| Research Article | ||
Open Vet. J.. 2025; 15(2): 714-723 Open Veterinary Journal, (2025), Vol. 15(2): 714-723 Research article Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, IraqThanaa Ismael Jawad1* and Ghaidaa Abass Jasim21Nursing Techniques Department, Technical Institute of Al-Dewaniyah, AL-Furat AL-Awsat Technical University, Najaf, Iraq 2Department of Microbiology, College of Veterinary Medicine, University of Al-Qadisiyah, Al-Diwaniyah, Iraq *Correspondence to: Thanaa Ismael Jawad. Nursing Techniques Department, Technical Institute of Al-Dewaniyah, AL-Furat AL-Awsat Technical University, Najaf, Iraq. than.dw [at] atu.edu.iq Submitted: 25/10/2024 Accepted: 24/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
AbstractBackground: Avian coccidiosis is one of the most significant and impactful diseases affecting the commercial poultry industry globally. Aim: This study aimed to identify the Eimeria species associated with chicken coccidiosis. Methods: Conducted from November 2023 to April 2024, the research involved 150 avian samples. Intestinal samples were collected from chickens suspected of having coccidiosis at veterinary hospitals and clinics in Diwaniyah Governorate. To confirm the infection, a longitudinal incision was made in the ceca of the suspected infected chicken, and a direct swab was obtained. Part of the cecal contents was placed in a test tube for the fecal flotation method, while the remainder was stored in a separate test tube and refrigerated for DNA extraction. Additionally, a 1-cm tissue sample was collected from the cecum and preserved in formalin to assess histopathological changes. Samples were analyzed using microscopy and conventional polymerase chain reaction (PCR), focusing on the small subunit 18S rRNA gene of the parasite, specifically internal transcribed spacer 1 (ITS 1). Results: Microscopic analysis revealed parasitic oocysts in 19.3% of the fecal samples from the examined poultry. Furthermore, the PCR results indicated that Eimeria tenella was present in 22% of the fecal samples. The ITS-1 sequences of E. tenella from this study showed a high degree of similarity to those recorded in GenBank at 95%, with sequences from India (GQ856301, JX853825.1, JX853831.1, and PP002176.1), China (JX477100.1, FJ449692.1, GQ153631.1, and FJ449691.1), and Turkey (HQ680474.1) species. Overall, E. tenella, which causes coccidiosis in poultry, leads to significant histopathological alterations in the intestinal tissues of infected birds, including epithelial damage. Conclusion: This study concludes that E. tenella is present in the tested poultry within the governorate. Keywords: Broiler, Eimeria tenella, Iraq, PCR, Poultry IntroductionAvian coccidiosis is one of the most significant and devastating diseases affecting the commercial poultry industry globally. Coccidiosis is the most prevalent intestinal parasitic disease affecting poultry populations worldwide (Amae et al., 2012). There are nine well-established species of Eimeria coccidia that commonly infect chickens. These include Eimeria acervulina, E. brunetti, E. maxima, E. mitis, E. necatrix, E. praecox, E. mevati, E. hagani, and E. tenella. Each Eimeria species exhibits a predilection and localizes to a specific region within the chicken’s digestive tract. Some overlap and interference in the development and proliferation of these Eimeria species can occur within the avian gastrointestinal system (Gasser et al., 2005). All species of Eimeria that infect chickens can cause coccidiosis, but four of these (E. acervulina, E. maxima, E. necatrix, and E. tenella) are generally considered most important due to their pathogenicity (Blake et al., 2021). Due to the challenges of reliably identifying some chicken Eimeria species based solely on their morphological characteristics, diagnostic laboratories are increasingly turning to DNA-based analytical technologies for the precise and definitive identification of these parasitic protozoa (Morgan et al., 2009). The advancement of molecular techniques and tools has enabled not only the diagnosis of coccidial infections but also the study of genetic diversity among these parasitic pathogens. This is achieved by analyzing small quantities of coccidial oocysts using molecular markers as an analytical approach (Costa et al., 2001). This study aimed to determine the genetic diversity of E. tenella in broiler chickens in Al-Diwaniyah province, Iraq. Understanding the histopathological changes associated with coccidiosis is vital for assessing disease severity and its effects on poultry health and production. Furthermore, this research seeks to deepen our understanding of the disease’s impact on tissue structure and overall poultry health. Materials and MethodsFecal sample collectionA total of 150 avian fecal