| Research Article | ||
Open Vet. J.. 2025; 15(2): 820-826 Open Veterinary Journal, (2025), Vol. 15(2): 820-826 Research Article Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomographyMohamed Aref1*, Walaa O. M. Youssef2, Ahmed Abdelbaset-Ismail2, Gamal A. Salem3*, Mohamed A. Nassan4 and Esraa Elsheikh11Department of Anatomy and embryology, Faculty of Veterinary Medicine, Zagazig University, Al-Sharqia, Egypt 2Department of Surgery, Anesthesiology, and Radiology, Faculty of Veterinary Medicine, Zagazig University, Al-Sharqia, Egypt 3Department of Pharmacology, Faculty of Veterinary Medicine, Zagazig University, Al-Sharqia, Egypt 4Department of Clinical Laboratory Sciences, Turabah University College, Taif University, Taif, Saudi Arabia *Corresponding Author: Gamal A. Salem. Department of Pharmacology, Faculty of Veterinary Medicine, Zagazig University, El-Sharkia, Egypt. Email: gamal_vet_85 [at] yahoo.com Submitted: 30/10/2024 Accepted: 29/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
AbstractBackground: Cross-sectional imaging is set to become the standard method for diagnosing various pathological conditions of the urinary tract in goats, and the concept for this atlas originated from this understanding. Until now, there has not been a comprehensive comparative atlas detailing the structures of the urinary tract in goats and their relationships with other organs. Aim: The main aim and objective of this study was to compare anatomically frozen cross-sections and detailed computed tomography (CT) images of the urinary tract, which may be used to diagnose urinary abnormalities and affections in goats. Methods: Eight healthy adult goats (Capra hircus) of both sexes were collected for anatomical and diagnostic examinations; average age: 15–25 ± 0.52 months and average weight: 25–35 ± 0.42 kg. The study focused on the pelvic and abdominal cavities, along with their contents and relationship with urinary organs. The examination included the analysis of bony and soft tissues using median, sagittal, and cross-anatomical sections. Subsequently, the specimens were subjected to diagnostic screening by CT. Results: We found that both kidneys were readily distinguishable in both anatomical and CT scans as spherical to bean-shaped, but in the CT scan, the kidneys exhibited hypoechoic features with an anechoic hilus, and there was no sharp demarcation between the cortex and medulla, although the cortex was slightly denser than the medulla. In the dorsal-plane CT scan, the right ureter was positioned dorsally adjacent to the caudal vena cava, extending toward the urinary bladder. The egress of the left ureter from the left renal hilus was observed in the transverse CT image. On transverse- and dorsal plane CT, the urinary bladder may be identified at the sacrum level on the pelvic floor, located beneath the uterus and rectum. A dorsal-plane CT scan revealed the presence of the urethra exiting the urine bladder in a caudal direction. Conclusion: The cross-sectional layout of CT images enables both students and clinicians to observe the anatomical connections and features of the goat urinary tract, which might not be easily seen through dissection alone. Furthermore, this atlas could serve as a helpful resource for study and clinical reference to enhance the understanding of goat urinary tract anatomy, which can ultimately help address pathological conditions. Keywords: Frozen cross sections; CT; kidney; Urinary bladder; Goat. IntroductionThe use of goats as animal models for scientific research has increased significantly in recent years. Goats have metabolic and bone remodeling rates close to those of humans, and they are widely used for studies of infectious diseases, bone, cartilage, and ligaments, cardiovascular conditions, and urinary conditions (Fulton et al., 1994; Larsen, 2015; Stieger-Vanegas and McKenzie, 2021). The renal system comprises the kidney, ureter, and urethra. This system’s primary function was to filter 200 L of fluid per day from the renal blood flow, allowing the expulsion of toxins, metabolic waste products, and excess ions while maintaining vital substances in the blood. The primary role of the urinary system was the maintenance of water and electrolyte homeostasis and the excretion of many toxic metabolic waste products, predominantly the nitrogenous compounds urea and creatinine, from the body (Ogobuiro and Tuma, 2019; El Karmoty et al., 2022). The goats’ kidneys were smooth, bean-shaped, and retroperitoneal. The goats’ kidneys were in the sublumbar region, with the