| Research Article | ||
Open Vet. J.. 2025; 15(2): 738-745 Open Veterinary Journal, (2025), Vol. 15(2): 748-745 Research Article Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus)Mohamed Aref1*, Noha Ali Abd-Almotaleb2, Eman H. Elsheikh3, Gamal A. Salem4*, Mohamed A. Nassan5, Esraa Elsheikh11Department of Anatomy and Embryology, Faculty of Veterinary medicine, Zagazig University, Al-Sharqia, Egypt 2Department of Anatomy and Embryology, Faculty of Medicine, Zagazig University, Al-Sharqia, Egypt 3Department of Comparative Anatomy, Faculty of Science, Zagazig University, Al-Sharqia, Egypt 4Department of Pharmacology, Faculty of Veterinary medicine, Zagazig University, Al-Sharqia, Egypt 5Department of Clinical Laboratory Sciences, Turabah University College, Taif University, Taif, Saudi Arabia *Corresponding Author: Gamal A. Salem. Department of Pharmacology, Faculty of Veterinary Medicine, Zagazig University, El-Sharkia, Egypt. Email: gamal_vet_85 [at] yahoo.com; Submitted: 28/10/2024 Accepted: 29/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
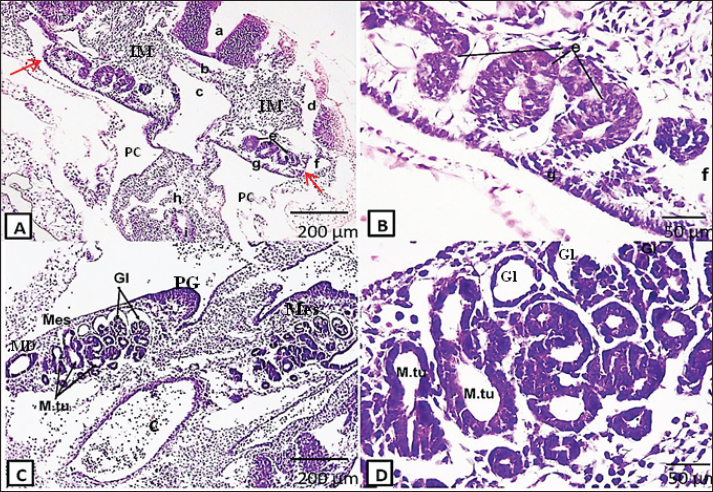
AbstractBackground: Rabbits are easily available and docile animals; thus, they are a promising model for medical research. White New Zealand rabbits are usually employed for research. Aim: This study aimed to determine the kidney’s histological structure using rabbit embryos and fetuses at various stages of gestation. Methods: Morphogenesis and histogenesis of the kidney were assessed using Harris’s hematoxylin and eosin staining at different gestational periods in rabbits. Results: Around the 10th day of gestation, the intermediate mesoderm started to differentiate into kidney tissue. The first primitive form of the kidney was the pronephros, which consisted of pronephric tubules and ducts. Large mesonephroi occupied a large part of the abdominal cavity from the 13th day of gestation and persisted for several days. At 15th day of gestation, the permanent kidney, the metanephros, began its differentiation and then persevered until formation of the adult kidney tissue at 24th day of gestation. Conclusion: This study clarified normal kidney histogenesis in rabbits; this would provide better understanding of kidney diseases aid in kidney transplant. Keywords: Embryo, Kidney, Mesonephros, Metanephros, Rabbit. IntroductionSeveral medical research fields use laboratory animal models in their experimental investigations. The wide use of animal models was attributed to the difficult application of these researches to human beings due to practical and ethical reasons. Rabbits are easily available and docile animals (Mapara et al., 2012); they are a promising model for medical research. White New Zealand rabbits, among other types, are frequently used in research projects. Compared with other breeds, these strains are less aggressive and have fewer health issues. Furthermore, rabbits are commonly used as animal models for a variety of human disorders because they are genetically closer to primates than rodents (Bosze and Houdebine, 2011; Sengupta and Dutta, 2020). One of the body’s most intricate organs is the mammalian kidney. The primary homeostatic organ required for maintaining general fluid balance, pH, and electrolyte management is the kidney. The kidney produces a number of hormones and humoral factors, including prostaglandins, calcitriol, erythropoietin, and renin, in addition to their excretory tasks (Santoro et al., 2015; Khoshdel Rad et al., 2020). The developing kidney systems are derived from the embryonic intermediate mesoderm in the form of three successive systems: pronephros, mesonephros, and metanephros. These developing nephric systems begin in the cervical region (pronephros) and are sequentially replaced in the thoracolumbar (mesonephros) and sacral regions (metanephros) due to changes in the position of the developing surrounding structures (Selman et al., 2016; Abbas et al., 2023). The pronephros is the most primitive, rudimentary, and nonfunctional set of the kidney. It appears as clusters of cells in the cervical region (in the form of pronephric tubules