| Research Article | ||
Open Vet. J.. 2025; 15(2): 795-803 Open Veterinary Journal, (2025), Vol. 15(2): 795-803 Research Article Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosionHanadi J. Al-Zubaidi1, Mohammed T. Mohammed2, Aoula Al-Zebeeby1*, Azhar A. Alkaby1 and Wurood Razzaq Hassan31Department of Pathology and Poultry Diseases, Faculty of Veterinary Medicine, University of Kufa, Al-Najaf Al-Ashraf, Iraq 2Department of Veterinary Microbiology, Faculty of Veterinary Medicine, University of Kufa, Kufa, Al-Najaf Al-Ashraf, Iraq 3Department of Anatomy and Histology, Faculty of Veterinary Medicine, University of Kufa, Kufa, Al-Najaf Al-Ashraf, Iraq *Corresponding Author: Aoula Al-Zebeeby, Department of Pathology and Poultry Diseases, Faculty of Veterinary Medicine, University of Kufa, Al-Najaf Al-Ashraf, Iraq. Email: aoulae.alzebeeby [at] uokufa.edu.iq Submitted: 04/11/2024 Accepted: 28/1/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
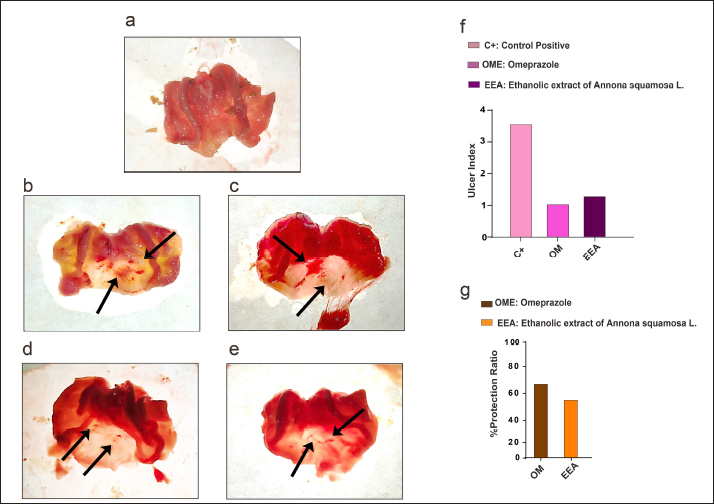
AbstractBackground: Annona squamosa L. is a medically important fruit that belongs to the family Annonaceae and has many medical benefits, including antiulcer effects. Aim: The present study explored the potential protection and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa L. (EEA) against ethanol-mediated peptic sever erosion using a rodent model in comparison with a synthetic compound broadly used in clinics to tackle peptic disturbances, known as Omeprazole. Methods: To do that, twenty-four both sex Wistar rats were assigned into four groups (six rats per group), namely control negative (C−) and control positive (C+) both received normal saline, Omeprazole (OM) administered OM at a dose of 20 mg/kg of body, and crude EEA received the extract at a dose of 200 mg/kg for two weeks. All groups except the C- group received absolute ethanol at a dose of 5 ml/kg of body weight for 1 hour. After euthanization, the gross ulcer index and protection ratio results exhibited a good percentage of protection in the EEA group compared with the C+ and OM groups. Results: These results were correlated with histopathological examination, which indicated that the gastric lesion of the EEA showed erosion in the pits area and isthmus, including destruction of parietal cells, whereas the gastric lesion of the C+ group showed severe necrosis of epithelial cells of the gastric mucosa, resulting in the absence of all mucosal layers of the affected area. In addition, the results revealed the anti-inflammatory properties of EEA through its ability to control the regulation of TNF-α and IL-1α. Both proteins were downregulated in response to the presence of EEA. Conclusion: To our knowledge, this is the first study to detect high IL-1α in ethanol-induced severe gastric erosion. These results suggested that EEA exhibited protective and anti-inflammatory effects against ethanol-mediated gastric erosion. Keywords: Annona squamosa L. fruit, antiulcer, IL-1 α, Omeprazole, ulcer index. IntroductionA gastric ulcer is a disturbance impairing the mucosal layer integrity of the stomach. This condition occurs due to a hierarchical inflammatory process activity leading to local injury and excavation through gastric acid and pepsin secretion (Shivhare and Vinode, 2023). Gastric ulcers and hyperacidity are two common causes of discomfort in people. According to a new statistical study, this disease affects 1 out of 10 patients throughout their lifetime (Xu et al., 2021). High mortality and morbidity rates are associated with gastric ulcers as a result of hemorrhage, perforation, and obstruction (Bertleff and Lange, 2010). Globally, one of the most common health issues is the gastric ulcer, especially with increasing risk factors, such as smoking, nutritional deficiency, stress, and alcohol drinking (Ravisankar et al., 2016). At the molecular level, gastric ulcers are characterized by increased proinflammatory cytokines expression and oxidative stress (Mei et al., 2012; Paikray et al., 2024). Different cytokines are involved in the inflammatory response as a result of gastric ulcer, including TNF-α and IL-6 (Aziz et al., 2018). Through the years, efforts have continued to develop new medications for treating gastric ulcers successfully. One of these drugs, a proton pump inhibitor (PPI), known as Omeprazole (OM), is a medication considered a first-line treatment for acute gastric ulcers by reducing the secretion