| Research Article | ||
Open Vet. J.. 2025; 15(2): 835-846 Open Veterinary Journal, (2025), Vol. 15(2): 835-846 Research Article Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and Selenium nanoparticlesMayada A.M. Abou Zeid1, Lamiaa Okasha1, Yamen Hegazy2,3 and Mohamed Abdelmegeid2,4*1Bacteriology , Kafrelsheikh Regional Laboratory, Animal Health Research Institute, Agricultural Research Center (ARC), Giza, Egypt 2Department of Animal Medicine, Faculty of Veterinary Medicine, Kafrelsheikh University, Kafrelsheikh, Egypt 3Clinical Studies Department, College of Veterinary Medicine, King Faisal University, Al-Ahsa 31982, Saudi Arabia 4Higher Colleges of Technology, Sharjah, UAE *Corresponding Author: Mohamed Abdelmegeid. Department of Animal Medicine, Faculty of Veterinary Medicine, Kafrelsheikh University, Kafrelsheikh, Egypt; Veterinary Program, Faculty of Health Sciences, Higher Colleges of Technology, Sharjah Men’s Campus, Sharjah, United Arab Emirates. Email: drmak3351 [at] gmail.com Submitted: 09/11/2024 Accepted: 09/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
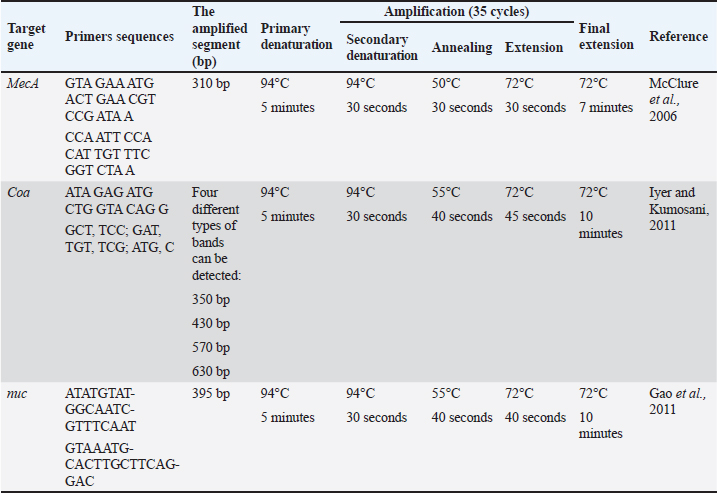
AbstractBackground: Bovine subclinical mastitis is a significant disease that significantly impacts the health of dairy cows and subsequently affects milk production. Among the various causes of subclinical mastitis, bacterial infections, particularly Staphylococcus aureus (S. aureus), play a crucial role. Aim: The aim of this investigation was to isolate and identify S. aureus strains from cows with subclinical mastitis, assess their antimicrobial susceptibility, virulence genes, and investigate the potential antibacterial activity of Moringa oleifera ethanolic extract and Selenium (Se) nanoparticles against both somatic cell count (SCC) and S. aureus count in subclinical mastitic cows. Methods: We collected 110 milk samples from dairy cows with subclinical mastitis in the Kafrelsheikh governorate before treatment with intramammary antibiotics. Samples were identified using pH detection, SCC at ≥200,000 cells/ml, and California Mastitis Test. Results: The effect of M. oleifera ethanolic extract, Se nanoparticles, and Ciprofloxacin antibiotic in reducing SCC and S. aureus counts was significant. Selenium nanoparticles and Ciprofloxacin antibiotics showed the highest reduction in the S. aureus count, comparable to M. oleifera ethanolic extract, while ciprofloxacin followed by M. oleifera ethanolic extract, and Selenium nanoparticles were effective against somatic cell count. Conclusion: This study emphasizes the widespread occurrence of S. aureus in subclinical bovine mastitis and highlights the potential of M. oleifera ethanolic extract and Se nanoparticles as promising treatment options. Selenium nanoparticles demonstrated notable antibacterial effects against S. aureus and contributed to a reduction in somatic cell counts. Considering these findings, we propose additional studies on the potential of Selenium nanoparticles as antibacterial agents, as they demonstrate effectiveness in preventing and managing S. aureus subclinical mastitis. Keywords: mecA gene, MRSA, Moringa oleifera, Staph aureus, Selenium nanoparticles, Subclinical mastitis. IntroductionMastitis, both in its clinical and subclinical forms, is a detrimental disease that significantly impacts the health of dairy animals, leading to reduced milk quality and quantity (Aamir et al., 2009). It is a complex condition influenced by three major factors: host resistance, causative agents, and environmental factors (Al Emon et al., 2024). The causative agents of mastitis typically include Gram-negative and Gram-positive bacteria, mycoplasmas, yeasts, and algae (Bhatt et al., 2012). Staphylococcus aureus is one of the main causes of contagious subclinical mastitis in cattle, causing severe economic losses (Farabi et al., 2024). It is also accountable for foodborne intoxication transmitted to humans via the ingestion of contaminated milk (Ewida and Al-Hosary, 2020). The pathogenesis of Staphylococcus aureus, including intracellular parasitism, biofilm formation, and increasingly severe antimicrobial resistance, renders traditional treatment methods ineffective (Algharib et al., 2020). One type of S. aureus mastitis is methicillin-resistant S. aureus (MRSA), which was initially introduced as a hospital-acquired (HA-MRSA) infection in humans (Deurenberg et al., 2007). Recently, this pathogen emerged as livestock-acquired MRSA (LA-MRSA) (Ahangari et al., 2017), causing serious concern due to difficulties in therapeutic trials at both the human and livestock levels. Milk samples from two Egyptian governorates—Assiut and El-Mansoura—contained methicillin-resistant S. aureus (Elshall, 2019). One of the most important problems associated with MRSA-induced mastitis is its controversial susceptibility to antibiotic therapy due to the cross-resistance of these isolates to most beta-lactams (Hoque et al., 2023). Furthermore, some MRSA isolates are multidrug-resistant against many other antimicrobial families. In addition, it poses a potential threat to the selection and dissemination of resistance genes (Khazaie and Ahmadi, 2021). The use of Selenium (Se) nanoparticles has become popular because of their potential applications as