connected to the pronephric duct). The pronephric tubules degenerate after a short period, whereas most parts of the pronephric ducts persist as mesonephric ducts. Mesonephros is the second system of the kidney that develops caudally to the pronephros and appears as a large, elongated excretory organ. It consists of a number of glomeruli with mesonephric tubules, which degenerate also (McGeady et al., 2017). The earliest form of permanent kidneys is the metanephros. It arises early in embryogenesis from two sources: the metanephric mass of mesenchyme, also known as the metanephric diverticulum or blastema, and the ureteric bud. The mesonephric duct persists as a Wolffian duct and gives rise to the ureteric bud, which develops into the kidney duct system. The blastema gives rise to the nephron (Sadler, 2012). The kidneys of rabbits consist of the cortex and medulla. The cortex is the outer part of the kidney that is stained darker, while the inner part, called the medulla, is stained lighter. Externally, the cortex is enclosed by a dense irregular connective tissue called the renal capsule. The higher magnification of the cortex illustrates the proximal and distal tubules, glomerulus, and medullary rays. The medullary rays consist of the nephron, blood vessels, and collecting tubules connected to the medulla by the collecting duct. The nephron consists of renal tubules and renal corpuscles (part of them is the glomerulus). The afferent arteriole, Bowman’s capsule, glomerulus, and proximal and distal convoluted tubules were observed at higher magnification of the nephron (Farhana and On, 2019). Here, we provide sufficient data on kidney development in rabbits. The improved knowledge of the normal development of the kidney in rabbits would benefit renal disease research, kidney teratology, and kidney transplantation. Material and MethodsAnimal selection and experimental proceduresTen pregnant New Zealand female rabbits (weighing approximately 2–4 kg) that were gathered from the farms of Zagazig University’s faculties of agriculture and veterinary medicine were used in the current study. The rabbits were 1.5–3 years old. Twenty embryos and fetuses ranging in gestational age from the 10th to 25th days (10th, 13th, 15th, 18th, 20th, 24th, and 25th days) were utilized to study kidney histogenesis and development. An intramuscular injection of five milligrams of xylazine (Xylaject, 20 mg/ml, ADWIA, Egypt) and 35 mg/kg of ketamine Hcl (KETALAR, 100 mg/ml, Pfizer, NY) was used to anesthetize the pregnant rabbits, according to Enoka (2013) and Pandian (2020). The embryos and fetuses that were retrieved were preserved using either Bruin’s solution (which was followed by 70% ethyl alcohol) for 6–8 hours or 10% neutral-buffered formalin for 24–72 hours. Dehydration, clearing, embedding, blocking, cutting, and staining with Harris’ H&E stain were all applied to all embryos and fetuses. A normal light microscope (Objectives Xs 4, 10, 40) was used to examine the stained sections, and a Moticam X3 plus camera, a small Wi-Fi camera with a resolution of four megapixels, was used to take photographs. The histological technique was performed, according to Drury and Wallington (1980); Berg et al. (2011); Nomina Anatomica Veterinaria (2017); Nomina Embryologica Veterinaria 2nd Edn. (2017); and Nomina Histologica Veterinaria 1st Edn. (2017) embraced the nomenclature used in this work. Ethical approvalThe Zagazig University Institutional Animal Care and Use Committee examined the research procedures (No, ZU-IACUC/2/F/198/2024). Every surgical procedure was performed while the animal was under general anesthesia, and every attempt was made to reduce its suffering. ResultsThe findings showed that kidney development in rabbits starts early, on the 10th day of gestation. The principal phase (pronephros) during kidney development arises from the ventral region of the intermediate mesoderm as a mesenchymal cell population called the nephric cords. These cords formed the pronephric tubules and duct, and the pronephric duct was connected with the coelomic cavity by the nephrostome (Fig. 1a and b).