right kidney extending from the last rib to the third lumbar transverse process. The left kidney extended from the caudal border of the third to the fifth lumbar transverse process. The right kidney connected dorsally to the sub-lumbar muscles and cranially to the first rib, forming the renal impression with the liver’s right and caudate lobes. The ascending colon’s intestinal disc lodged the right kidney medially. The rumen connects craniomedially and caudally to the small intestine and the descending colon to the left kidney (Hussain, 2010; Ragab et al., 2010; El Karmoty et al., 2022). Different diagnostic imaging techniques, such as ultrasonography, radiography, and computed tomography (CT), play an important role in veterinary practice. The data obtained from diagnostic imaging were used as baseline data for educational, clinical, and research purposes (Alnahrawy et al., 2021). CT is a valuable modality providing standard live animal cross-sectional image review. CT has an advantage over radiography in eliminating superimposed images and eliminating the need for contrast materials. Ultrasound examination may be an invaluable tool in the diagnosis of some urinary abnormalities, such as polycystic kidney disease, important congenital nephropathy in goats (Steffen and Tontis, 1996), pyelonephritis (Craig et al., 2008), kidney tumors (Dobry and Danuser, 2009; Griffin et al., 2009), kidney trauma (Lee et al., 2007), and urinary tract disorders (Washburn et al., 2009). The main goal of this research was to identify the anatomical features of the urinary tract in goats and to determine their connections to other abdominal and pelvic organs. This was achieved by directly comparing the anatomical structures observed in cross-sections with those observed in CT imaging. Materials and MethodsAnimalsThe current investigation was conducted on a sample of eight goats, both male and female, with a weight range of 25–35 ± 0.42 kg and an age range of 15–25 ± 0.52 months. All goats underwent thorough clinical examination at the Department of Surgery, Anesthesiology, and Radiology, Faculty of Veterinary Medicine, Zagazig University. The female goats were those who were neither pregnant nor lactating after clinical examination at the Department of Theriogenology, Faculty of Veterinary Medicine, Zagazig University. CT imagingInitially, the goats underwent CT scanning after sedation via intravenous administration of Xylazine HCl 2% (0.1 mg/kg, Xyla-Ject®, ADWIA, Egypt). The CT scan was conducted at a privately owned CT center located in the Al-Sharkia Governorate, Egypt. The experimental setup utilized a third-generation multislice helical CT and 3D CT scanners, specifically, a GE Light Speed Ultra 8 system manufactured by General Electric Company in the United States. This scanner consists of eight body slices per rotation. In this study, the soft tissue window width was measured to be 500 HU (Hounsfield unit). The experimental setup consisted of a 53.2-kW generator, a tube size of 6.3 MHU, and a rotation time of 0.5 seconds. The abdominal region was subjected to a scanning procedure using a layer thickness of 5 mm intervals with an exposure of 120 kV and 270 mA. Contrast material was not used in the acquisition of CT images. The soft tissue window was used to assess the configuration, morphology, and uniformity of the urinary tract, utilizing the guidance of bony features. Following a 24-hours fast, the goats were administered sedatives and placed in a sternal recumbent position during the scanning phase. The CT scan was performed following the prescribed protocol outlined (Braun et al., 2011). Frozen cross-sectional techniqueThe identical goats were euthanized and thereafter placed in a deep freezer set at a temperature of −20°C at the Department of Anatomy and Embryology, Faculty of Veterinary Medicine, Zagazig University, while assuming a sternal recumbent position. After complete freezing of the cadavers, they were subsequently subjected to transverse sectioning with an estimated spacing of 3 cm between each section, commencing from the 12th thoracic vertebra level and extending to the sacrum. The successive transverse incisions were made at a right angle to the longitudinal axis of the goat trunk. The photography was conducted on each cross-section, and subsequently, a comparison was made between each photograph and the matching CT images at an equivalent level. The nomenclature used in this study was the following (Nomina Anatomica Veterinaria, 2017). Ethical approvalThe Zagazig University Ethical Committee