samples were collected directly from the cecum of poultry and placed in clean, labeled plastic containers. Subsequently, the samples were transported to the Parasitology Laboratory of the College of Veterinary Medicine at the University of Al-Qadisiya. The collection of samples occurred in various regions of Al-Diwaniyah province from November 2023 to April 2024. Additionally, cecal samples were placed in separate test tubes and refrigerated to facilitate DNA extraction. A 1-cm segment was taken from the cecum of chickens where the presence of oocysts had been confirmed and preserved in 10% formalin for histological sectioning. Direct examination method (microscopic examination)The first step in the current study was to perform a direct smear of the intestinal contents of chickens suspected of being infected and suffering from bloody diarrhea. The size of a matchstick head was taken from this content and placed on a glass slide, and a drop of distilled water was placed and mixed well. Then, the slide was covered and then examined. Under a microscope with a magnification of 40×. All samples were examined using the flotation method (Eckert et al., 1995) to investigate the presence of Eimeria oocysts. Polymerase chain reaction and DNA sequencingFor DNA extraction, genomic DNA was extracted from the cecal contents of 150 chickens using the AddPrep Genomic DNA Extraction Kit (Geneaid, USA). Following extraction, the DNA was purified. To measure the purity and concentration of the extracted DNA, a nanodrop spectrophotometer was used. The primers used in this study were designed based on the internal transcribed spacer-1 (ITS-1) sequence using lyophilized primers provided by Bioneer Company (Korea). The E. tenella primers were amplified from genomic DNA using a species-specific forward primer (5’-AATTTAGTCCATCGCAACCCTTG-3’) and a reverse primer (5’-CGAGCGCTCTGCATACGACA-3’), targeting a segment of approximately 450 base pair (bp). The primer preparation involved subjecting the reaction mixtures to denaturation at 96°C for 5 minutes, followed by 35 cycles of denaturation at 94°C for 25 seconds, annealing at 61°C for 25 seconds, and extension at 72°C for 40 seconds. The final extension step was performed at 72°C for 5 minutes, as shown in Table 2. Each sample (10 μl) was analyzed by electrophoresis under specific conditions. An amplification mixture was prepared in polymerase chain reaction (PCR) tubes for the detection of the 18S rRNA region using primers (Su et al., 2003). Two concentrations of agarose gel were prepared: 1% and 1.5%. The 1% agarose gel was used for electrophoresis after DNA extraction, whereas the 1.5% agarose gel was used following PCR detection. Subsequently, the PCR products were purified and sent to Macrogen (Korea) for partial gene sequencing. Finally, a phylogenetic tree was generated using MEGA X (Kumar et al., 2018), and similar sequences from around the world were identified using the NCBI database. Histopathological examinationA tissue sample was collected from the cecum of a chicken affected by coccidiosis, sourced from poultry farms in the Diwaniyah Governorate, and from infected cases presented at veterinary clinics. The sample was rinsed with water to remove blood and debris and then preserved in 10% formalin for subsequent tissue sectioning. The specimens were then cut into small cubic pieces, approximately 1 cm³ in size, and washed with tap water for 2 hours. Table 1. The primer names and sequences according to product size (ITS-1).
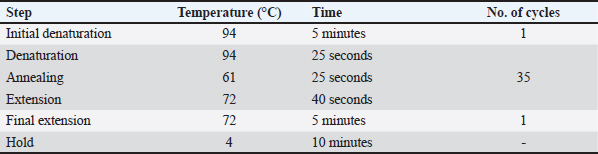
Table 2. PCR Program for the amplification of the ITS-1sSequence.
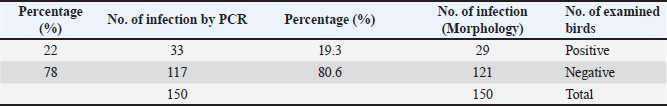
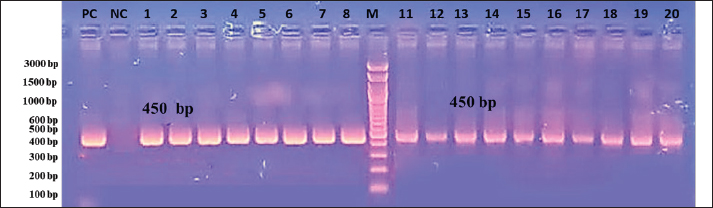
The tissue was then processed according to the standard protocol described by Bancroft and Stevens (2002). The procedures included: first, fixing the samples immediately after dissection in 10% formalin for a period ranging from 24 to 48 hours. After fixation, the samples were washed with running water for half an hour to remove any residual fixative. Next, dehydration was performed using a series of alcohol solutions, starting with 50%, followed by 60%, 70%, 80%, 90%, and finally 100%, with each concentration applied for 2 hours. Subsequently, clearing was achieved using xylene twice, with each solution lasting for half an hour. The tissue specimens were infiltrated by placing them in paraffin wax at 58°C–60°C for two sessions of 2 hours each. After infiltration, the specimens were embedded in paraffin wax and allowed to cool for 24 hours. The specimens were then sectioned to a thickness of 5–7 µm using a rotary microtome. For mounting, a thin smear of Mayer’s egg albumin was applied to fix the sections onto glass slides. Rehydration was performed using an alcohol series, starting at 100%, followed