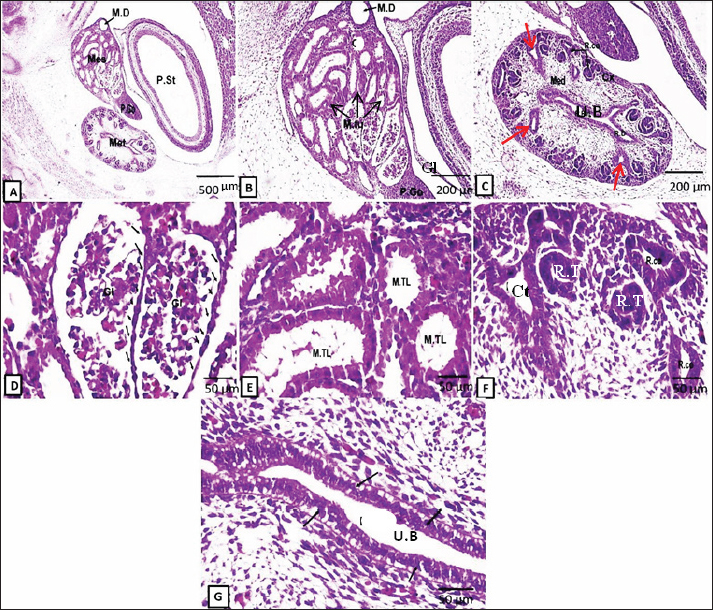
Fig. 1. Representative photomicrographs of H&E-stained sections at the D10 of gestation (A, B) and D13 (C-D): (A) (D10: Scale bar 200 μm, ×~10): primitive neural tube (a), primitive notochord (b), primitive dorsal aorta (c), primitive cardinal vein (d), primitive pronephric tubules (e), primitive pronephric duct (f), genital ridge (g), dorsal mesentery (h), primitive gut (i), intermediate mesoderm (IM), primitive coelomic cavity (PC), nephrostome (red arrows). (B) (D 10: -Scale bar 50 μm, ×~40): higher magnifications of the primitive pronephric tubules (e), primitive pronephric duct (f), and genital ridge (g). (C) (D13: Scale bar 200 μm, ×~10): showing the mesonephros (Mes) occupying a large area of the embryonic abdominal cavity containing glomeruli (Gl) at different stages of development, mesonephric tubules (M.tu), mesonephric duct (MD), primitive gonad (PG), and dorsal aorta (c). (D) (D 13: Scale bar 50 μm, ×~ 40): higher magnification of mesonephros containing glomeruli (Gl) surrounded by capsule lined by a single layer of low-cuboidal epithelium and mesonephric tubules lined by high-cuboidal epithelium. At the 13th day of gestation, cross-section of the embryo at this stage showed that the mesonephros (two, large and oval in shape) occupied a great part of the abdominal cavity, with the dorsal aorta in-between (Fig. 1c). The mesonephros at this embryonic stage consisted of the mesonephric duct, mesonephric tubules, and a tuft of blood capillaries called the glomeruli (derived from the dorsal aorta). Both mesonephric tubules and duct are lined with cuboidal epithelium. The mesonephros did not show well-differentiated cortex and medulla at this age. The mesonephric tubules were divided into proximal blind and distal parts. The proximal parts of the tubules formed the future Bowman’s capsule, which enclosed the glomeruli: forming the renal corpuscles. At this stage, the capsule is lined with low cuboidal cells. The vascular mesenchymal tissue filled the intertubular spaces (Fig. 1d). On the 15th day of gestation, both large, well-developed mesonephroi and undifferentiated metanephros appeared (Fig. 2a–g). At this stage, the mesonephric glomerulus is surrounded by a capsule lined by low-cuboidal epithelium. The cuboidal epithelium lined the mesonephric tubules, and some of the cells began to deteriorate. (Fig. 2d,e). The primitive metanephros consisted of the outer metanephrogenic mass derived from the intermediate mesoderm. The branched ureteric bud (future renal pelvis) was encircled by this mass. In addition to the formation of the renal cortex, the metanephrogenic mass initiates the formation of many solid and canalized renal tubules or future nephrons. The ureteric bud occupying the medulla was lined with simple columnar epithelium and received the collecting tubules (Fig. 2c,g).