for Animal Care and Use approved all techniques used in the current study, which followed the worldwide criteria outlined in the approved Institutional Animal Care and Use Committee (IACUC) protocol (ZUIACUC/2/F/197/2024). ResultsThe current study relied on the utilization of anatomical cross sections as a point of reference for the objective identification of structures within CT images at a consistent level. This study compared CT soft tissue windows in the transverse plane with serial frozen cross-slice images at each vertebra, spanning from the 12th thoracic vertebra to the sacrum. The identification of soft tissue structures on CT was based on their radiostatic density, morphology, and spatial distribution in both the cross-sectional and dorsal plane images. The thoracic and lumbar vertebrae, ribs, and sternum were used as anatomical landmarks to determine the positions of the organs under examination. The bony boundaries of the abdomen and pelvis were demonstrated by 3D CT scan (Fig. 1). Both kidneys were readily distinguishable based on their morphology and associations in both anatomical and CT examinations (Figs. 2, 4). In both the transverse and dorsal planes of the CT scan, the objects exhibited a spherical to bean-shaped morphology. On CT, both kidneys exhibited hypoechoic structures characterized by an anechoic hilus. A clear differentiation was observed between the cortex, medulla, and adjacent perirenal fat, particularly in the frozen sections. On CT, both kidneys appeared ovoid and sharply demarcated with homogenous renal parenchyma. There was no sharp demarcation between the cortex and medulla, although the cortex was slightly denser than the medulla. Figure 1 shows the visualization of the right kidney, located at the 12th thoracic vertebral level, specifically to the right of the median plane. The structure in question exhibited a dorsal relationship with the thoracic vertebrae, a lateral relationship with the ribs, a medical association with the dorsal ruminal sac, and an embedded location inside the renal impression of the caudate lobe of the liver. The left solitary kidney was observed at the second lumbar vertebral level, positioned to the right of the median plane (Fig. 2). The dorsal ruminal sac, as observed in the dorsal-plane CT scan (Fig. 4), resulted in the displacement of the left kidney toward the right side of the abdomen and the caudal migration toward the right kidney. The left kidney exhibited a dorsal relationship with the lumbar spine, a medial relationship with the dorsal ruminal sac, and a ventrolateral relationship with the intervening intestine. A dorsal-plane CT scan revealed the presence of the right renal vein, which extended from the right kidney toward the caudal vena cava (Fig. 4). Both transverse and dorsal plane CT scans revealed the presence of the right renal artery, which originates from the aorta and extends toward the right kidney (Figs. 2, 4). Following its entry into the renal hilus (Fig. 2), the structure underwent bifurcation into its dorsal and ventral branches. In the dorsal-plane CT scan, the right ureter was positioned dorsally adjacent to the caudal vena cava, extending toward the urinary bladder. The lumen was smaller in size compared with the renal vein (Fig. 4). The egress of the left ureter from the left renal hilus was observed in the transverse CT image (Fig. 2). On transverse- and dorsal-plane CT scans (Figs. 3, 4), the urinary bladder may be identified at the sacrum level on the pelvic floor, located beneath the uterus and rectum. A dorsal-plane CT scan revealed the presence of the urethra exiting the urine bladder in a caudal direction (Fig. 4). DiscussionThis study defined urinary tract CT in goats at a greater depth than another study. The CT scans provided precise data regarding the shape, position, and relationships of the studied organs. The images were recognized based on the anatomical slices without the use of landmark identification (Shojaei et al., 2006). In an earlier study, both kidneys were observed in the dorsal plane and on cross-sectional CT imaging. By using anatomically frozen cross sections, the skeletal structure from the 12th thoracic vertebra on the right side to the second lumbar vertebra (Dyce et al., 2009) in ruminants (Steininger and Tierheilkd, 2012) and goats can be observed.
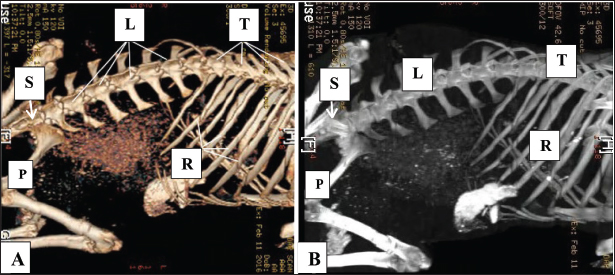
Fig. 1. Normal 3D CT scans of the abdomen and pelvis in healthy goats (Panels A, B) show the thoracic vertebrae (T), lumber vertebrae (L), ribs (R), pelvic bone (P), and sacrum (S).