of gastric acid (Malfertheiner et al., 2007; Sharma, 2023). However, therapy that is already used in the clinic may fail due to a direct or indirect cause, such as arrhythmia, impotence, hypersensitivity, gynecomastia, and hematopoietic changes (Kuna, 2019). Therefore, the global trend toward the utilization of alternative medicine, or what is known as plant-derived medicine, is due to an increase in up-to-date knowledge regarding medical properties, including the safety, efficacy, and quality of medicinal products of such types of medication (Falcão et al., 2008; Naji et al., 2018). One of these medically important plants, Annona squamosa L., is a tropical, domestic species of India, Bermuda, the Bahamas, Mexico, Brazil, Peru, Ecuador, the West Indies, and Central and South America (El-Chaghaby et al., 2014; Salmerón-Manzano et al., 2020; Al Kazman et al., 2020; Safira et al., 2022). A. squamosa L. belongs to the Annonaceae family, and the tree grows as a small shrub, with large randomly organized branches having a light brownish to brownish coat with thin leaves (Kalidindi et al., 2015). The A. squamosa L. is an edible fruit that contains vitamins (such as vitamin B1 (thiamine) and vitamin C), minerals, and dietary fiber. It has also been used in different food applications (Zahid et al., 2018). Many studies have revealed that A. squamosa L. has medical properties. Extracts yielded from different parts of the A. squamosa L. plant are used to treat a variety of diseases, including tumors, fever, dysentery, hemorrhage, and epilepsy (Anaya-Esparza et al., 2020). Therefore, the present study aimed to evaluate the protective and anti-inflammatory effects of the crude ethanolic extract of A. squamosa L. on ethanol-induced gastric erosion using a rodent model. MethodsAnimalsTwenty-four healthy, both sexes, and adult Wistar rats with a weight range of 150-200 g were obtained from the Faculty of Science/University of Kufa. Rats were placed in plastic cages (two rats per cage) with wood chips for bedding and housed at an animal house, Faculty of Science/University of Kufa, under controlled conditions. Rats were provided with free access to standard rat pellets and drinking water. Rats were grouped randomly (using the matched pairs method, according to the sex) and adapted for a one-week experimental period. Experimental designThe animals were divided into four groups: each group consisted of six rats, namely control negative (C−), control positive (C+), OM (Germany), and crude ethanolic extract of Annona squamosa L. (EEA). Each group was subjected to receiving the compounds by gavage daily for 2 weeks at a specific dose as follows: the C− group received normal saline, while the C+ group received normal saline before receiving absolute ethanol (Spain) at a dose of 5 ml/kg of body weight (Park et al., 2021). The OM group received OM at a dose of 20 mg/kg (Nworgu et al., 2019), and the EEA group received EEA at a dose of 200 mg/kg (Hamid et al., 2012) before receiving absolute ethanol at a dose of 5 ml/kg of body weight. All rats were euthanized 1 hour after receiving absolute ethanol via exsanguination under general anesthesia using ketamine and xylazine. Collection and preparation of crude ethanolic extract of A. squamosa L. fruitA fresh of Anonna Squamosa L. fruit was purchased from the local market in Al-Najaf. The plant was classified by a professional plant taxonomist in the Faculty of Science/University of Kufa according to the formal document No. 1,315 on April 2, 2023. For the extract preparation, the fruit Anonna squamosa L. was washed in water, cut into small pieces, dried in shade, and then converted to fine powder using a grinder; 50 mg of the produced fruit powder were mixed with 500 ml of 70% ethanol for 24 hours on a magnetic stirrer at 45°C. The extract was filtered using Whatman No.1 filter paper twice, which was then concentrated under reduced pressure using a rotary evaporator at 40°C and 90 rpm (Harborne, 1998). The final concentrated extract was refrigerated in sterile sample bottles for further investigation. Induction of gastric ulcerThe ulcer was induced after 24 hours of fasting by administration of 99.9% absolute ethanol at a dose of 5 ml/kg of body weight by gavage. One hour later, the animals were anaesthetized with ketamine and xylazine. The stomach samples were dissected and opened along the greater curvature to determine the number and