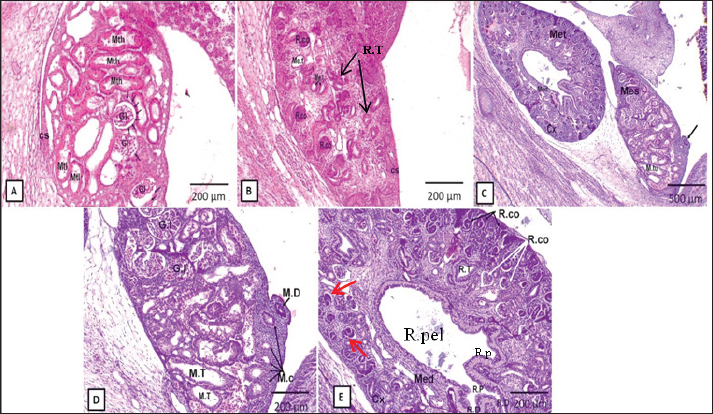
Fig. 2. Representative photomicrographs of H&E-stained sections at the D15 of embryonic life (A.G): (A) (Scale bar 500 μm, ×~4): mesonephros (Mes) and metanephros (Met) together at abdominal cavity, mesonephric duct (M.D), primitive gonad (P.Go), and primitive stomach (P.St). (B) (Scale bar 200 μm, ×~10): higher magnification of primitive mesonephros containing glomeruli (Gl), mesonephric tubules (M.tu), mesonephric duct (M.D), and primitive gonad (P.Go). (C) (Scale bar 200 μm, ×~10): showing higher magnification of primitive metanephros as outer cortex (Cx), inner medulla (Med), renal corpuscles (R.Co), renal duct (R.D), collecting tubules (red arrows), and ureteric bud (UB). (D) (Scale bar 50 μm, ×~40): higher magnification of mesonephros glomeruli (Gl) surrounded by capsule lined by low cuboidal epithelium (black arrows). (E) (Scale bar 50 μm, ×~40): higher magnification of mesonephric tubules (M.Tl) lined by a single layer of low cuboidal epithelium. (F) (Scale bar 50 μm, ×~40): higher magnifications of renal corpuscles (R.co), renal tubules (R.T), and collecting tubules (Ct). (G) (Scale bar 50 μm, X40): higher magnification of the ureteric bud (U.B) (primitive renal pelvis) lined by the S. columnar epithelium. At the 18th–20th days of fetal life, the rabbit metanephros was well differentiated compared with the previous stages, besides the late stages of the mesonephros (Fig. 3). The mesonephros could still be detected at this stage and showed degeneration and decreased size in some mesonephric tubules and glomeruli. The parenchyma of the metanephros was divided into the inner medulla and outer cortex in the same previous stage (Fig. 3c and e). The developing cortex consisted of renal tubules (the developing nephrons), with more differentiated tubules observed at the corticomedullary junction and newly formed tubules appearing at the external portion of the cortex. The Bowman’s capsule was created when one end of these tubules developed into a funnel shape. The glomeruli comprising the renal corpuscles were contained in this capsule (Fig. 3e).
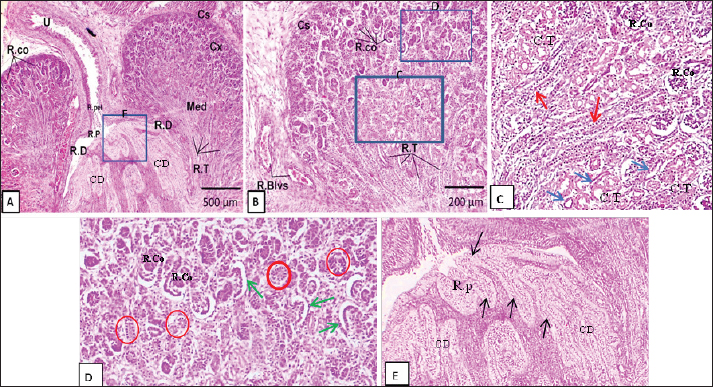
Fig. 3. Representative photomicrographs of H&E-stained sections at the D18 of rabbit fetal life (A, B) and D20 (C.E): (A) (D18: Scale bar 200 μm, ×~10): mesonephros contain glomeruli of mesonephros (Gl), mesonephric tubules (lower part) (Mtl), mesonephric tubules (higher part) (Mth) and capsule (Cs) formed from mesenchymal tissue. (B) (D18: Scale bar 200 μm, ×~10): metanephros, containing renal corpuscles (R.Co), metanephric tubules (Me.t), and capsule (Cs). (C) (D20: Scale bar 500 μm, ×~4): primitive mesonephros (Mes) and metanephros (Met); mesonephric tubules (M.tu); mesonephric duct (black arrow); metanephros divided into outer cortex (Cx) and inner medulla (Med). (D) (D20: Scale bar 200 μm, ×~10): magnification of mesonephroi (Gl), mesonephric tubules (M.T), mesonephric duct (M.D) that appear cranially and raw of mesonephric corpuscles that appear caudally (M.C). (E) (D20: Scale bar 200 μm, ×~10): magnification of metanephros divided as the outer cortex (Cx), inner medulla (Med), renal corpuscles (R.co) as new/outer (black arrows) and old/inner (white arrows), Bowman’s capsule (red arrows), renal tubules (R.T), renal duct (R.D), renal pelvis (R.pel), and renal papilla (R.p). At the 24th–25th days of fetal life, the rabbit’s metanephros (Fig. 4b) demonstrated a distinct separation of the cortex and medulla. Nephrons were localized and abundant in the cortex at the corticomedullary junction and its outer regions. There were various newly formed renal tubules of different widths and wall thicknesses at the outer part of the cortex. The renal tubules at the corticomedullary junction were differentiated into mature nephrons. The latter consisted of renal corpuscles, a loop of Henle, and proximal and distal tubules convoluted and straight (Fig . 4c). Bowman’s capsule had a funnel-shaped form, with cuboidal epithelium lining the inner visceral layer and S. squamous epithelium lining the outer parietal layer (Fig. 4d). The collecting tubules in the cortex region were small and lined by simple low columnar epithelium (Fig. 4c).
