| Short Communication | ||
Open Vet. J.. 2025; 15(2): 1050-1055 Open Veterinary Journal, (2025), Vol. 15(2): 1050-1055 Short Communication The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, IraqZeinab A. M. Al-Tememe*, Mustafa Hadi Hamid and Mohanad Fadhl Hussain Al- MusodiDepartment of Animal Production, College of Agriculture, Kerbala University, Karbala, Iraq *Corresponding Author: Zeinab A. M Al-Tememe. Department of Animal Production, College of Agriculture, Kerbala University, Karbala, Iraq. Email: zainab.mohammed [at] uokerbala.edu.iq Submitted: 01/11/2024 Accepted: 21/01/2024 Published: 28/02/2025 © 2025 Open Veterinary Journal
AbstractBackground: The spread of fungi in agricultural products in Iraq poses a great challenge to the productivity of meat broilers because of the contamination of most components of feed leeches at various ages, including poor storage. Fungi were isolated from wheat grains. Aim: This study aimed to investigate the effect of the fungi associated with poultry feed on gizzard and liver relative weight in a number of breeding halls in Karbala, Iraq. Methods: Fungi were isolated from wheat grains brought from some poultry farming halls in Holy Karbala Province. Three random samples were taken for each site. The results were analyzed using complete randomized design, and means were compared using the least significant difference test. The results showed the appearance of Aspergiulls niger and Aspergiulls flavus with a frequency of 70% and 40%, respectively. The isolated fungi were diagnosed using approved taxonomic keys. Results: The chickens treated with the mycotoxin of A. flavus reached 0.95 g, while the A. niger gave a weight loss rate of 1.20 g, with a significant difference from the fungus A. flavus. The gizzard weight of chickens treated with the treatment recorded a significant difference of 1.34 and 0.96 g, respectively. Regarding the effect of mycotoxins on the relative level of the liver, the fungal poison of A. flavus recorded a weight of 2.36 g, while the fungus A. niger gave a weight loss rate of 2.51 g, a significant difference from the poison of A. flavus. Conclusion: The results showed that spoilage of poultry feed, including wheat, caused by A. niger and A. flavus has a negative impact on poultry. Aspergiulls niger and A. flavus cause decreased liver and gizzard weight in meat birds as a result of treatment with mycotoxins. Keywords: Aspergiulls niger, Aspergiulls flavus, Mycotoxin, Poultry feeds, Iraq. IntroductionThe scientific name of wheat is Triticum aestivum. It is one of the most important grain crops because of its high nutritional value. The grains are among the crops on which toxin-producing fungi grow (Moretti et al., 2017). Developed countries incur many losses due to mycotoxins commonly associated with trade usually, in developing countries, the losses are economical and health through decreased availability of food and decreased market value of contaminated products as well as a decrease in the marketability of crops and an increase in diseases and deaths of livestock and humans (Gbashi et al., 2018). There are five main groups of mycotoxins that can be present in wheat crops: Afla toxins, fumonisin, deoxynivalenol, okra, and zearalenone (ZEN) toxins, fumonisin and ZEN toxins may begin to be produced in the field before harvest while afla and okra poison is mostly produced during post-harvest storage mechanical infection, insect infestation and storage conditions are the main factors for increasing mycotoxin contamination (Khodaei et al., 2021). Fungi are important microorganisms in Iraq and around the world because of their importance in the lives of humans, animals, and plants, in addition to their importance as food and their use as products of vital metabolism. However, the resulting damage is very serious because it produces fungal toxins that have devastating effects on human and animal health (Summerell et al., 2010). It causes an increase in the production of reactive oxygen species. This has harmful effects on various organs and body systems, As well as poor absorption of various nutrients, which leads to malnutrition and stunted growth. It is also responsible for weak cellular immunity by inhibiting the production of productive immune cells and thus causing infectious diseases (Yilmaz and Bag, 2022). Mycotoxins are toxic secondary metabolites produced by many fungi during the stationary phase of growth in various types of food and grain crops (Ropejko and Twarużek, 2021). Mycotoxins have direct or indirect harmful effects on humans and animals, even at low concentrations (Vila-Donat et al., 2018). Storage fungi are more dangerous to the crop during storage, as storage fungi pose a serious problem for grains. Fungal infections result in negative effects represented in reducing the viability of grain germination