by 95%, and finally 90%. Finally, the specimens were stained with hematoxylin and eosin (H&E), and the slide was mounted by adding DPX before placing a cover slip over the sections on the glass slides (Suvarna et al., 2013). Statistical analysisThe case–control test was applied in SPSS to determine the statistical significance of the data. A p value of <0.05 was considered significant. ResultsMacroscopic examinationIn the macroscopic examination, the predominant pathological signs observed in most infected chickens are substantial hemorrhage in the cecum, which can be either liquid or clotted. The ceca walls are noted to be dark brown, appearing almost black in color. Microscopic examinationThe microscopic examination results indicated that the infection rate was 19.3% among 150 samples taken from intestinal contents suspected of being infected with Eimeria sp. (Table 3). In this study, traditional microscopic examination revealed that 19.3% of the samples were indeed infected with Eimeria species. This infection rate aligns with the findings reported by a researcher, which indicated a total Eimeria species infection rate of 19.3% in house-reared chickens from Muthanna Governorate (Fig. 1). Molecular examinationDNA extraction was successfully performed on 150 samples collected from the cecum of chickens suspected of coccidiosis, particularly those exhibiting bloody diarrhea. After confirming the presence of eggs through microscopic examination, these samples were selected for molecular analysis. The results of the molecular examination revealed that the percentage of chickens infected with Eimeria sp. was consistent at 22%, as shown in Table 2. A single infection of E. tenella was observed. Among the samples, 22% tested positive, indicating successful ITS-1 amplification using conventional PCR techniques. Furthermore, the results confirmed that all field isolates were identified as E. tenella, as they produced a specific DNA fragment of 450 bp, resulting in a DNA product within a defined size range (Fig. 2). DNA sequencer technique and phylogenetic analysisThe genetic tree diagram was generated using the molecular sequence of ITS-1. The genetic analysis revealed similar genetic affinity among the local species. Three PCR-positive samples underwent DNA sequencing to identify avian Eimeria species molecularly, and the results confirmed that the samples were diagnosed as E. tenella (NCBI-registration numbers: H240724, H240725, and H240726), as illustrated in Figure 3. DiscussionThis percentage of infection was comparable to the 10% infection rate documented by another study on broiler chickens in Sudan (Alzib and Abdelnabi, 2017). However, this rate is considered low compared with the 87% infection rate reported in China (Huang et al., 2017). In Iran, the prevalence rate was found to be 31.8%, while a separate study in Tabriz, northern Iran reported an infection rate of 55.96% (Nematollahi et al., 2009). Additionally, another report indicated an infection rate of 30.68% in Diwaniyah Governorate (Meteab et al., 2021). Several factors may have significantly contributed to the reduction of the infection rate in Diwaniyah. Among these factors is the maintenance of environmental cleanliness, which minimizes the presence of viruses and parasites, thereby reducing the risk of infection. In addition, decreasing the number of birds in designated spaces can lower the risk of disease transmission. Providing appropriate care, including vaccination and regular medical examinations, further reduces the risk of infection. Table 3. Infection rates of Eimeria spp. microscopically and by PCR.
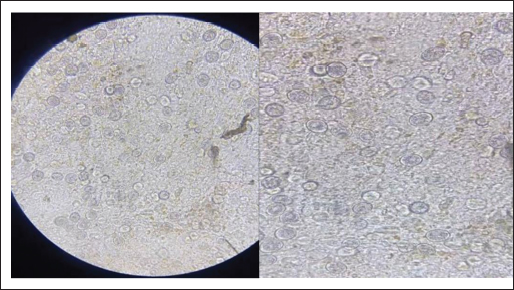
Fig. 1. Non-sporulated oocyst: 40×.
Fig. 2. PCR product of agarose gel electrophoresis of Eimeria ITS-1 sequence (450 bp), where M: 3,000 bp DNA ladder; lanes 1–9 positive samples. In comparing the present study with findings from foreign research, the results are inconsistent with those reported by another researcher (Fatoba et al., 2020), who used nested species-specific ITS-1 primers for molecular diagnosis and identified five species of Eimeria (E. tenella, E. maxima, E. acervulina, E. brunetti, and E. mitis) in South Africa. However, this finding is similar to that reported by another study (Tan et al., 2017), in which E. maxima and E. tenella were successfully identified by a distinct 278 bp band on agarose gels. Additionally, species-specific PCR for E. maxima yielded 205-bp amplicons, with positive PCR reactions obtained from fecal samples in Korea. Moreover, our findings align precisely with those reported by another researcher (Gharekhani et al., 2014), who identified all isolates as E. tenella using species-specific PCR amplification of the ITS1 rDNA region from oocysts extracted from poultry fecal samples