length of gastric lesions (Morsy et al., 2012). Evaluation of gross gastric lesionsGastric ulcers were measured following the induction of each experimental group. First, the dissected stomach was placed on transparent glass or a board and then scanned using a camera. The ulcer scoring was made according to Takagi and Okabe (Takagi and Okabe, 1968): 0 no lesion, 1–2 mucosal petechial, 1–5 small lesions size 1–2 mm, and < 5 small lesions or one intermediate lesion 3–4 mm, 2 or more intermediate lesions or one gross lesion > 4 mm and perforated ulcers. The equations used to calculate the ulcer index and percentage of protective ratio are as follows: Histopathological preparing studyStomach samples were collected, washed once with PBS, and fixed within 10% formalin (Chemanol, KSA) for (48 hours) with changing the formalin of the collected samples 24 hours later. The stomach samples were then sliced into pieces measuring 0.5 cm in thickness each and excised from the affected tissue. After fixation steps, several steps were performed for preparing samples, namely the dehydration process (done by serially increasing concentrations of ethanol (Retouch, China), paraffin embedding (Chemact Petrochemicals, China) blocks step, sectioning and mounting on the slide, and staining step using hematoxylin and eosin (Leica Biosystems, USA). Finally, histopathological changes and imaging were detected using a light microscope at (10X and 40X) magnifications (Luna, 1968). Immunohistochemistry studyStomach samples in paraffin-embedded form were sliced into 4 µm thick sections, carefully placed in a water bath (FALC BI, Italy), and then mounted on positively charged glass slides (CrystalCruz® Electro-Statically Charged Micro Slides, sc-363562, SANTA CRUZ BIOTECHNOLOGY, USA). The stomach tissue section was deparaffinized (using a hot air oven at 55°C for 1 hour) and rehydrated (using xylene and gradually decreasing concentrations of ethanol for 3 minutes/concentration). Immunohistochemistry was performed according to the manufacturer’s protocol using a Dako EnVision detection immunohistochemistry kit (Envision FLEX, Dako, K8000, Denmark). In the following step, slides were incubated with the primary antibodies, namely anti- necrosis factor-α primary antibody (Polyclonal Rabbit Antibody: E-AB-40015, Elabscience, China) (TNF-α) and anti-interlukin-1α primary antibody (Polyclonal Rabbit Antibody: E-AB-40407, Elabscience, China) (IL-1 α). The sections were incubated with a secondary antibody labelled horseradish peroxidase (EnVision FLEX /HRP, SM802) and were then examined using DAB+ chromogen. All tissue sections were stained with Mayer haematoxylin (Bio-Optica, 05-06002/L, Italy), dehydrated, and cleared. Finally, all slides were mounted with a cover slide, the protein signal was detected using a light microscope, and the image was captured using 10X and 40X. Statistical analysisThe one-way F test with multiple comparisons (ANOVA) and the Fisher test (the least significant difference) P≤0.01 were achieved to compare the control positive group (C+), OM group, and the crude ethanolic extract of A. squamosa L. (EEA). The statistical analysis was carried out using GraphPad Prism 6 software for Windows (La Jolla, CA, USA). Ethical approvalAccording to ethical instructions, this study was approved by the Ethical Committee of Animal Care and Use/University of Kufa, as documented through formal application No. 11203 on 7 May 2023. ResultsEEA reduces stomach hemorrhage following absolute ethanol treatmentIn order to investigate the protective effect of the ethanolic extract of A. Squamosa L. (EEA), a gross examination took place and the observation revealed hemorrhagic lesions in the stomachs of all rats that received absolute ethanol. However, the severity of hemorrhagic lesions was varied, where hemorrhagic patches with the presence of intermediate-sized lesions were observed in the C+ group (Fig. 1b and c). While the petechial hemorrhagic lesions observed in the EEA group (Fig. 1d) compared with OM group (Fig. 1e) showed convergent results. In correlation with the gross results, the ulcer index showed a lower number of stomach ulcers in the EEA group compared with the C+ group. The OM-treated group indicated similar results to the EEA group (Fig. 1f). Along with these results, the protection ratio revealed a good protective percentage in the EEA group compared with the OM group (Fig. 1g).