and germination rate, in addition to the ability of many fungi officinalis to produce toxic metabolic compounds, these grains are called mycotoxins, which reduce their economic value and become unfit for human and animal consumption (Saeidi, 1985). The genus Aspergillus belongs to the order Eurotiales, the class of Ascomycota, and there are more than 60 species of Aspergillus fungi (Youssef, 2012). Some of which are of medical importance and cause infections in humans and animals (Hassan and Voigt, 2019). The principal and well-known mycotoxins produced by Aspergilli are ochratoxin A and aflatoxins, as well as less-prominent toxins like patulin (Keller et al., 2005). These toxins are present in various agricultural commodities (Varga et al., 2004). This study aimed to identify and diagnose fungi associated with wheat grain feed and the production of mycotoxins in fodder farms in Karbala province. Materials and MethodsFeed samplesSamples collected of wheat were brought from some poultry farming halls in the Holy Karbala province (Five halls). Three random samples were taken for each site, 200 g were weighed, the samples were mixed, and a portion of them was taken for the purpose of fungal isolation. Isolation of the pathogenThe wheat grains (T. aestivum) were washed with running water for half an hour, and then the pieces were surface sterilized using a 1% aqueous solution of sodium hypochlorate for 3 minutes. The pieces were removed from the solution, washed with sterile distilled water twice to remove the surface sterilant, and dried between two sterile Whatman No.10 Filter papers. They were placed in 9-cm-diameter sterile Petri dishes containing Potato dextrose agar (PDA) weighing 41 g of PDA per liter of distilled water, and added to the antibiotic chloromphenicol at a rate of 250 mg l-1 before hardening, with five seeds per plate after sterilizing in an autoclave for 20 minutes at 121°C and a pressure of 15 pounds/inch2, the plates were incubated at 25◦C ± 2°C for 4–5 days. The growing colonies were purified in Petri dishes containing potato extract medium, dextrose, and agar using Hyphal Tip Techniqul. Diagnosis of the fungi under study using approved taxonomic and molecular keysFor morphological identification, color, shapes, mycelia, and types of conidia were characterized and compared with previous descriptions. It was diagnosed based on the following classification keys: (Ellis et al., 2007). The frequency of each isolated fungus was calculated as follows: Preparation of mycotoxinsTwo fungal isolates were chosen based on the results of the previous paragraph, known for their presence in animal feed and for their production of mycotoxins (Moubasher, 1993; Pitt and Hocking, 1997). The medium was prepared potato dextrose broth which the antibiotic chloromphenicol 250 mg. l-1 was added, and then it was distributed into flasks with a capacity of 250 ml each. After sterilizing the medium and cooling it to room temperature, it was inoculated into discs with a diameter of 5 mm, from each selected fungal isolate, three replicates for each isolate. All flasks were incubated at a temperature of 25◦C ± 2°C for 2 weeks. The components of each beaker were mixed separately with an electric mixer for 3 minutes (taking into account washing and sterilizing the mixer and all other tools before and after each use). The contents of each flask were filtered using a Buechner funnel and a vacuum pump. Then, the extract was passed through a 0.45-micron diameter bacterial filter, the extracts were collected in clean, sterile glass bottles, and the mycotoxin was stored in the refrigerator at 4°C until use within 24 hours. Study of the effects of selected mycotoxins on chicken chicksMeat chickens were obtained from one of the private hatcheries in Karbala province. Hubbard Flex broiler chickens were obtained at 14 days of age. A special hall was prepared and equipped with all the necessary supplies for raising poultry (Al-Zajjaji, 1978). Adopting a special program for caring for and raising chicks and following a preventive program to protect them from diseases (Otto, 1979). These chicks were given a dose of 1 ml of mycotoxin per 50 g of body weight orally (Alasdi, 2006). Five birds for each fungal extract, with three replicates, in addition to the control group that was dosed with sterile distilled water, the experiment continued for 45 days, during which diets and water were given freely until the end of the experiment. Measure the relative weights of the gizzard and liverThe weights of the birds treated with mycotoxins were measured, as well as those of the control group treated with sterile distilled water and medical treatment. After 45 days, the birds were slaughtered, the livers and gizzards (gm/kg) were removed, and their relative weight was calculated using a sensitive balance in a meat laboratory.
