/?mno=224805 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.15 AMA (American Medical Association) Style Agustono B, Yunita MN, Lokapirnasari WP, Warsito SH, Marbun TD, Windria S. Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promoters. Open Vet. J.. 2025; 15(2): 668-679. doi:10.5455/OVJ.2025.v15.i2.15 Vancouver/ICMJE Style Agustono B, Yunita MN, Lokapirnasari WP, Warsito SH, Marbun TD, Windria S. Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promoters. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 668-679. doi:10.5455/OVJ.2025.v15.i2.15 Harvard Style Agustono, B., Yunita, . M. N., Lokapirnasari, . W. P., Warsito, . S. H., Marbun, . T. D. & Windria, . S. (2025) Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promoters. Open Vet. J., 15 (2), 668-679. doi:10.5455/OVJ.2025.v15.i2.15 Turabian Style Agustono, Bodhi, Maya Nurwartanti Yunita, Widya Paramita Lokapirnasari, Sunaryo Hadi Warsito, Tabita Dameria Marbun, and Sarasati Windria. 2025. Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promoters. Open Veterinary Journal, 15 (2), 668-679. doi:10.5455/OVJ.2025.v15.i2.15 Chicago Style Agustono, Bodhi, Maya Nurwartanti Yunita, Widya Paramita Lokapirnasari, Sunaryo Hadi Warsito, Tabita Dameria Marbun, and Sarasati Windria. "Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promoters." Open Veterinary Journal 15 (2025), 668-679. doi:10.5455/OVJ.2025.v15.i2.15 MLA (The Modern Language Association) Style Agustono, Bodhi, Maya Nurwartanti Yunita, Widya Paramita Lokapirnasari, Sunaryo Hadi Warsito, Tabita Dameria Marbun, and Sarasati Windria. "Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promoters." Open Veterinary Journal 15.2 (2025), 668-679. Print. doi:10.5455/OVJ.2025.v15.i2.15 APA (American Psychological Association) Style Agustono, B., Yunita, . M. N., Lokapirnasari, . W. P., Warsito, . S. H., Marbun, . T. D. & Windria, . S. (2025) Optimizing male layer chicken performance and health with probiotic supplementation: A sustainable alternative to antibiotic growth promoters. Open Veterinary Journal, 15 (2), 668-679. doi:10.5455/OVJ.2025.v15.i2.15 |