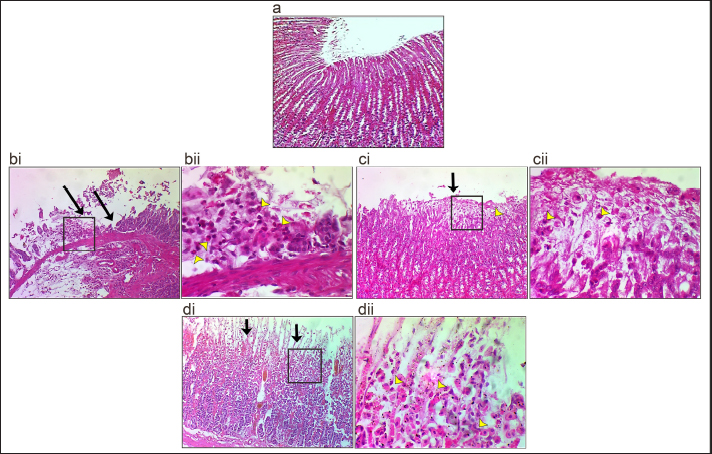
Figure 1. EEA showed high protection ability against ethanol-mediated ulcer formation. (a) C- group (b and c). C+ group: hemorrhagic patches and intermediate hemorrhagic lesions (black arrows) were observed in the body area of the affected stomach. (d) OM group: Two petechial hemorrhagic lesions (arrows) were observed in the body area of the affected stomach. (e) EEA group: three petechial hemorrhagic lesions were observed in the body area of the affected stomach. (f) Ulcer index. (g) Percentage of protection ratio, mean, n=6. Further investigation, histopathological examination showed that in the C+ group, the lesion was characterized by severe erosion in gastric mucosa due to the necrosis of epithelial cells in pits, isthmus, and neck of mucosal layer accompanied with infiltration of neutrophils (Fig. 2 bi and bii) compared with the C- group did not exhibit any pathological changes (Fig.2a). While the OM group showed a small area of erosion and necrosis of epithelial cells observed in the surfaces of gastric mucosa involving the upper pits layer with the presence of inflammatory cell infiltration (Fig. 2ci and cii). In the case of EEA, necrotic areas of epithelial cells were observed in the pits of gastric mucosa that involved all pits with the presence of inflammatory cell infiltration in the affected area. These results indicated that EEA reduced the effect of ethanol-mediated stomach erosion.
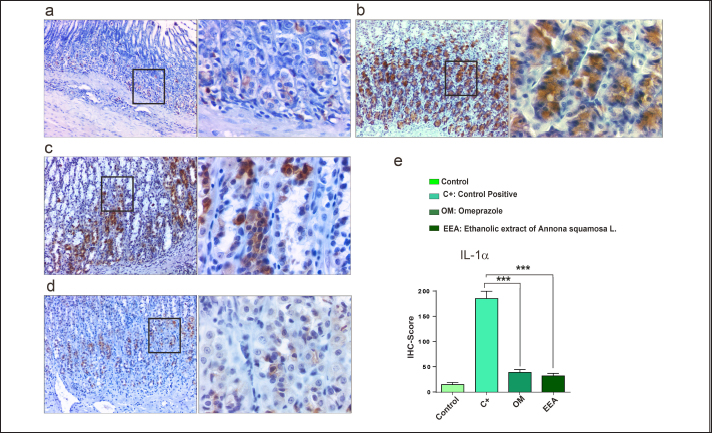
Figure 2. Histopathological section of stomach. (a) C- group showed normal gastric histological architectures. (bi and bii) C+ group showed severe necrosis (black arrows) of epithelial cells of gastric mucosa, where the necrosis of epithelial cells led to the absence of all mucosal layers of the affected area. In addition, infiltration of inflammatory cells (mostly neutrophil) (yellow arrowhead) was observed in the necrotic area. (ci and cii) OM group showed that the necrosis of epithelial cells (black arrows) was observed in surfaces of the gastric mucosa that involved the upper pits layer with the presence of inflammatory cell infiltration (yellow arrowhead) in the affected area. (di and dii) EEA group showed necrosis of epithelial cells (black arrows) was observed in the pits layer of the gastric mucosa that involved the all-pits layer with the presence of inflammatory cell infiltration (yellow arrowhead) in the affected area (H and E: 10X and 40X). Downregulation of TNF−α and IL-1 α correlates with anti-inflammatory effect of EEAFor further confirmation and to check the anti-inflammatory properties of EEA, the expression levels of TNF-α and IL-1α were examined. The results demonstrated that TNF-α was highly expressed in the C+ group (Figs. 3b, 4b) compared with the C- group (Figs. 3a and 4a). Interestingly, the expression level of IL-α was high as well. In contrast, a significant decrease in the expression levels of both TNF-α and IL-1α was observed in the OM group (Figs. 3c, 4 c) and EEA group (Figs. 3d, 4d) compared with the C+ group (Figs. 3e, 4e). Taken together, these results confirmed the anti-inflammatory properties of EEA following the induction of an ethanol-mediated stomach erosion.