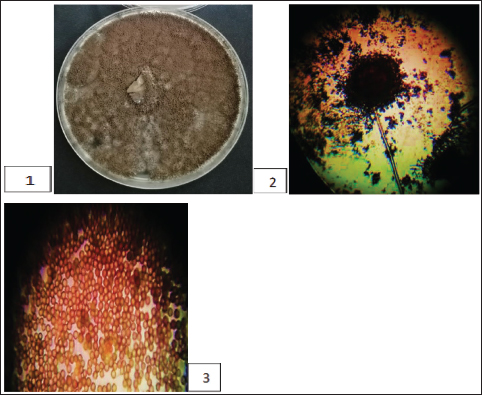
Fig. 1. The A. niger (1) colony appearance on PDA; (2) conidia; (3) the conidiophore ends with a swollen head containing the conidia.
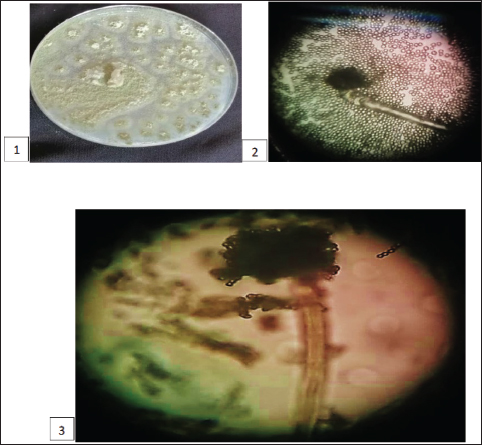
Fig. 2. A. flavus (1) colony appearance on PDA; (2) conidia; (3) an undivided, unbranched conidiophore with a head containing conidia in chains.
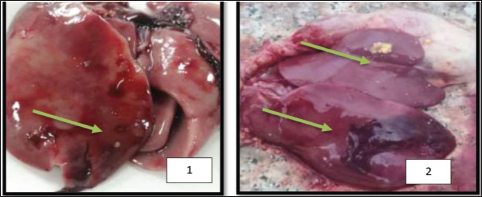
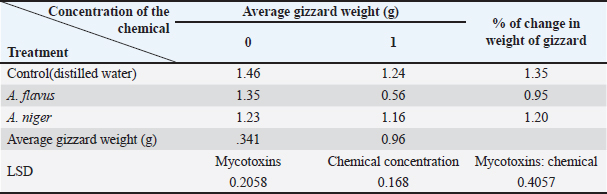
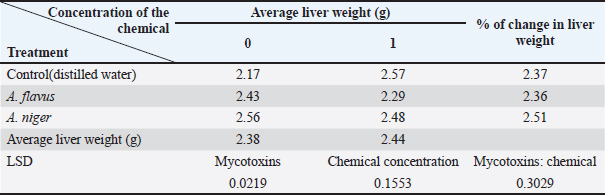
Fig. 3. Effect of mycotoxins on liver tissue: (1) 10 days old; (2) 45 days after the dose. Statistical analysisThe results were analyzed using a complete randomized design, and means were compared using the least significant difference test (LSD) at a probability level of 0.05 using SAS statistical analysis software (S.A.S, 2012). Results and DiscussionIsolation of the pathogenThe isolation results showed fungal growth on PAD medium, represented by two fungi Aspergiulls niger Aspergiulls niger and Aspergiulls flavus with a frequency of 70% and 40%, respectively. Aspergiulls nigerThe fungus grows rapidly over three to four days. The colony has a cottony appearance that is initially white to yellow and then turns green or black (Fig. 1). TMyceliumis characterized by abundant growth and is branched and divided. The conidia are spherical in shape, single-celled, with a rough outer wall, 3.5–5 mm in diameter, and dark brown to black in color (Someren et al., 1990). Aspergiulls flavusFungal colonies initially appeared white and velvety in shape, and after 3–4 days, the isolates produced conidia, it is yellowish green, olive, or dark green in color, which dominate the appearance of the colony (Fig. 2), usually simple and flat at the edges but were raised in the middle and wrinkled, the conidiophore appeared colorless, with thick walls, rough bearing semi-spherical vesicles in some isolates it is spherical in other isolates, with diameters ranging between 1,800 and 2,000 mm. Cells were either uniseriate, biseriate, or both the phialide appeared on the Metwalau it is directly connected to the vesicles, and the conidia are spherical in shape they have thin, slightly rough walls and range between 250 and 450 mm in diameter (Layton et al., 1977; Klich, 2002). Effect of mycotoxins on the relative weight of the gizzardThe results in Table 1 showed that there were significant differences between the average weights of chicken gizzards after 45 days of treatment with mycotoxins. The comparison treatment recorded the highest rate of 1.35 g for gizzard weight, with a significant difference from poultry treated with mycotoxins. The gizzard weight