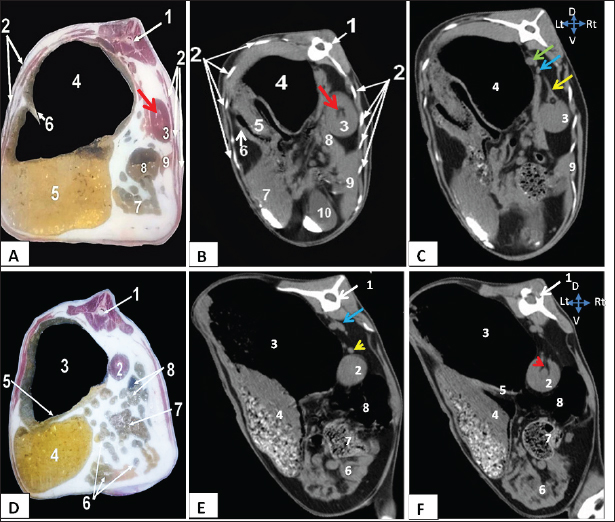
Fig. 2. Representative macrographic images of cross-sectional anatomy (Panel A-C) and CT image (5-mm thickness) (Panel B-D) of the goat abdomen. Panel A: 1- Twelfth thoracic vertebra; 2- Costal bony segments; 3- Right kidney; 4- Dorsal ruminal sac; 5- Ventral ruminal sac; 6- Right longitudinal ruminal pillar; 7- Jejunum; 8- Cecum; 9- Right lobe of liver; Red arrow: Renal hilus. Panel (B-C): 1- Twelfth thoracic vertebra; 2- Costal bony segments; 3- Right kidney; 4- Dorsal ruminal sac; 5- Ventral ruminal sac; 6- Greater omentum; 7- Abomasum; 8- Caudate lobe of liver; 9- Right lobe of liver; 10- Gall bladder; Red arrow: Renal hilus; yellow arrow: Right renal artery; Green arrow: Caudal vena cava; Blue arrow: Aorta. Panels (D-F): 1- Second lumbar vertebra; 2- Left kidney; 3- Dorsal ruminal sac; 4- Ventral ruminal sac; 5- Left longitudinal ruminal pillar; 6- Jejunum; 7- Cecum; 8- Colon; Blue arrow: Aorta; Yellow arrowhead: left renal artery; Red arrowhead: Left ureter. The findings of our study indicate that the right kidney spanned the thoracic vertebrae 12th to 13th, whereas the left kidney was situated between the second and third lumbar vertebrae. In goats, the left kidney was positioned between the first and fifth lumbar vertebrae, whereas the right kidney was located between the 11th and 4th lumbar vertebrae, contingent upon the level of rumen fullness with ingesta (Braun et al., 2011). Furthermore, it has been reported that in goats, the right kidney maintains a consistent position, ventrally to the final thoracic vertebra as well as the first and second lumber vertebrae. Additionally, the left kidney was positioned inferior to the right kidney and ventrally to the second, third, and fourth lumbar vertebrae (Ragab et al., 2010). In a previous study on Jebeer, the right kidney was observed at the level of the final thoracic to second lumbar vertebrae, whereas the left kidney was found to stretch from the second to the fourth lumbar vertebrae (Sajjadian et al., 2015). In previous studies, the optimal positioning of the right kidney in goats was shown to be near the ribs, thoracic vertebrae, lateral abdominal wall, and caudate lobe of the liver. The left kidney was associated with the lumbar vertebrae, and it was visually distinguished from the lateral abdominal wall by the presence of a gas-filled intestine. The distal portion of the left kidney was observed in the right flank region posterior to the right kidney. The observed phenomenon can be attributed to the medial displacement of the left kidney caused by the gas-filled dorsal ruminal sac (Alsafy et al., 2013). The results of this study were consistent with earlier findings reported by (Steininger and Tierheilkd, 2012), as well as (Braun et al., 2011) in goats.
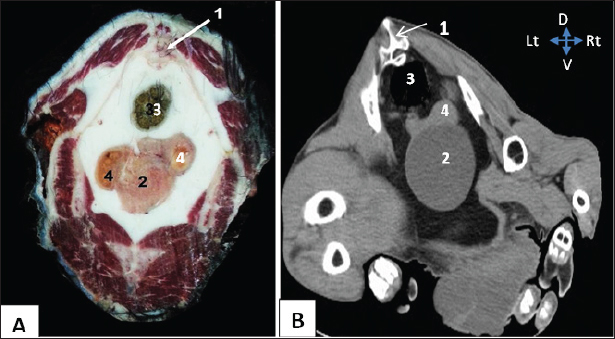
Fig. 3. Representative macrographic image of cross-sectional anatomy (Panel A) and computed tomography image (5-mm thickness) (Panel B) of the goat pelvis. Panel (A –B): 1- Sacrum; 2-Urinary bladder; 3- Rectum; 4-Uterus.