Figure 3. Low expression of TNF-α in response to the EEA protection effect. (a) Control negative group, (b) C+ group, (c) OM group, (d) EEA group, and (e) Immunohistochemistry (IHC) score showed a significant decrease of TNF-α in OM and EEA groups compared with the C+ group. ***p . 0.001, error bars=mean ±} standard error of mean (n=3). DiscussionGastric ulcers are well-described and commonly occur in clinics due to a variety of causes. It has been found that gastric ulcers result from an imbalance in the secretion of pepsin and gastric acid in addition to defensive factors of the gastric mucosal layer (Périco et al., 2020). The stomach defense lines can be debilitated and penetrated by chemical compounds, such as alcohol, though, creating lesions in the epithelial layer and increasing edema formation due to increasing the permeability of the vascular layers. Such lesions can be induced by ethanol by different mechanisms, including a decrease in gastric PH, resulting in lowering gastric motility, and reducing mucus and bicarbonate secretions (Abd-Alla et al., 2022). Stereotypically, ethanol is the most convenient material for inducing gastric ulcers in rat models. It causes a progressive disturbance in the microcirculation of the mucosal layer as well as ischemia, resulting in free radical production. In addition to increased gastric acid secretion, hemorrhage and areas of ulcer (Abd-Alla et al., 2022). In the current study and agreement with previous studies, using absolute ethanol only as a control positive showed the successful formation of gastric erosion, where gross observation showed the patches of hemorrhagic lesions in the affected stomach (Fig. 1b–e) and the histopathological observation showed the destruction of the gastric mucosa, which included all layers of gastric mucosa (Fig. 2bi and bii). Over the years, many chemical compounds have been discovered, developed, and used in clinics to tackle gastric ulcers. One of these well-known compounds, OM, is known as the principal and first-line medication used for peptic ulcer treatment through the reduction of gastric acid secretion (Malfertheiner et al., 2007; Sharma, 2023). In spite of successful outcomes of medical compounds that used to cure peptic ulcer, unpredictable and unwanted side effects have been recorded (Lambert et al., 2015; Kuna et al., 2019; Paz et al., 2020). Therefore, alternative or herbal medicine is becoming the most popular medicine for treating various diseases, including peptic ulcer (Naji et al., 2018; Kuna et al., 2019). Numerous studies have illustrated that many plants belonging to different plant families have medical utilization. One of these Annonaceae is, a plant family that possesses a promising property regarding gastric ulcer treatment (Mahima et al., 2020). The phytochemical screening of A. squamosa L. illustrated an excellent percentage of a wide variety of bioactive and antioxidant compounds, such as distinct metabolites, flavonoids, alkaloids, terpenoids, saponins, phytosterols, tannins, carbohydrates, phenolic compounds, and fixed oil (Othman et al., 2019; Avhad and Attarde, 2021). All these bioactive compounds have been used for their pharmacological properties to treat different medical conditions (Othman et al., 2019). These medical conditions can be summarized as follows: anti-inflammatory, anti-diabetic, antipyretic, anti-diarrhea and antifertility (Mahima et al., 2020). Other studies have demonstrated that extracts from A. squamosa L. twigs exhibit significant antiulcer activity. In the same context, it has been found that A. squamosa L. fruit manifested clear cryoprotection in the case of aspirin and alcohol models by controlling the mucin level (Yadav et al., 2011). In the present study, EEA and OM showed a convergent protective effect against ethanol-induced gastric erosion compared with the C+ group (Fig. 1f and g). Similarly, the histopathological results of the EEA group (Fig. 2ci and cii) and the OM group (Fig. 2di and dii) exhibited necrosis only involving epithelial cells that lined the pits with infiltration of inflammatory cells (neutrophils). These results suggested that the protective features observed in EEA group were similar to those of the OM-treated group compared with the negative and positive control groups. TNF-α is an important proinflammatory cytokine that plays a vital role in initiating and developing gastric ulcers. The development of this disorder occurs by controlling the primary acute inflammatory response. This is illustrated by increasing infiltration of neutrophils into the gastric mucosa (Fiorucci et al., 1998). In addition, TNF-α is modulating cell death in the gastric mucosa. Therefore, downregulation of TNF-α might facilitate the gastric ulcer healing process. Consequently, in the present study, we examined the expression level of TNF-α and the results revealed a significant increase in the C+ group, while the EEA group showed a significant reduction in TNF-α expression level (Fig. 3). These results correlate with a previous study done by Aziz et al. (2018), which suggested the amelioration of gastric ulcers and reduction in TNF-α expression levels following treatment with oxyresveratrol. IL-1α is another crucial and ubiquitous proinflammatory cytokine. Constitutively, IL-1α precursor is normally expressed in almost all types of healthy cells. However, it is released as a bioactive mediator due to the inflammatory process of necrosis (Cavalli et al., 2021). It has been found that the expression levels of TNF-α and IL-1β were high in the case of ethanol-mediated gastric ulcer (Amirshahrokhi and Khalili, 2015). Surprisingly, our findings revealed a significant increase in the expression level of IL-1α following ethanol induction, whereas the EEA treated group indicated a significant decrease in the expression level of IL-1α (Fig. 4). These results suggested a brand-new vital role of involvement of the IL-1α in the inflammatory response of gastric erosion induced by ethanol. IL-1α and IL-1β have been encoded through distinct genes with very low homology. However, both of these cytokines are binding the same IL-1 family receptor known as IL-1R. Therefore, they induce similar pro-inflammatory effects (Rider et al., 2013; Dincă et al., 2022). Interestingly, the intervention of IL-1α in the development of inflammatory diseases has relatively remained unnoticed, which may be because the records of different patients with inflammatory diseases illustrated that IL-1α is hardly detected in the circulation (Cavalli et al., 2021), which may explain the upregulation of IL-1α in the tissue. The current study investigated the protective features of EEA against ethanol-mediated gastric erosion in different aspects. The EEA exhibited a very good degree of protection with anti-inflammatory properties. The take-home message is the administration of crude EEA L. reduced ethanol-induced acute erosion in Wistar rats with relatively high protection and anti-inflammatory effects. In the context the inflammatory response, and to our knowledge, this is the first record for up-regulation of IL-1α in C+ and downregulation in protected groups with EEA.