collected across eight regions in China. In Iran, E. tenella was also identified as the dominant species, as outlined by another study (Alam et al., 2021), which detected E. maxima, E. necatrix, E. tenella, E. acervulina, and E. mitis, with a prevalence of Eimeria spp. at 31.5% (126 out of 400 samples), thus making E. tenella the most prevalent species in Khuzestan. The detection of highly pathogenic E. tenella, E. necatrix, and E. brunetti, along with the more prevalent E. acervulina and the less pathogenic E. mitis, underscores the diversity of Eimeria species in the study area. Furthermore, the use of ITS1-based PCR is recommended for the reliable diagnosis of chicken coccidiosis on a large scale to monitor its epizootiological prevalence in Bangladesh (Lee et al., 2011). Our findings were also consistent with those reported by another researcher (Patra et al., 2010), in which PCR using specific primers for E. tenella successfully amplified only the ITS1 regions of ribosomal DNA (rDNA). The primer pair designed for genus specificity and E. tenella exclusively amplified their intended target DNA, with no cross-species amplification observed from heterologous primer pairs. Notably, the percentage of the specific gene E. tenella was lower than the 59% reported in a study by another researcher (Ahmed and Albakri, 2021) for ITS1. The results of the DNA sequencing comparison between the local and global strains revealed that the ITS-1 sequences of E. tenella from this study demonstrated a high degree of similarity to E. tenella sequences available in GenBank. Specifically, there was a 95% identity with sequences from Indian species (GQ856301, JX853825.1, JX853831.1, and PP002176.1), Chinese species (JX477100.1, FJ449692.1, GQ153631.1, and FJ449691.1), and Turkish species (HQ680474.1). The genetic similarity between Eimeria isolates from local poultry and strains from countries such as China, India, and Turkey may be attributed to several factors. First, significant poultry trade in these countries may facilitate the exchange of pathogenic isolates. In addition, the shared environmental and agricultural conditions in these regions can support the adaptation of Eimeria, leading to genetic similarities. Furthermore, natural selection pressure from environmental and health factors may shape the isolates, resulting in the development of shared genetic traits. Advances in genome sequencing techniques have also enhanced the ability to compare genes among different isolates, thus making genetic similarities more apparent. These factors contribute to the observation that isolates from different regions can exhibit genetic similarities, especially given that these countries are neighboring Iraq and share overlapping borders. In contrast, the identity rate was lower at 76% with the Pakistani species (MN830381.1) and the strain isolated from broiler chickens in northern Iraq (Sulaymaniyah) (PP229210.1). This lower identity likely explains the amplification of Eimeria species by PCR. The observed genetic divergence may also be related to the size of the product, which reached 271 bp for the strain isolated from broiler chickens in northern Iraq. They demonstrated that 18S rDNA gene sequences and the ITS 1 region derived from ribosomal RNA (rRNA) genes can be effectively used for the genetic characterization and phylogenetic analysis of certain Eimeria species. Histological examinationThe cecum exhibited the strongest correlation among the diagnostic methods, showing moderate agreement between macroscopic examination and histopathology, as well as between macroscopic examination and PCR, and strong agreement between histopathology and PCR. This may be attributed to the greater intestinal content in this segment across all collected samples. The reasons for selecting the cecum for tissue sectioning were previously reported (Balestrin et al., 2022).
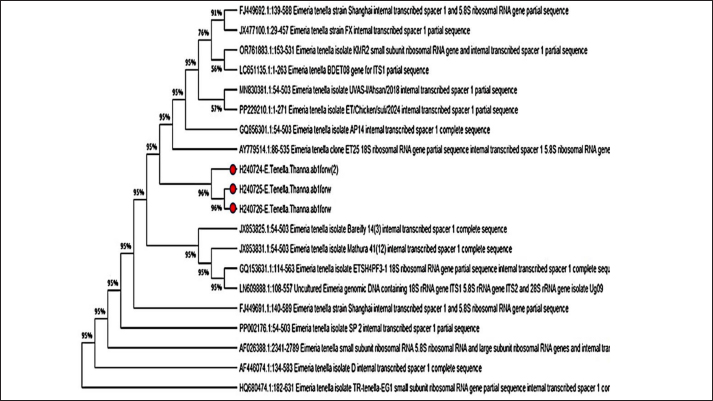
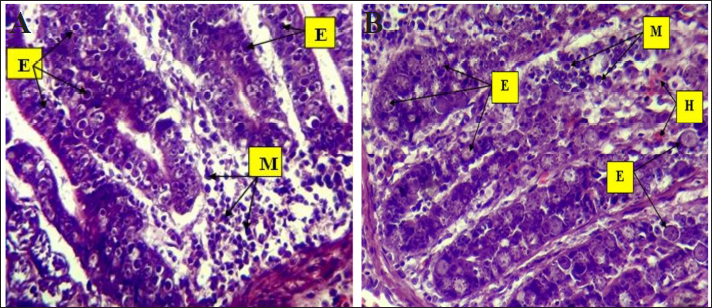
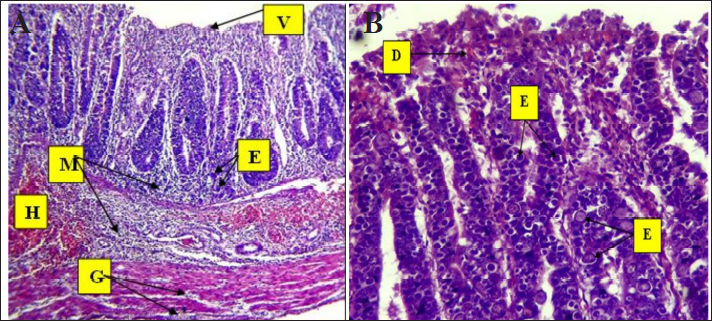
Fig. 3. Phylogenetic tree analysis of E. tenella in chickens (ITS-1) partial sequence constructed using multiple alignments using MEGA X software (Version: MEGA, 11.0.11) (Kumar et al, 2018). The identified sequences are shown with red circles, followed by the obtained accession numbers. These results were compared with sequences identified in China, India, Australia, and Turkey. Light microscopic analysis of hematoxylin and eosin-stained sections showed that the histological examination of cecum samples from chickens infected with Eimeria spp. indicated monocyte infiltration in the mucosal layer and muscle fiber degeneration. In addition, desquamation of the intestinal lining cells was noted, as illustrated in Figure 4. Eimeria parasites, which cause coccidiosis in poultry, induce significant