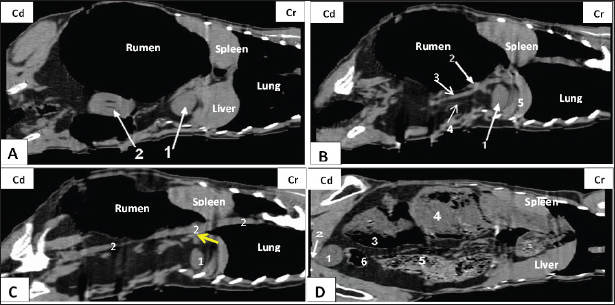
Fig. 4. Representative computed tomography image of the goat trunk in the dorsal plane. Panel (A): 1-Right kidney; 2-Left kidney. Panel (B): 1-Right kidney; 2-Right renal vein; 3-Caudal vena cava; 4-Right ureter; and 5- caudate lobe of the liver. Panel (C): 1-Right kidney; 2-Aorta. Panel (D): (1) urinary bladder; (2) urethra; (3) intra-ruminal gases; (4) rumen; (5) intestinal; (6) colon. Based on the echogenicity analysis, the kidneys exhibited intermediate echogenicity (gray) in conjunction with the presence of anechoic (black) perirenal fat in their vicinity. The renal pelvis had a darker hue (black) than the renal parenchyma, which can be attributed to the presence of adipose tissue. The distinctive morphology facilitated the recognition of the kidneys in the CT scan examination. These findings are in the same line with previous work on goats(Braun et al., 2011) and humans (Sagel et al., 2002). Furthermore, the substantial amount of perirenal fat present in the goat kidney contributed to its enhanced stability in its anatomical position (Jabbar et al., 2018). The transverse CT images revealed the presence of the renal hilus. Visible renal veins, arteries, and ureters were observed. The ureters had a smaller diameter than the renal vessels and were devoid of any associated urine content. The aforementioned result was confirmed in goats (Braun et al., 2010; Braun et al., 2011) and calves (Ohlerth and Scharf, 2007; Braun et al., 2014), while (Hathcock and Stickle, 1993; Ragab et al., 2010) asserted that in goats, the visualization of renal arteries and ureters in the renal hilus was occasionally challenging. Additionally, transverse CT revealed the primary source of the right renal artery to be the abdominal aorta. In contrast to the left renal artery, the right renal artery exhibits bifurcation into dorsal and ventral branches before entering the renal hilus. This observation was consistent with the findings reported by (Jabbar et al., 2018) on goats. Similarly, it was found that the urine bladder can be located at several levels, including the sacrum, pelvic floor below the uterus, and rectum, depending on its degree of fullness (Jabbar et al., 2018; Alnahrawy et al., 2021), and in calves (Braun et al., 2014). As in previous studies, the urine bladder was identified at the 6th lumbar vertebral level (Sajjadian et al., 2015). ConclusionThe findings of this study demonstrated that CT could be a significant imaging modality for examining the urinary system in goats. The use of anatomically frozen cross-slice images played a crucial role in verifying the morphology, location, and interconnections of the structures observed in CT images. The comprehensive visual representations were a valuable resource for surgeons and anatomists, facilitating the enhanced diagnostic evaluation of urinary tract disorders in goats. AcknowledgmentsThe authors express their gratitude to the staff members of the Anatomy and Embryology and Surgery, Anesthesiology and Radiology Departments, Faculty of Veterinary Medicine at Zagazig University, Egypt, for their support during our work. Conflict of interestThe authors confirm that we do not have any conflicts of interest to declare. FundingThe authors did not receive any external funding for this study. Authors’ contributionsM.A., W.Y., AAI, M.N., and E.E. executed the experiments, validated the results, shared in writing the original manuscript, and performed data curation and analysis. M.A. and G.A.S. conceptualized the study, abstracted the work, and wrote, reviewed, and edited the manuscript. All authors have read and agreed to the publication of the finale version of