Fig. 4. Representative photomicrographs of H&E-stained sections from rabbit kidney tissue (A, B) and D24-25 showing: (A) (D18: Scale bar 500 μm, ×~4): showing metanephros divied as outer corted (Cx), inner medulla (Med), capsule (Cs), renal corpuscles (R.Co), renal tubules (R.T), renal duct (R.D), renal papilla (R.p), renal pelvis (R.pel), collecting ducts (CD) and ureter (U). (B) (Scale bar 200 μm, ×~10): showing magnification of metanephros contain renal corpuscles (R.Co), renal tubules (R.T), capsule (Cs), renal blood vessels (R.Blvs). (C) (at cortico-medullary junction, magnification of B, ×~10): Showing renal corpuscles (R.Co), convoluted tubules (C.T), collecting tubules (Red arrows) lined by columnar epithelium, loop of Henle (blue arrows) lined by S. squamous epithelium. (D) (at the cortex, magnification of B, ×~10): showing renal corpuscles (R.Co), newly formed renal tubules (Red circles), Bowman’s capsule (green arrows) was funnel-shaped with outer parietal layer lined by S. squamous edpithelium and inner visceral layer lined by cuoidal epithelium. (E) (Magnification of A. ×~10) showing: the renal medulla consisting of collecting ducts (CD) and the renal papilla (R.p) lined by columnar epithelium (black arrows). The outer part of the developing cortex was surrounded by undifferentiated mesenchymal tissue that would share in kidney capsule development (Fig. 4b). The collecting tubules in the developing medulla were encircled by dense, undifferentiated mesenchymal tissue; these tubules were broader in lumen and larger in size than the collecting tubules in the cortex. The collecting tubules were lined with columnar epithelium containing central nuclei. The renal papillae are united to form a single renal papilla (unipapillary rabbit kidney). The renal papilla was also lined with columnar epithelium (Fig. 4e). The renal pelvis began demarcating and consisted of the renal ducts, ureters, and renal blood vessels (Fig. 4a). DiscussionThe present study clarified kidney histogenesis at different gestational periods. This approach would help improve our understanding of kidney diseases and experimental teratology. Similarly, in a previous study on mice, the kidneys developed from the ventral part of the intermediate mesoderm and persisted along the caudal region of the embryo during gastrulation. The onset of kidney development was detected on the 10th day of rabbit gestation in this study. The first primitive kidney at this stage is called the pronephros (Nagalakshmi and Yu, 2015), except that the pronephros in mice is first detected at the 8.5th–10.5th embryonic days. In this study, the mesonephros was observed first at the 13th day of embryonic life. They appeared as large structures occupying a large part of the abdominal cavity. While Abbas and Rabie (2012) observed in rabbit mesonephros at the 14th day of embryonic life, Fayez et al. (2014) showed mesonephros for the first time at the 15th day of gestation. In an earlier study, normal physiological herniation of the developing intestinal loop was produced by the relatively large size of the liver and mesonephros (Noden and Lahunta, 1987). In similar findings, the mesonephros at this stage showed mesonephric tubules lined by cuboidal epithelium, a clump of blood capillaries known as a glomerulus that invaded some tubules, eventually forming the renal corpuscles (Fayez et al., 2014). In other similar studies, the development of the definitive and permanent rabbit kidney, the metanephros, developed from the interaction between the metanephric mesenchyme (nephrogenic ducts) and the ureteric bud (mesonephric duct). The metanephros was first detected in this study on the 15th day of rabbit gestation. At this stage, ureteric bud branching is detected, and nephron induction occurs at the periphery of the kidney: the onset of renal cortex formation. Simple columnar epithelium with a round or oval nucleus lines the ureteric bud (Fayez et al., 2014) in rabbits. In contrast, rabbits showed the onset of metanephros appearance on the 12th day of gestation (Gaber, 2017), whereas metanephros formation around the E15.5th day of gestation (Nagalakshmi and Yu, 2015) in mice, while the beginning of metanephros differentiation at the 5th week of gestation (Rosenblum, 2008; Costantini, 2010) in humans. Moreover, in human, noted the first ureteric bud branching and renal pelvis formation as well as the cranial and caudal lobes of the kidney were observed