pathological changes in the cecum of infected birds. These changes generally encompass as follows: Mucosal Damage: The parasite invades the intestinal epithelium resulting in necrosis and shedding of the mucosal layer, thereby compromising the integrity of the intestinal lining, as depicted in Figure 3. Monocyte Infiltration: There is an infiltration of monocytes and other immune cells in response to the infection, indicating an inflammatory response, as shown in Figures 4 and 5. The infiltration was pronounced, particularly involving eosinophils and macrophages, as illustrated in Figures 5 and 6. These findings were consistent with previous observations (Nonkookhetkhong and Chalalai, 2021), indicating that monocytes were the most effective white blood cells in response to E. tenella infection, with monocytosis associated with this infection. This is consistent with another study on Eimeria sp. infection (Taubert et al., 2009). As explained by Qureshi (2003), monocytes and macrophages operate through chemotaxis, which involves their migration toward an inflammatory gradient. As noted in monocyte infiltration, there is an infiltration of monocytes and other immune cells in response to the infection, indicating an inflammatory response, as shown in Figures 5 and 6. The infiltration was pronounced, particularly involving eosinophils and macrophages, as illustrated in Figures 6 and 7. These findings were aligned with previous observations (AL-Saegh, 2018), indicating that monocytes were the most effective white blood cells in response to E. tenella infection, with monocytosis associated with this infection. This result is consistent with that of another study on Eimeria sp. infection (Chapman, 2001). As explained by Alzib and Abdelnabi (2017), monocytes and macrophages operate through chemotaxes, which involves their migration toward an inflammatory gradient. The clusters of developmental stages of the Eimeria parasite, including schizonts and oocysts, were also noted. Additionally, necrosis and hemorrhage were observed in the submucosal and muscular layers, as shown in Figures 5 and 6. This result was inconsistent with an observation (Adamu et al., 2013) that only a small number of E. brunetti oocysts were found, and no schizonts were identified. Helal et al. (2019) observed that while a significant number of E. tenella oocysts and clusters of large schizonts were present, only a small number of E. brunetti oocysts were identified, and no schizonts were found. The cells lining the intestine may undergo shedding, further disrupting the absorptive surface of the brush border and contributing to diarrhea and malabsorption. The findings of this study are consistent with those reported by Helal et al. (2019). Villus atrophy and destruction of the brush border were also noted, as shown in Figures 6 and 7. The cause was linked to Zulpo et al. (2007), who noted that Eimeria spp. disrupt the balance between cell proliferation and extrusion in intestinal villi, resulting in cell loss. Two phases of the Eimeria sp. life cycle occur within epithelial cells, leading to their destruction. In this study, the histopathological findings of atrophy and increased spacing between intestinal villi in experimentally inoculated broiler chickens were consistent with the findings reported previously. In cases of severe injury, significant bleeding was observed due to the developmental stages of the Eimeria parasite. There was notable infiltration of inflammatory cells, particularly mononuclear cells, accompanied by necrosis in the submucosal layer and fluid accumulation in both the muscular and submucosal layers, as shown in Figures 6 and 8. The bleeding can be linked to the presence of developmental forms such as oocysts and schizonts. This observation is consistent with the findings of Chanie et al. (2009), who noted a large number of oocysts, schizonts, and extensive tissue damage in the ceca, indicating the severity of E. tenella infection. The second-generation schizont, known for causing substantial tissue damage, hemorrhage, disruption of the cecal glands, and destruction of both the mucosal and muscularis layers, has been identified as the most harmful stage of E. tenella (Jihad et al., 2021).
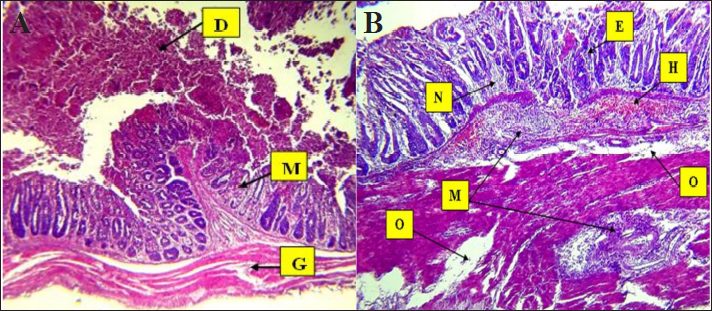
Fig. 4. Histopathological changes in Eimeria tenella infection in poultry. A. Desquamation of epithelial enterocytes lining villi (D), mononuclear cell (M) infiltration in mucosa, and degeneration of muscle fibers 4× H&E. There is marked degeneration (G) muscular smooth fibers (G). B. Clusters of Eimeria at different developmental stages (E) with severe hemorrhage (H). There is heavy infiltration of mononuclear cells (M) with necrosis (N) of the submucosa and severe edema (O) in the submucosa and muscularis: 4× H&E.