of chickens treated with A. flavus mycotoxin was 0.95 g, while the A. niger gave a percentage of weight loss amounting to 1.20 g, a significant difference from the A. flavus, while the gizzard weight of chickens treated with the treatment recorded a significant difference of 1.34 and 0.96 g, respectively; on the other hand, the interaction between the mycotoxin of the fungus A. flavus and chemotherapy recorded an average weight of 0.56 g a significant difference from the negative comparison amounted to 1.35 g, while the interaction between the A. niger and chemotherapy gave an average weight of 1.16 g without a significant difference with the negative comparison, which amounted to 1.34 g, the interaction with the positive comparison differed significantly for birds treated with mycotoxin of the A. flavus. These results are consistent with (Kayode et al., 2012), which indicated a significant decrease in gizzard weight after dosing meat birds with A. niger. Effects of mycotoxins on the relative weight of chicken liversFrom the results of Table 2, it was shown that there were significant differences between the average weights of chicken livers 45 days after treatment with mycotoxins. The comparison treatment recorded an average of 2.37 g for relative liver weight, a significant difference from poultry treated with mycotoxins, the fungal poison of A. flavus recorded a weight of 2.36 g, while the A. niger gave a weight loss rate of 2.51 g, a significant difference from the poison of A. flavus, the treatment did not differ significantly in its effect on the liver weight of poultry, recording an average of 2.38 and 2.44 g, respectively; on the other hand, the interaction between the mycotoxin of the A. flavus and chemotherapy recorded an average weight of 2.29 g, with a non-significant difference from the negative comparison of 2.43 g, while the interaction between the A. niger and chemotherapy gave an average weight of 2.48 g without a significant difference with the negative comparison, which amounted to 2.56 g, while the interaction with the positive control differed significantly for birds treated with the mycotoxin of the A. niger. The results of the current study are consistent with (Muhammad and Oloyede, 2010) what they said, which indicated a decrease in liver weight in meat birds after exposure to A. niger (Fig. 3). Table 1. Effect of mycotoxins of the A. flavus and A. niger mycotoxins and chemotherapy on the relative weight of the gizzard in chicken chicks.
Table 2. Effect of mycotoxins of the A. flavus and A. niger, mycotoxins and chemotherapy on the relative weight of the liver of chicken chicks.