Figure 4. Low expression level of IL-1α in response to the EEA protection effect. (a) Control negative group, (b) C+ group, (c) OM group, (d) EEA group, and (e) Immunohistochemistry (IHC) score showed a significant decrease of IL-1α in OM and EEA groups compared with the C+ group. ***p . 0.001, error bars=mean ±} standard error of mean (n=3). AcknowledgmentsWe would like to acknowledge Asst. Prof. Dr. Hutheyfa AlSalih Department of Pathology and Poultry Diseases/Faculty of Veterinary Medicine, University of Kufa, for his help in immunohistochemistry processing. Conflicts of interestThe authors declare no conflicts of interest regarding this publication. FundingThis research received no specific grant. Author’s contributionHJA. and MTM. designed the study and performed the experiment. AA analyzed data, wrote the manuscript, and prepared the figures. AAA. and WRH. processed tissues and prepared slides. ReferencesAbd-Alla, H.I., Ibrahim Fouad, G., Ahmed, K.A. and Shaker, K. 2022. Alloimperatorin from Ammi majus fruits mitigates Piroxicam-provoked gastric ulcer and hepatorenal toxicity in rats via suppressing oxidative stress and apoptosis. Biomarkers 27(8), 727–742. Al Kazman, B.S.M., Harnett, J.E. and Hanrahan, J.R. 2022. Traditional uses, phytochemistry and pharmacological activities of Annonacae. Molecules 27(11), 3462. Amirshahrokhi, K. and Khalili, A.R. 2015. The effect of thalidomide on ethanol-induced gastric mucosal damage in mice: Involvement of inflammatory cytokines and nitric oxide. Chem. Biol. Interact. 225, 63–69. Anaya-Esparza, L.M., de García-Magaña, M. L., Abraham Domínguez-Ávila, J., Yahia, E.M., Salazar-López, N.J., González-Aguilar, G.A. and Montalvo-González, E. 2020. Annonas: underutilized species as a potential source of bioactive compounds. Food Res. Int. 138, 109775. Aqubi, A.E., Al-Naimi, R.S. and Al-Taee, E.H. 2011. Comparative pathological and cytogenetical study of ethanolic extract of Vinca rosea L. and Vinblastine in treating mammary gland adenocarcinoma implanted mice. KJVMS 2(1), 146–163. Avhad, R. and Attarde, D. 2021. A review on Annona squamosa L .: pharmacognosy , phytochemical analysis, and pharmacological activities. IJPPR 21(3), 598–608. Aziz, R.S., Siddiqua, A., Shahzad, M., Shabbir, A. and Naseem, N. 2019. Oxyresveratrol ameliorates ethanol-induced gastric ulcer via downregulation of IL-6, TNF-α NF-ĸB, and COX-2 levels, and upregulation of TFF-2 levels. Biomed. Pharmacother. 110, 554–560. Bertleff, M.J.O.E. and Lange, J.F. 2010. Perforated peptic ulcer disease: a review of history and treatment. Dig. Surg. 27(3), 161–169. Cavalli, G., Colafrancesco, S., Emmi, G., Imazio, M., Lopalco, G., Maggio, M.C. and Dinarello, C.A. 2021. Interleukin 1α: a comprehensive review on the role of IL-1α in the pathogenesis and treatment of autoimmune and inflammatory diseases. Autoimmun. Rev. 20(3), 102763. El-Chaghaby, G.A., Ahmad, A.F. and Ramis, E.S. 2014. Evaluation of the antioxidant and antibacterial properties of various solvents extracts of Annona squamosa L. leaves. Arabian J. Chem. 7(2), 227–233. Falcão, H.S., Mariath, I.R., Diniz, M.F.F.M., Batista, L.M. and Barbosa-Filho, J.M. 2008. Plants of the American continent with antiulcer activity. Phytomedicine 15(1–2), 132–146. Fiorucci, S., Antonelli, E., Migliorati, G., Santucci, L., Morelli, O., Federici, B. and Morelli, A. 1998. TNFα processing enzyme inhibitors prevent aspirin-induced TNFα release and protect against gastric mucosal injury in rats. Aliment. Pharmacol. Ther. 12(11), 1139–1153. Hamid, R.A., Foong, C.P., Ahmad, Z. and Hussain, M.K. 2012. Antinociceptive and anti ulcerogenic activities of the ethanolic extract of Annona muricata leaf. Rev. Bras. Farmacogn. 