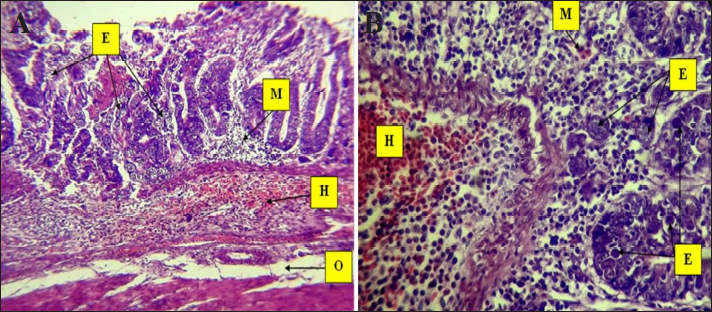
Fig. 5. Histopathological changes in Eimeria tenella infection in poultry. A. Clusters of Eimeria different (E) developmental stages with severe hemorrhage and heavy infiltration of mononuclear cells (M) with submucosal necrosis and submucosal and muscularis with severe hemorrhage (H). Severe edema (O) appears in the submucosa and muscularis 4× H&E. B. Different stages of Eimeria (E) in the epithelium of crypts and mucosa with severe and infiltration of mononuclear cells (M), mainly eosinophils, in the submucosa. There is severe hemorrhage (H): 40× H&E.
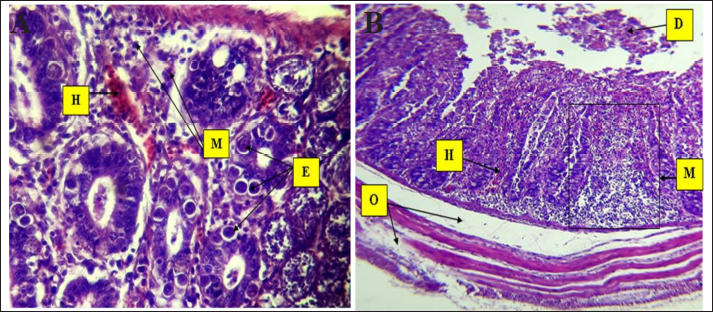
Fig. 6. Histopathological changes in Eimeria tenella infection in poultry. A. Different stages of Eimeria schizonts in the epithelium of crypts and brush borders of villi with severe infiltration of mononuclear cells (mainly eosinophils and macrophages in the submucosa. The infiltration of inflammatory cells (M) and hemorrhage (H) in the submucosa. The clusters of Eimeria at different developmental stages (E) 40× H&E. B. Marked desquamation (D) of epithelial enterocytes lining the villi, loss of villi and epithelial tissue, disruption of the intestinal mucosa, severe degeneration and muscular edema (O), infiltration of inflammatory cells (M) and hemorrhage (H) in the submucosa: 4× H&E.
Fig. 7. Histopathological changes in Eimeria tenella infection in poultry. A. Higher magnification of different stages of Eimeria schizonts (E) in the epithelium of crypts and brush borders of villi with severe infiltration of mononuclear cells (M), mainly eosinophils and macrophages in the submucosa: 40× H&E. B. Higher magnification image of heavy infestation of Eimeria schizonts (E) in the crypts with high infiltration of mononuclear cells (M) and hemorrhage (H) in the submucosa: 40× H&E.