This confirms the contamination of poultry feed, including wheat, and many fungi, including the genus Aspergillus, which were brought from various places in Holy Karbala. These fungal species are contagious and cause various diseases, especially the respiratory system of poultry, causing their death. These results were consistent with what Alasadi (2006) and Al-tememe et al. (2023). Aflatoxin is one of the substances that does not stimulate the immune system due to its small molecular weight, which may be related to beneficial microorganisms in the gastrointestinal tract. In addition, aflatoxin causes immune suppression resulting from inhibition of protein synthesis, a decrease in serum albumin and globulin levels, a decrease in levels of circulating antibodies in the blood, weakness in the endothelial-reticulo system, decreased immunity mediated by cells. Also, it affects the development of the thymus gland and the bursa of Fabricius (Miles and Bootwalla, 1991; Devegowda et al., 1998). We suggest the long-term effects of mycotoxin exposure on poultry health, developing effective strategies for mycotoxin decontamination, and exploring the potential of natural feed additives to mitigate the negative effects of mycotoxins. ConclusionThe results showed that spoilage of poultry feed, including wheat, because A. niger and A. flavus, hence its negative impact on poultry. AcknowledgmentsThe authors would like to be grateful to everybody who extended a helping hand to complete this research. Conflict of interestThere is no conflict of interest in this work. FundingThere is no financial support for researchers’ work done from their personal account. Authors’ contributionsZeinab A.M. Al-tememe, wrote the paper. Mustafa Hadi Hamid collected the data. Mohanad Fadhl Hussain Al-musodi contributed data and anaylsis tools. Data availabilityData are available to everyone. ReferencesAlasadi, S.H.A. 2006. Effect of filtrates of fungi isolated from poultry feed on the growth of chicken chicks in some areas of Karbala. Thesis, Faculty of Education, University of Kerbala, Iraq. Al-tememe, Z.A.M., Awad, H., Al-Hakeem, S.M. and Bajlan, S.G.Sh. 2023. Use nano-chitosan to inhibit the two fungi Aspergillus niger. and Nigrospora sphaerica which isolated from Zea mays L. laboratory. IJAS and T. 6( 1), 1–11. Al-Zajjaji, R. 1978. Practical lessons in poultry. Tahrir Library Publications, College of Agriculture-Sulaymaniyah University, Iraq. Ellis, D.H., Davis, S., Alexiou, H., Handke, R. and Bartley, R. 2007. Description of medical fungi, 2nd ed. Adelaide, South Australia: Nexus Print Solutions. Gbashi, S., Madala, N.E., De Saeger, S., De Boevre, M., Adekoya, I., Adebo, O.A. and Njobeh, P.B. 2018. The socio-economic impact of mycotoxin contamination in Africa. pp: 4–24. DOI: 10.5772/intechopen.79328. Hassan, M.I.A. and Voigt, K. 2019. Pathogenicity patterns of mucormycosis: epidemiology, interaction with immune cells, and virulence factors. Mycol. 57, 245–256; doi:10.1093/mmy/myz011. Kayode, R., Sani, A., Apata, D., Joseph, J. and Obalowu, M. 2012. Performance and carcass characteristics of broiler chickens fed on fungal mixed-culture (Aspergillus niger and Penicillium chrysogenum) fermented mango kernel cake. GR. J. Microbiology 2(1), 67–75. Keller, N.P., Turner, G. and Bennett, J.W. 2005. Fungal secondary metabolism—from biochemistry to genomics. Nat. Rev. Microbiol. 3, 937–947; doi:10.1038/nrmicro1286. Khodaei, D., Fardin, J. and Amin Mousavi, K. 2021. Global overview of the occurrence of mycotoxins in cereals: a three-year survey. Curr. Opin. Food Sci. 39, 36–42. Klich, M.A. 2002. Identification of Common Aspergillus Species. Centraalbureau poor Schimmelcultures. Utrecht, The Netherlands, pp: 426–432. Available at: https://wi.knaw.nl/images/publications/AspergillusSpecies.pdf Layton, Y.M. 1977. Medically important fungi: a guide to identification. Proc. R. Soc. Med. 70, 359. Moretti, A., Antonio, F., Logrieco, M. and Antonio, S. 2017. Evaluation of the environmental and human health impact of road construction activities. J. Cleaner Pro. 172(1), 1004–1013. Moubasher, A.H. 1993. Soil fungi in Qatar and other Arab