22(3), 630–641. Harborne, JB. 1998. Textbook of Phytochemical Methods. A Guide to Modern Techniques of Plant Analysis. Third Edition. Chapman and Hall Ltd. Kalidindi, N., Thimmaiah, N.V., Jagadeesh, N.V., Nandeep, R., Swetha, S. and Kalidindi, B. 2015. Antifungal and antioxidant activities of organic and aqueous extracts of Annona squamosa Linn. leaves. J. Food Drug Anal. 23(4), 795–802. Kuna, L., Jakab, J., Smolic, R., Raguz-Lucic, N., Vcev, A. and Smolic, M. 2019. Peptic ulcer disease: a brief review of conventional therapy and herbal treatment options. J. Clin. Med. 8(2), 179. Lambert, A.A., Lam, J.O., Paik, J.J., Ugarte Gil, C., Drummond, M.B. and Crowell, T.A. 2015. Risk of community-acquired pneumonia with outpatient proton-pump inhibitor therapy: a systematic review and meta-analysis. PLoS One 10(6), 1–18. Luna, L.G. 1968. Manual of histologic staining methods of the Armed Forces Institute of Pathology. McGraw-Hill, New York: Blakiston Division. Mahima, R., Rupanshi, G., Sidra, Z., Shrashti, S., Karishma, V., Shweta, T. and Kirti. 2020. A review on biochemical and pharmacological property of Annona squamosa. Inter. J. Multi. Educ. Res. 5(2), 43–48. Malfertheiner, P., Mégraud, F., O’Morain, C., Bazzoli, F., El-Omar, E., Graham, D.Y. and Xiao, S. 2007. Current concepts in the management of helicobacter pylori infection: the maastricht III consensus report. Gut. 56(6), 772–781. Mei, X., Xu, D., Xu, S., Zheng, Y. and Xu, S. 2012. Novel role of Zn(II)–curcumin in enhancing cell proliferation and adjusting proinflammatory cytokine-mediated oxidative damage of ethanol-induced acute gastric ulcers. Chem. Biol. Inter. 197(1), 31–39. Morsy, M.A., Heeba, G.H., Abdelwahab, S.A. and Rofaeil, R.R. 2012. Protective effects of nebivolol against cold restraint stress-induced gastric ulcer in rats: Role of NO, HO-1, and COX-1,2. Nitric Oxide Biol. Chem. 27(2), 117–122. Naji, H.A., Rhiyf, A.G. and Al-Zebeeby, A. 2018. Protective features of Myrtus communis leaves against the genotoxic effects of arsenic in Wistar rats. JPSR 10(11), 2921–2923. Nworgu, C., Celestine, A., Ugwuishi, E., Okorie, P., Anyaeji, P., Ugwu, P. and Nwachukwu, D. 2019. Evaluation of the cytoprotective effects of antiulcer agents in acid-alcohol induced gastric ulceration in wistar rats. J. Physiol. Pathophysiol. 10(1), 10–16. Othman, L., Sleiman, A. and Abdel-Massih, R.M. 2019. Antimicrobial activity of polyphenols and alkaloids in middle eastern plants. Front. Microbiol. 10, 911. Paikray, E., Chawala, S., Bathrachalam, C. and Jena, S. 2024. Protective effect of goat milk on alcohol-induced peptic ulcer in Balb/C mice. JPTCP 31(1), 2230–2234. Park, H., Seo, C.-S., Baek, E.B., Rho, J., Won, Y.-S. and Kwun, H. 2021. Gastroprotective Effect of myricetin on ethanol-induced acute gastric injury in rats. Evid. Based Complement. Alternat. Med. 2021, 1–9. Paz, M.F.C.J., De Alencar, M.V.O.B., De Lima, R.M.I.P., Sobral, A.L.P., Do Nascimento, G. T.M., Dos Reis, C.A. and Melo Cavalcante, A.A.D.C. 2020. Pharmacological effects and toxicogenetic impacts of omeprazole: genomic instability and cancer. Oxid. Med. Cell Longev. 2020, 3457890. Périco, L.L., Emílio-Silva, M.T., Ohara, R., Rodrigues, V.P., Bueno, G., Barbosa-Filho, J. M. and Hiruma-Lima, C.A. 2020. Systematic analysis of monoterpenes: advances and challenges in the treatment of peptic ulcer diseases. Biomolecules 10(2), 265. Ravisankar, P., Koushik, O.S., Reddy, A.A., Eswar Kumar, U., Anvith, P.S. and Pragna, P. 2016. A detailed analysis on acidity and ulcer in esophagus, gastric and duodenal ulcers and management. IOSR J. Dental Med. Sci. 15(1), 2279–2861. Rider, P., Carmi, Y., Voronov, E. and Apte, R. 2013. Interleukin-1α. Semin. Immunol. 