Fig. 8. Histopathological changes in Eimeria tenella infection in poultry. A. Marked shortening and loss of villi (V), Eimeria schizonts (E), infiltration of inflammatory cells (M) in the intestinal mucosa and submucosa, severe hemorrhage (H) in submucosa, and marked degeneration (G) of muscular smooth fibers: 4× H&E. B. Developmental forms of Eimeria (E) are detected in epithelial cells of brush borders of villi and disruption of the intestinal mucosa (D): 40× H&E. ConclusionThe study concludes that E. tenella is present in the tested poultry within the governorate. AcknowledgmentsThe authors would like to thank the College of Veterinary Medicine, University of Al-Qadisiyah, Al-Diwaniyah City, Iraq. Conflict of interestThere are no conflicts of interest to declare. FundingThe study did not receive any financial support, but the authors’ personal assets funded the study. Authors’ contributionsAll authors participated in all parts of the study. Data availabilityThe data presented in this study are available upon request from the corresponding author. ReferencesAdamu, M., Boonkaewwan, C., Gongruttananun, N. and Vongpakorn, M. 2013. Hematological, biochemical and histopathological changes caused by coccidiosis in chickens. Kasetsart J. Nat. Sci. 47, 238–246. Ahmed, AM. and Albakri, H.S. 2021. Phynotypic and genotypic identification of Eimeria species in backyard chicken in Nineveh governorate, Iraq. Iraqi J. Vet. Sci. 35(Supplement II), 41–46. Alam, M.Z., Dey, A.R., Parvin, S., Akter, S., and Rony, S.A. 2021. ITS1-PCR based identification of chicken Eimeria species in poultry litter from Mymensingh district, Bangladesh. Adv. Vet. Anim. Res. 8(3), 489–493. AL-Saegh, H.A.H. 2018. Hemato-biochemical and histopathological changes caused by coccidiosis in broilers. Kufa J. Vet. Med. Sci. 9(1), 11–20. Alzib, A.A. and Abdelnabi, G.H. 2017. Eimeria Spp. Infection in some broiler farms in Khartoum State, Sudan. SOJ. Vet. Sci. 3(4), 1–3. Amae, A., Worku, W. and Negussie, H. 2012. Coccidiosis prevailing in parent stocks: a comparative study between growers and adult layers in Kombolcha poultry breeding and multiplication centre, Ethiopia. Global Vet. 8, 285–291. Balestrin, P.W.G., Balestrin, E., Santiani, F., de Cristo, T.G., Pereira, D.G., Bonatto, G.R., Bilick, J.V. and Casagrande, R.A. 2022. Comparison of macroscopy, histopathology, and PCR for diagnosing Eimeria spp. in broiler chickens. Pesq. Vet. Bras. 42, e06968–e06975. Bancroft, J.D. and Stevens, H. 2002. Theory and practice of histological techniques. 4th ed. Nottingham, UK: Queen’s Medical Center, University of Hospital NHS Trust. Blake, D., Vrba, V., Xia, D., Jatau, I.D., Spiro, S., Nolan, M.J., Underwood, G. and Tomley, F. 2021. Genetic and biological characterization of three cryptic Eimeria operational taxonomic units that infect chickens (Gallus gallus domesticus). Int. J. Parasitol. 51(8), 621–634. Chanie, M., Negash T. and Tilahun S.B. 2009. Occurrence of concurrent infectious diseases in broiler chickens is a threat to commercial poultry farms in Central Ethiopia. Trop. Anim. Health Prod. 41(7), 1309–1317. Chapman, H.D. 2001. Use of anticoccidial drugs in broiler chickens in the USA: analysis for the years 1995 to 1999. Poult. Sci. 80, 572–580. Costa, C.A.F., Gomes, R.F., Melo, M.N., Ribeiro, M.F.B. 2001. Eimeria parasites of domestic fowl: genetic relationships of different isolates estimated from random polymorphic DNA. Parasitol. Res. 87, 459–466. Eckert, J., Taylor, M., Cat chpole, J., Licois, D., Coudert, P. and Bucklar, H. 1995. Identification of Eimeria species and strains. Morphological characteristics of oocysts. Biotechnology: guidelines on techniques in coccidiosis research. (16602), 103–119. Fatoba, A.J., Zishiri, O.T., Blake, D.P., Peters, S.O., Lebepe, J., Mukaratirwa, S. and Adeleke, M.A. 2020. Study on the prevalence and genetic diversity of Eimeria species from broilers and free-range chickens in KwaZulu-Natal province, South Africa. Onderstepoort J. Vet. Res. 87(1), e1–e10. Gasser, R.B., Skinner, R., Fadavi, R., Richards, G. and Morris, G. 2005. High-throughput capillary electrophoresis for the identification and differentiation of seven species of Eimeria from chickens. Electrophoresis 26, 3479–3485. Gharekhani, J., Sadeghi-Dehkordi, Z. and Bahrami, M. 2014. Prevalence of coccidiosis in broiler chicken farms in Western Iran. J. Vet. Med. 2014, 980604–980607. Helal, I.B., Ahmed, S.N., Amer, S.E. and El Shershaby, R.E. 2019. Histopathological studies on experimentally Eimeria tenella-infected broiler chicken. Egypt. J. Exp. Biol. (Zool.). 15(1), 25–34. Hemzah, M.A. 2013. Prevalence of subclinical coccidiosis associated with house reared chickens in Al-Muthanna province, Iraq. Kufa J. Vet. Med. Sci. 4(1), 128–133. Huang, Y., Ruan, X., Li, L. and Zeng, M. 2017. Prevalence of Eimeria species in domestic chickens in Anhui province, China. J. Parasit. Dis. 41(4), 1014–1019. Jihad, W.S., Ahmed, J.A. and Najim, H.A. 2021. Clinical and pathological scoring of avian coccidiosis in Basra Province, Iraq. Turk. J. Physiother. Rehabil. 