the manuscript. Data availabilityData not already included in the publications are available from the authors upon reasonable request. ReferencesAlnahrawy, E.H., Rashed, R., Shogy, K. and Erasha, A. 2021. Morphological and diagnostic imaging studies on pelvic cavity of Egyptian female baladi goat (Capra hircus). J. Cur. Vet. Res. 3, 32–40. Alsafy, M., El-Gendy, S., El-kammar, M.H. and Ismaiel, M. 2013. Contrast radiographic, ultrasonographic and computed tomographic imaging studies on the abdominal organs and fatty liver infiltration of zaraibigoat. J. Med. Sci. 13, 316–326. Braun, J., Van Den Berg, R., Baraliakos, X., Boehm, H., Burgos-Vargas, R., Collantes-Estevez, E., Dagfinrud, H., Dijkmans, B., Dougados, M., Emery, P., Geher, P., Hammoudeh, M., Inman, R.D., Jongkees, M., Khan, M.A., Kiltz, U., Kvien, T.K., Leirisalo-Repo, M., Maksymowych, W.P., Olivieri, I., Pavelka, K., Sieper, J., Stanislawska-Biernat, E., Wendling, D., Özgocmen, S., Van Drogen, C., Van Royen, B.J. and Van Der Heijde, D. 2010. Update of the ASAS/EULAR recommendations for the management of ankylosing spondylitis. Ann. Rhe. Dis. 70(6), 896–904. Braun, U., Irmer, M., Augsburger, H. and Ohlerth, S. 2011. Computed tomography of the abdomen in saanen goats: III. Kidneys, ureters and urinary bladder. Schw. Archiv. Fur Tie. 153, 321–329. Braun, U., Schnetzler, C., Augsburger, H., Bettschart, W. and Ohlerth, S. 2014. Computed tomography of the abdomen of calves during the first 105 days of life: III. Urinary tract and adrenal glands. Arch. Tierheilkd. 156, 237–247. Craig, W.D., Wagner, B.J. and Travis, M.D. 2008. Pyelonephritis: radiologic-pathologic review. Radiographics 28, 255–276. Dobry, E. and Danuser, H. 2009. Imaging of the kidney and the urinary tract. Therapeutische Umschau. Rev. Therap. 66, 39–42. Dyce, K., Sack, W. and Wensing, C. 2009. Textbook of veterinary anatomy (E-Book. Philadelphia, PA: Saunders-Elseveir Puplisher. El Karmoty, A., Daghash, S. and Shymaa, A.E. 2022. Some anatomical studies on the kidney of goat (Capra hircus) with special reference to angioarchitecture, ultrasonography and doppler. Turk. J. Vet. Anim. Sci. 46, 565–574. Fulton, L.K., Melody, S.C. and Farris, H.E. 1994. The goat as a model for biomedical research and teaching. Ilar J. 36, 21–29. Griffin, N., Grant, L.A., Bharwani, N. and Sohaib, S.A. 2009. Computed tomography in metastatic renal cell carcinoma. Semin Ultrasound CT MRI 30, 359–366. Hathcock, J.T. and Stickle, R.L. 1993. Principles and concepts of computed tomography. The veterinary clinics of North America. Small Anim. Pract. 23, 399–415. Hussain, MS. 2010. Essentials of caprine anatomy. Faisalabad: University of Agriculture Faisalabad Pakistan. Jabbar, A.I., Kareem, H., Raad, A., Ibrahim, S. and Lateef, A.N. 2018. Anatomical and histological invistgation of the kidney in goat (Capra hircus). Agri. Sci. J. 10, 1–12. Larsen, G.D. 2015. A reliable ruminate for research. Lab. Animal 44, 337. Lee, Y.J., Oh, S.N., Rha, S.E. and Byun, J.Y. 2007. Renal trauma. Radiol. Clin. North Am. 45, 581–592. Nomina Anatomica Veterinaria. 2017. Prepared by the International Committee on Veterinary Gross Anatomical Nomenclature and Authorized by the General Assembly of the World Association of Veterinary Anatomists. Hanover (Germany), Ghent (Belgium), Columbia, MO (USA), Rio de Janeiro: International Committee on Veterinary Gross Anatomical Nomenclature. Ogobuiro, I. and Tuma, F. 2019. Physiology, renal. Treasure Island, FL: StatPearls Publishing. Ohlerth, S. and Scharf, G. 2007. Computed tomography in small animals: basic principles and state of the art applications. Vet. J. 173, 254–271. Ragab, G.A., Seif, M.M. and Hagag, U. 2010. Radiologic