countries. The Center for Scientific and Applied Research Publishing. University of Qatar, Qatar. Muhammad, N. and Oloyede, O. 2010. Growth performance of broiler chicks fedan Aspergilluss niger- fermented Terminalia catappa seedmeal–based diet. Int. J. Biol. Chem. Sci. 4(1), 107–114. Otto, H.S. 1979. Merck veterinary manual, 5th ed. Merck & Co. Inc. Pitt, J.I. and Hocking, A.D. 1997. Fungi and food spoilage, 2nd ed. Britain, UK: Chapman & Hall Publishing. University Press. Ropejko, K. and Twarużek, M. 2021. Zearalenone and its metabolites-general overview, occurrence, and toxicity. Toxins 13(1), 35. Saeidi, K.K. 1985. The presence of aflatoxin and arenose in some grains and their food products in some Iraqi governorates. IJAS (IZNCO). 3(2), 177–165. Someren, J.W., Eveline, E.O., Hagebeuk, B. and Cees Lijzenga, A.C. 1996. Circadian rest—activity rhythm disturbances in alzheimer’s disease. Biol. Psychiat. 40(4), 259–270. S.A.S. 2012. Statistical analysis system, users guide. Statistical Version 9.1. Cary, NC: SAS. Instituted Inc. Summerell, B.A., Laurence, M.H., Liew, E.C. and Leslie, J.F. 2010. Biogeography and phylogeography of Fusarium: a review. Fungal Divers. 44(1), 3–13. Vila-Donat, P., Marín, S., Sanchis, V. and Ramos, A.J. 2018. A review of the mycotoxin adsorbing agents for animal feed decontamination with an emphasis on their multi-binding capacity, for animal feed decontamination. Food and Chem. Toxicol. 114, 246–259. Yilmaz, S. and Bag, H. 2022. Aflatoxin B1: mechanism, oxidative stress, and effects on animal health. Insights Vet. Sci. 6, 17–24. Youssef, M.M.S. 2012. Maintenance of the manuscripts science and work. House of Arab Writer for Printing and Publishing. Varga, J., Tóth, B., Mesterházy, Á., Téren, J. and Fazekas, B. 2004. Mycotoxigenic fungi and mycotoxins in foods and feeds in Hungary. In An overview on toxigenic fungi and mycotoxins in Europe. Eds., Logrieco, A. and Visconti, A. Dordrecht: Springer. | ||
| How to Cite this Article |
| Pubmed Style Al-tememe ZAM, Hamid MH, Al-musodi MFH. The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, Iraq. Open Vet. J.. 2025; 15(2): 1050-1055. doi:10.5455/OVJ.2025.v15.i2.53 Web Style Al-tememe ZAM, Hamid MH, Al-musodi MFH. The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, Iraq. https://www.openveterinaryjournal.com/?mno=227002 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.53 AMA (American Medical Association) Style Al-tememe ZAM, Hamid MH, Al-musodi MFH. The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, Iraq. Open Vet. J.. 2025; 15(2): 1050-1055. doi:10.5455/OVJ.2025.v15.i2.53 Vancouver/ICMJE Style Al-tememe ZAM, Hamid MH, Al-musodi MFH. The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, Iraq. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 1050-1055. doi:10.5455/OVJ.2025.v15.i2.53 Harvard Style Al-tememe, Z. A. M., Hamid, . M. H. & Al-musodi, . M. F. H. (2025) The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, Iraq. Open Vet. J., 15 (2), 1050-1055. doi:10.5455/OVJ.2025.v15.i2.53 Turabian Style Al-tememe, Zeinab A. M., Mustafa Hadi Hamid, and Mohanad Fadhl Hussain Al-musodi. 2025. The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, Iraq. Open Veterinary Journal, 15 (2), 1050-1055. doi:10.5455/OVJ.2025.v15.i2.53 Chicago Style Al-tememe, Zeinab A. M., Mustafa Hadi Hamid, and Mohanad Fadhl Hussain Al-musodi. "The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, Iraq." Open Veterinary Journal 15 (2025), 1050-1055. doi:10.5455/OVJ.2025.v15.i2.53 MLA (The Modern Language Association) Style Al-tememe, Zeinab A. M., Mustafa Hadi Hamid, and Mohanad Fadhl Hussain Al-musodi. "The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, Iraq." Open Veterinary Journal 15.2 (2025), 1050-1055. Print. doi:10.5455/OVJ.2025.v15.i2.53 APA (American Psychological Association) Style Al-tememe, Z. A. M., Hamid, . M. H. & Al-musodi, . M. F. H. (2025) The effect of the mycotoxins of some fungi isolated from poultry feeds on the relative weight of the liver and gizzard of broiler chicks in different regions of Karbala, Iraq. Open Veterinary Journal, 15 (2), 1050-1055. doi:10.5455/OVJ.2025.v15.i2.53 |