32(3), 18757–18765. Kumar, S., Stecher, G., Li, M., Knyaz, C. and Tamura, K. 2018. MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 35, 1547–1549. Lee H.A., Hong, S., Chung, Y. and Kim, O. 2011. Sensitive and specific identification by polymerase chain reaction of Eimeria tenella and Eimeria maxima, important protozoan pathogens in laboratory avian facilities. Lab. Anim. Res. 27(3), 255–258. Meteab, B. K., Kareem, H. I., Hammadi, H. A. and Abedalrazzaq, M. 2021. Prevalence of coccidiosis in the chickens of Diwaniya city, Iraq. Iran. J. Ichthyol. 8, 155–162. Morgan, J.A., Morris, G.M., Wlodek, B.M., Byrnes, R., Jenner, M., Constantinoiu, C.C., Anderson, G.R., Lew-Tabor, A.E., Molloy, J.B., Gasser, R.B. and Jorgensen, W.K. 2009. Real-time polymerase chain reaction (PCR) assays for the specific detection and quantification of seven Eimeria species that cause coccidiosis in chickens. Mol. Cell Probes. 23(2), 83–89. Nematollahi, A., Moghaddam G. and Pourabad R.F. 2009. Prevalence of Eimeria species among broiler chicks in Tabriz (northwest of Iran). Mun. Ent. Zool. 4(1), 53–58. Nonkookhetkhong, T. and Chalalai, T. 2021. Lesion scores, oocysts output, hematological and histopathological changes of the 7 days life cycle of Eimeria tenella in broilers. Res. Sq. 32(1), 13–21 Patra, G., Ali, M.A., Chanu, Kh. V., Jonathan, L., Joy, L.K., Prava, M., Ravindran, R., Das, G. and Devi, L.I. 2010. PCR based diagnosis of Eimeria tenella infection in broiler chicken. Int. J. Poultry Sci. 9(8), 813–818. Qureshi, M.A. 2003. Avian macrophage and immune response: an overview. Poultry Sci. 82(5), 691–698. Su, Y.C., Fei, A.C. and Tsai, F.M. 2003. Differential diagnosis of five avian Eimeria species by polymerase chain reaction using primers derived from the internal transcribed spacer 1 (ITS-1) sequence. Vet. Parasitol. 117, 221–227. Suvarna, S.K., Layton, C. and Bancroft, J.D. 2013. Bancroft’s theory and practice of histological techniques. Nottingham, UK: Elsevier. Tan, L., Li, Y., Yang, X., Ke, Q., Lei, W., Mughal, M.N., Fang, R., Zhou, Y., Shen, B. and Zhao, J. 2017. Genetic diversity and drug sensitivity studies on Eimeria tenella field isolates from Hubei Province of China. Parasit. Vectors. 10, 137–146. Taubert, A., Behrendt, J.H., Sühwold, A., Zahner H. and Hermosilla, C. 2009. Monocyte- and macrophage-mediated immune reactions against Eimeria bovis. Vet. Parasitol. 164(2–4), 141–153. Zulpo, D.L., Peretti, J., Ono, L.M., Longhi, E., Oliveira, M.R., Guimarães, I.G., Headley, S.A. and Guimarães, J.S. 2007. Pathogenicity and histopathological observations of commercial broiler chicks experimentally infected with isolates of Eimeria tenella, E. acervulina, and E. maxima. Semina (Londrina). Ciências Agrárias. 28, 97–104. | ||
| How to Cite this Article |
| Pubmed Style Jawad TI, Jasim GA. Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, Iraq. Open Vet. J.. 2025; 15(2): 714-723. doi:10.5455/OVJ.2025.v15.i2.20 Web Style Jawad TI, Jasim GA. Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, Iraq. https://www.openveterinaryjournal.com/?mno=226129 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.20 AMA (American Medical Association) Style Jawad TI, Jasim GA. Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, Iraq. Open Vet. J.. 2025; 15(2): 714-723. doi:10.5455/OVJ.2025.v15.i2.20 Vancouver/ICMJE Style Jawad TI, Jasim GA. Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, Iraq. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 714-723. doi:10.5455/OVJ.2025.v15.i2.20 Harvard Style Jawad, T. I. & Jasim, . G. A. (2025) Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, Iraq. Open Vet. J., 15 (2), 714-723. doi:10.5455/OVJ.2025.v15.i2.20 Turabian Style Jawad, Thanaa Ismael, and Ghaidaa Abass Jasim. 2025. Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, Iraq. Open Veterinary Journal, 15 (2), 714-723. doi:10.5455/OVJ.2025.v15.i2.20 Chicago Style Jawad, Thanaa Ismael, and Ghaidaa Abass Jasim. "Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, Iraq." Open Veterinary Journal 15 (2025), 714-723. doi:10.5455/OVJ.2025.v15.i2.20 MLA (The Modern Language Association) Style Jawad, Thanaa Ismael, and Ghaidaa Abass Jasim. "Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, Iraq." Open Veterinary Journal 15.2 (2025), 714-723. Print. doi:10.5455/OVJ.2025.v15.i2.20 APA (American Psychological Association) Style Jawad, T. I. & Jasim, . G. A. (2025) Molecular detection and histopathological changes caused by Eimeria tenella in broilers in Al-Diwaniyah Province, Iraq. Open Veterinary Journal, 15 (2), 714-723. doi:10.5455/OVJ.2025.v15.i2.20 |