and ultrasonographic studies of kidneys in goat. J. Vet. Med. Res. 20, 30–37. Sagel, S., Stanley, R. and Levitt, R. 2002. Computed tomography of the kidney. J. Urol. 167, 1028–1038. Sajjadian, S.M., Bahador S., and Zade, B.S. 2015. Computed tomographic anatomy of the abdominal cavity in the Jebeer (Gazella bennettii). Anat. Sci. J. 12, 37–44. Shojaei, B., Vajhi, A.R., Rostami, A., Molaei, M.M., Arashian, I. and Hashemnia, S. 2006. Computed tomographic anatomy of the abdominal region of cat. Iran. J. Vet. Res. 7, 45–52. Steffen, F. and Tontis, A. 1996. Clinical findings and pathology of congenital polycystic renal disease in the goat. Tierarztliche Praxis 24, 453–458. Steininger, K. and Tierheilkd, U.B. 2012. Ultrasonography of the urinary tract in 29 female saanen goats. Arch. Tierheilkd. 154, 67–74. Stieger-Vanegas, S.M. and McKenzie, E. 2021. Imaging of the urinary and reproductive tract in small ruminants. The veterinary clinics of North America. Food Anim. Pract. 37, 75–92. Washburn, Z.W., Dillman, J.R., Cohan, R.H., Caoili, E.M. and Ellis, J.H. 2009. Computed tomographic urography update: an evolving urinary tract imaging modality. Semin. Ultrasound CT MR 30, 233–245. | ||
| How to Cite this Article |
| Pubmed Style Aref M, Youssef WOM, Abdelbaset-ismail A, Salem GA, Nassan MA, Elsheikh EA. Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomography. Open Vet. J.. 2025; 15(2): 820-826. doi:10.5455/OVJ.2025.v15.i2.31 Web Style Aref M, Youssef WOM, Abdelbaset-ismail A, Salem GA, Nassan MA, Elsheikh EA. Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomography. https://www.openveterinaryjournal.com/?mno=226815 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.31 AMA (American Medical Association) Style Aref M, Youssef WOM, Abdelbaset-ismail A, Salem GA, Nassan MA, Elsheikh EA. Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomography. Open Vet. J.. 2025; 15(2): 820-826. doi:10.5455/OVJ.2025.v15.i2.31 Vancouver/ICMJE Style Aref M, Youssef WOM, Abdelbaset-ismail A, Salem GA, Nassan MA, Elsheikh EA. Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomography. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 820-826. doi:10.5455/OVJ.2025.v15.i2.31 Harvard Style Aref, M., Youssef, . W. O. M., Abdelbaset-ismail, . A., Salem, . G. A., Nassan, . M. A. & Elsheikh, . E. A. (2025) Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomography. Open Vet. J., 15 (2), 820-826. doi:10.5455/OVJ.2025.v15.i2.31 Turabian Style Aref, Mohamed, Walaa O. M. Youssef, Ahmed Abdelbaset-ismail, Gamal A. Salem, Mohamed A. Nassan, and Esraa A. Elsheikh. 2025. Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomography. Open Veterinary Journal, 15 (2), 820-826. doi:10.5455/OVJ.2025.v15.i2.31 Chicago Style Aref, Mohamed, Walaa O. M. Youssef, Ahmed Abdelbaset-ismail, Gamal A. Salem, Mohamed A. Nassan, and Esraa A. Elsheikh. "Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomography." Open Veterinary Journal 15 (2025), 820-826. doi:10.5455/OVJ.2025.v15.i2.31 MLA (The Modern Language Association) Style Aref, Mohamed, Walaa O. M. Youssef, Ahmed Abdelbaset-ismail, Gamal A. Salem, Mohamed A. Nassan, and Esraa A. Elsheikh. "Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomography." Open Veterinary Journal 15.2 (2025), 820-826. Print. doi:10.5455/OVJ.2025.v15.i2.31 APA (American Psychological Association) Style Aref, M., Youssef, . W. O. M., Abdelbaset-ismail, . A., Salem, . G. A., Nassan, . M. A. & Elsheikh, . E. A. (2025) Morphological examination of the normal kidney and urinary bladder in goats (Capra hircus) using anatomical sections and computed tomography. Open Veterinary Journal, 15 (2), 820-826. doi:10.5455/OVJ.2025.v15.i2.31 |