at the 6th week of development (Upadhyay and Silverstein, 2014). The metanephros kidney is located next to the advanced stages of the degenerated mesonephros on the 20th day of gestation. The glomeruli shrank in size and the mesonephric tubules showed signs of moderate degeneration. However, Fayez et al. (2014), in rabbits, showed these outcomes at the 19th day of gestation. However, mouse mesonephroi started to degrade at day 14.5 and the mesonephric tubules completely underwent apoptosis before disappearing at day 15.5 (Sainio et al., 1997). In a previous study, the inner medulla and outer cortex of the metanephros could separate during the 18th–20th days of gestation. The metanephrogenic mesoderm initiates the nephron-forming stage within the developing cortex. The newly formed renal tubules were of different shapes and sizes and were in the form of solid, canalized, round, or oval renal vesicles. Some of these vesicles become elongated tubules; one end of these tubules became funnel-shaped and surrounded the glomerulus forming the renal corpuscle (share in the formation of future mature nephrons). Some tubules undergo canalization to form convoluted and straight tubules and a loop of Henle (Nagata, 2012; Fayez et al., 2014). In other earlier studies, on days 24 and 25, the freshly created nephrons were concentrated in the outer region of the cortex, whereas the corticomedullary region contained almost mature old nephrons. The medulla at this stage was well distinguished from the cortex, and the collecting ducts (excretory ducts of the kidney) were well differentiated. A single layer of columnar epithelium lined the collecting ducts (Fayez et al., 2014). On the other hand, collecting ducts in mice became apparent on the 28th day of gestation (Dickinson et al., 2005). Additionally, studies on domestic animals showed that the epithelial cells of collecting tubules were columnar near the renal papilla and cuboidal toward the distal portions of the tubules (Dellmann and Brown, 1976). In previous studies, the papillae were fully united at this point in development, creating a common renal papilla or rabbit unipapillary kidney. The papilla received the collecting ducts and was lined by columnar epithelium (Abbas and Rabie, 2012; Gaber, 2017) in rabbits. In similar findings, nephrogenesis was an extremely intricate process that resulted in the development of nephrons, which were the kidney’s functional units. The renal cortex of the kidney comprises the nephrons, which filter blood from wastes such as uric acid, water, and other ions. The renal vesicles, which were the antecedents of the nephrons, were created by the conversion of mesenchyme to epithelium (Shah et al., 2004; Hsu, 2021; Allegaert and Iacobelli, 2022). Furthermore, in humans, nephrogenesis is completed by the 36th gestational week, and urine production begins at the 10th week of gestation (Zandi-Nejad et al., 2006). In a similar previous study, the mature form of the rabbit nephrons consisted of numerous renal corpuscles surrounded by the renal convoluted tubules and loop of Henle. Cuboidal epithelium lined the convoluted tubules. S. squamous epithelium lined the loop of Henle. Each renal corpuscle consists of a glomerulus encircled by the parietal and visceral layers of Bowman’s capsule. The urine space separated the simple squamous epithelium that lined the parietal layer of this capsule from the cuboidal epithelium that lined the visceral layer (Al-Kelaby et al., 2018; Elsebay et al., 2022) in rabbits and (Bacha and Bacha, 2012; Eurell and Frappier, 2013) domestic animals. The same results were in accordance with Gaber (2017) in rabbits, except that the simple columnar epithelium lined the renal tubules, and the visceral layer of the Bowman’s capsule was observed to have a low columnar epithelial layer. ConclusionThe goal of the current study was to illustrate kidney morphogenesis and histogenesis in rabbits during fetal development. The development of the kidney in rabbits begins at the early stages of gestation. The first rudimentary form of the kidney, the pronephros, appeared on the 10th day of gestation. The mesonephros, the second transitory phase, persisted for several days and formed part of the urinary and genital systems. Beginning on the 15th day of gestation, the metanephros, the permanent kidney, continued until the 25th day, when adult kidney tissue began to form. AcknowledgmentsThe authors express their gratitude