25(6), 430–438. Safira, A., Widayani, P., An-Najaaty, D., Mahesa Rani, C.A., Septiani, M., Syah Putra, Y.A. and Raharjo, H.M. 2022. A review of an important plants: Annona squamosa leaf. Pharmacol. J. 14(2), 456–463. Salmerón-Manzano, E., Garrido-Cardenas, J.A. and Manzano-Agugliaro, F. 2020. Worldwide research trends on medicinal plants. Int. J. Environ. Res. Public Health 17(10), 3376. Sharma, P. 2023. Over 30 years of omeprazole. Assoc. Physic. India. 71(8), 11–12. Shivhare, V.D. and Vinode, P. 2023. Role of Indian herbal medicines and nitrogen imidazole derivatives in the management of ulcer. Adv. Pharma. J. 8(6), 155–164. Takagi, K. and Okabe, S. 1968. The effects of drugs on the production and recovery processes of the stress ulcer. Japan. J. Pharma. 18(1), 9–18. Waterman, P.G. 1993. Phytochemical dictionary. A handbook of bioactive compounds from plants. Biochem. System. Ecol. 21(8), 849. Xu, H., Yao, H., Jiang, Z., Wu, X., Chen, Z., Hu, W. and Wang, Y. 2021. Gastric ulcer and traditional Chinese medicine. Vascular Dis. Therap. 6(2), 1–5. Yadav, D.K., Singh, N., Dev, K., Sharma, R., Sahai, M., Palit, G. and Maurya, R. 2011. Anti-ulcer constituents of Annona squamosa twigs. Fitoterapia 82(4), 666–675. Zahid, M., Mujahid, M., Singh, K., Farooqui, S., Singh, K., Parveen, S. and Arif, M. 2018. Annona squamosa Linn. (Custard Apple): an aromatic medicinal plant fruit with immense nutraceutical and therapeutic potentials. Inter. J. Pharm. Sci. Res. 9(5), 1745. | ||
| How to Cite this Article |
| Pubmed Style Al-zubaidi HJ, Mohammed MT, Al-zebeeby A, Alkaby AA, Hassan WR. Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosion. Open Vet. J.. 2025; 15(2): 795-803. doi:10.5455/OVJ.2025.v15.i2.28 Web Style Al-zubaidi HJ, Mohammed MT, Al-zebeeby A, Alkaby AA, Hassan WR. Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosion. https://www.openveterinaryjournal.com/?mno=227385 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.28 AMA (American Medical Association) Style Al-zubaidi HJ, Mohammed MT, Al-zebeeby A, Alkaby AA, Hassan WR. Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosion. Open Vet. J.. 2025; 15(2): 795-803. doi:10.5455/OVJ.2025.v15.i2.28 Vancouver/ICMJE Style Al-zubaidi HJ, Mohammed MT, Al-zebeeby A, Alkaby AA, Hassan WR. Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosion. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 795-803. doi:10.5455/OVJ.2025.v15.i2.28 Harvard Style Al-zubaidi, H. J., Mohammed, . M. T., Al-zebeeby, . A., Alkaby, . A. A. & Hassan, . W. R. (2025) Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosion. Open Vet. J., 15 (2), 795-803. doi:10.5455/OVJ.2025.v15.i2.28 Turabian Style Al-zubaidi, Hanadi J., Mohammed T. Mohammed, Aoula Al-zebeeby, Azhar A. Alkaby, and Wurood Razzaq Hassan. 2025. Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosion. Open Veterinary Journal, 15 (2), 795-803. doi:10.5455/OVJ.2025.v15.i2.28 Chicago Style Al-zubaidi, Hanadi J., Mohammed T. Mohammed, Aoula Al-zebeeby, Azhar A. Alkaby, and Wurood Razzaq Hassan. "Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosion." Open Veterinary Journal 15 (2025), 795-803. doi:10.5455/OVJ.2025.v15.i2.28 MLA (The Modern Language Association) Style Al-zubaidi, Hanadi J., Mohammed T. Mohammed, Aoula Al-zebeeby, Azhar A. Alkaby, and Wurood Razzaq Hassan. "Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosion." Open Veterinary Journal 15.2 (2025), 795-803. Print. doi:10.5455/OVJ.2025.v15.i2.28 APA (American Psychological Association) Style Al-zubaidi, H. J., Mohammed, . M. T., Al-zebeeby, . A., Alkaby, . A. A. & Hassan, . W. R. (2025) Exploring the potential protective and anti-inflammatory effects of the crude ethanolic extract of Annona squamosa Linn fruit against Ethanol-mediated gastric erosion. Open Veterinary Journal, 15 (2), 795-803. doi:10.5455/OVJ.2025.v15.i2.28 |