to the staff members of the Anatomy and Embryology Department, Faculty of Veterinary Medicine at Zagazig University, Egypt, for their support during our work. Conflict of interestThe authors confirm that they do not have any conflicts of interest to declare. FundingThe authors did not receive any external funding for this study. Authors’ ContributionsMA, EE, N.A, E.E, and MAN: Conceptualized the study, executed all experiments, analyzed the data, and participated in writing the manuscript. MA, GAS: Supervised the study, validated the results, abstracted the work, and reviewed and edited the manuscript. All authors have read, reviewed, and approved the final manuscript. ReferencesAbbas, J.G. and Rabie, F. 2012. Histomorphological study of tubular system and collecting tubules in domestic rabbit’s fetuses (Oryctolagus Cuniculus). Kufa. J. Vet. Med. Sci. 3(1), 67–76. Abbas, G., Rehman, A.U., Sarwar, N., Fatima, Z., Hussain, S., Ahmed, M., Raza, M.A., Kan, M., Doğan, H., Khan, M.A. and Ahmad, S. 2023. Deciding sowing-window for maize-based cropping system in arid and semiaridenvironments in Punjab, Pakistan. Tur. J. Agri. Fore. 47, 1078–1098. Al-Kelaby, W.J.A., Almhanna, H., Hussein, H.J., Abdulridha, W.M. and Al-Shaibani, S. 2018. Histological and biochemical evaluation of the efficiency of rabbit kidney after partial or radical nephrectomy. Biochem. Cell. Arch. 18, 2033–2042. Allegaert, K. and Iacobelli, S. 2022. Editorial: the developing kidney: perinatal aspects and relevance throughout life. Front. Pediatr. 10, 990854. Bacha, W.J. and Bacha, L.M. 2012. Color atlas of veterinary histology. 662. Wiley Blackwell. UK. Berg, D., Malinowsky, K., Reischauer, B., Wolff, C. and Becker, K.F. 2011. Use of formalin-fixed and paraffin-embedded tissues for diagnosis and therapy in routine clinical settings. Methods Mol. Biol. 785, 109–122. Bosze, Z. and Houdebine, L.M. 2011. Application of rabbits in biomedical research: a review. Wor. Rabbit Sci, 14, 1–14. Costantini F, K.R. 2010. Patterning a complex organ: branching morphogenesis and nephron segmentation in kidney development. Dev Cell. 18 (5), 698–712. Dellmann, H., Brown, E.1976. Textbook of Veterinary Histology. Textbook of veterinary histology. Lea & Febiger.Philadelphia. USA. Dickinson, H., Walker, D. W., Cullen-McEwen, L., Wintour, E. M., Moritz, K. 2005. The spiny mouse (Acomys cahirinus) completes nephrogenesis before birth. Am J Physiol Renal Physiol. 289(2), F273–9. Drury, R.A. and Wallington, E.A. 1980. Carleton’s histological technique. 5th ed. New York: Oxford University Press. Elsebay, S., Abdelnaby, A., Khalaf, G. and Abou-Rabia, N. 2022. Characterization of the decellularized male rabbit kidney as a three-dimensional natural scaffold for tissue engineering. A histological study. Egy. J. Hist. 45, 1146–1169. Enoka, B. 2013. Rabbit anaesthesia. SOP. 103(1), 1–7. Eurell, J.O. and Frappier, B.L. 2013. Dellmann’s textbook of veterinary histology [WWW Document]. John Wiley & Sons. Wiley Blackwell. UK. Farhana, S. and On, F.W. 2019. Anatomical and histological study of male uropoetic organs of common rabbit (Oryctolagus cuiculus). Proc. Int. Conf. Sci. Eng. 2, 149–152. Fayez, E., Sayed-Ahmed, A. and Abo-Ghanema. 2014. Morphogenesis of rabbit kidney pre-and postnatal. Alex. J. Vet. Sci. 41, 47. Gaber, W. 2017. Development of the rabbit (Oryctolagus cuniculus) metanephros. J. Vet. Anat. 10, 109. Hsu Y, C.T. 2021. The first thousand days: kidney health and beyond. Healthcare 9, 1332. Khoshdel Rad, N., Aghdami, N. and Moghadasali, R. 2020. Cellular and molecular mechanisms of kidney development: from the embryo to the kidney organoid. Front. Cell. Dev. Biol. 8, 183. Nagalakshmi, V.K. and Yu, J. 2015. The ureteric bud epithelium: morphogenesis and roles in metanephric kidney patterning. Mol Reprod Dev. 82(3), 151–66. Nagata, M. 2012. Glomerulogenesis and the role of endothelium. Curr. Opin. Nephrol. Hypertens. 27(3), 159–164. Mapara, M., Thomas, B. and Journal, K.B.-D. 2012. Rabbit as an animal model for experimental research. Dent. Res. J. 9(1), 111–118. McGeady, T., Quinn, P., Fitzpatrick, E. and Ryan, M. 2017. Veterinary embryology. 2nd ed. Pondicherry, India. Noden, D. and Lahunta, A. 1987. The embryology of domestic animals. Developmental mechanisms and malformations. Quar. Rev. Biol. 62, 85. Nomina Anatomica Veterinaria. 2017. Prepared by the International Committee on Veterinary Gross Anatomical Nomenclature and Authorized by the General Assembly of the World Association of Veterinary Anatomists. Hanover (Germany), Ghent (Belgium), Columbia, MO (USA), Rio de Janeiro. International Committee on Veterinary Gross Anatomical Nomenclature. Nomina Embryologica Veterinaria 2nd Edn. 2017. Prepared by the International Committee on Veterinary Embryological Nomenclature and Authorized by the General Assembly of the World Association of Veterinary Anatomists . Knoxville, U.S.A, pp: 11–12. Nomina Histologica Veterinaria 1st Edn. 2017. Prepared by the International Committee on Veterinary Histological Nomenclature and Authorized by the General Assembly of the World Association of Veterinary Anatomists. Cornell University, pp: 10–31. Pandian, S. 2020. Ketamine-xylazine anesthesia in rabbits. Ind. Vet. J. 82, 388–389. Rosenblum, N.D. 2008. Developmental biology of the human kidney. Semin. Fetal. Neonatal. Med. 13, 125–132. Sadler, T. 2012. Langman’s Medical Embryology. 12th ed. Philadelphia: Lippincott Williams & Wilkins, Baltimore. Sainio, K., Hellstedt, P., Kreidberg, J. A., Saxén, L., Sariola, H. 1997. Differential regulation of two sets of mesonephric tubules by WT-1. Devel. 124(7), 1293–1299. Santoro, D., Caccamo, D., Lucisano, S., Buemi, M., Sebekova, K., Teta, D. and De Nicola, L. 2015. Interplay of vitamin D, erythropoiesis, and the renin-angiotensin system. Biomed. Res. Int. 2015(1), 145828. Selman, M.O., Al-Bakri, N. and Al-Salmany, K.A.M. 2016. Histogenesis and histomorphometric study on kidney disorder by lamortigine in postnatal. Rat. Wor. J. Pharm. Res. 5(11), 190–204. Sengupta, P. and Dutta, S. 2020. Mapping the age of laboratory rabbit strains to human. Int. J. Prev. Med. 11, 194. Shah, M., Sampogna, R., Sakurai, H. and Bush, K. 2004. Branching morphogenesis and kidney disease. Development 131(7), 1449–1462. Upadhyay, K.K. and Silverstein, D.M. 2014.Renal development: a complex process dependent on inductive interaction.Curr Pediatr Rev. 10(2), 107–14. Zandi-Nejad, K., Luyckx, V. A., Brenner, B. M. 2006.Adult hypertension and kidney disease: the role of fetal programming. Hypertension. 47(3), 502–8. | ||
| How to Cite this Article |
| Pubmed Style Aref M, Abd-almotaleb NA, Elsheikh EH, Salem GA, Nassan MA, Elsheikh E. Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus). Open Vet. J.. 2025; 15(2): 738-745. doi:10.5455/OVJ.2025.v15.i2.23 Web Style Aref M, Abd-almotaleb NA, Elsheikh EH, Salem GA, Nassan MA, Elsheikh E. Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus). https://www.openveterinaryjournal.com/?mno=226464 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.23 AMA (American Medical Association) Style Aref M, Abd-almotaleb NA, Elsheikh EH, Salem GA, Nassan MA, Elsheikh E. Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus). Open Vet. J.. 2025; 15(2): 738-745. doi:10.5455/OVJ.2025.v15.i2.23 Vancouver/ICMJE Style Aref M, Abd-almotaleb NA, Elsheikh EH, Salem GA, Nassan MA, Elsheikh E. Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus). Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 738-745. doi:10.5455/OVJ.2025.v15.i2.23 Harvard Style Aref, M., Abd-almotaleb, . N. A., Elsheikh, . E. H., Salem, . G. A., Nassan, . M. A. & Elsheikh, . E. (2025) Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus). Open Vet. J., 15 (2), 738-745. doi:10.5455/OVJ.2025.v15.i2.23 Turabian Style Aref, Mohamed, Noha Ali Abd-almotaleb, Eman H. Elsheikh, Gamal A. Salem, Mohamed A. Nassan, and Esraa Elsheikh. 2025. Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus). Open Veterinary Journal, 15 (2), 738-745. doi:10.5455/OVJ.2025.v15.i2.23 Chicago Style Aref, Mohamed, Noha Ali Abd-almotaleb, Eman H. Elsheikh, Gamal A. Salem, Mohamed A. Nassan, and Esraa Elsheikh. "Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus)." Open Veterinary Journal 15 (2025), 738-745. doi:10.5455/OVJ.2025.v15.i2.23 MLA (The Modern Language Association) Style Aref, Mohamed, Noha Ali Abd-almotaleb, Eman H. Elsheikh, Gamal A. Salem, Mohamed A. Nassan, and Esraa Elsheikh. "Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus)." Open Veterinary Journal 15.2 (2025), 738-745. Print. doi:10.5455/OVJ.2025.v15.i2.23 APA (American Psychological Association) Style Aref, M., Abd-almotaleb, . N. A., Elsheikh, . E. H., Salem, . G. A., Nassan, . M. A. & Elsheikh, . E. (2025) Prenatal morphogenic and histogenic development of the kidney in rabbits (Oryctolagus cuniculus). Open Veterinary Journal, 15 (2), 738-745. doi:10.5455/OVJ.2025.v15.i2.23 |