antioxidants, antimicrobials, and enzyme inhibitors (Ramamurthy et al., 2013). Selenium helps heal udder epithelial cells inflamed by S. aureus by inhibiting the activity of nuclear factor kappa B, mitogen-activated protein kinase, and toll-like receptor 2 signaling pathways, while also promoting the expression of microRNA-146a (Wang et al., 2018). Moringa oleifera leaves have recently been recognized as a valuable source of nutrition with various beneficial activities. These leaves have promising antitumor, anti-inflammatory, antiulcer, antiatherosclerotic, and anticonvulsant properties (Dahiru et al., 2006). Researchers have also explored the antimicrobial properties of plant-based compounds, including M. oleifera, as potential therapeutic alternatives (Bugno et al., 2007). The investigation of alternative treatments, such as Se nanoparticles and M. oleifera extracts, shows great potential for combating subclinical mastitis and improving the overall health of dairy animals. Understanding the antimicrobial properties and effects of subclinical mastitis-causing microorganisms, including their susceptibility and virulence factors, can provide valuable insights for developing effective treatment strategies. Thus, this study aimed to estimate the prevalence of S. aureus subclinical mastitis among apparently healthy animals, identify the antibiotic resistance scheme of the isolated strains, and explore the in-vivo antibacterial efficacy of Se nanoparticles and M. oleifera extract on S. aureus subclinical mastitis, contributing to the expansion of novel beneficial methods for managing subclinical mastitis in dairy animals. Material and MethodsThis study was conducted according to research ethics approved by the committee on research of the College of Veterinary Medicine, Kafr-Elsheikh University, Kafr El-Shaikh, Egypt (KFS-IACUC/95/2019). Clinical examinationClinical examination of the cows’ udders and regional lymph nodes was performed before sampling, as previously described (Constable et al., 2016). Milk samplesA total of 110 samples of milk from subclinical mastitic animals were collected after pH detection. Subclinical mastitis was detected using somatic cell count (SCC) and the California Mastitis Test (CMT). SCC was determined by automated counters with a threshold of ≥200,000 cells/ml to indicate subclinical cases, consistent with European guidelines. CMT was performed by mixing milk samples with a reagent on a paddle, with gel formation indicating elevated somatic cell counts in response to intramammary inflammation, following the method described by Ebrahimie et al. (2018). The samples were collected from dairy cow farms at the Kafrelsheikh governorate before the cows were treated with intramammary antibacterial agents. Before collecting milk samples, milkers thoroughly cleaned the udders, teat orifices, and hands with soap and water, followed by disinfection using 70% ethyl alcohol. The initial milk streams were disposed of, and approximately 20 ml of milk was kept in clean, sterile bottles, each labeled with a unique serial number. All samples were sent directly to the laboratory for bacteriological examination (Al Emon et al., 2024; Farabi et al., 2024). Bacteriological examinationsA cow was considered to be subclinically infected after counting somatic cells, detecting pH, and indicating positivity for subclinical mastitis (SCM). Milk pH was measured at 37°C using a digital pH meter (modelAdwa1030, Romania) after calibration of the pH probe immediately after collecting milk samples, with beyond 6.8 indicating the prevalence of subclinical mastitis (Modh et al., 2019). Furthermore, the SCC cut-off was employed to determine the frequency of SCM as having at least 200,000 cells/ml with or without positive isolation of the udder pathogen and when the cow was presented with no clinical signs (Ebrahimie et al., 2018). Isolation and identification of S. aureusMilk samples were collected aseptically and centrifuged at 3,000 rpm for 20 min, after which the supernatant was discarded. A loopful of the sediment was then inoculated into Tryptic soy broth and aerobically incubated at 37°C for 18–24 hours. Subsequent subculturing was performed on Baird-Parker selective agar plates incubated aerobically at 37°C. Plates were examined after 24 hours and again at 48 hours for the presence of black, shiny, convex colonies with narrow white margins and surrounding clear zones (Baird-Parker, 1969). Mannitol salt agar (MSA) (DM160D) (Mast Diagnostics, Merseyside, United Kingdom) was also used to inoculate the samples (Wedley et al., 2014) and incubated at 37°C for 24 hour. Clusters of S. aureus with a yellowish color on MSA plates were again examined for morphological and biochemical characteristics (Cheesbrough, 2006). Antimicrobial susceptibility testingaccording to the guidelines provided by CLSI, 2011, the susceptibility profiles of 10 isolated S. aureus strains to 9 commercially available antibiotic discs were determined using the disk diffusion technique. The following antibiotic discs were used in this study: Amoxicillin clavulanic acid (AMC) 30 g, Sulfa trimethoprim (COT) 25 g, methicillin (ME) 5 g, cefotaxime 30 g, tetracycline (TE) 30 g, fosfomycin 200 g, tylosine 15 g, Ciprofloxacin (CIP) 5 g, and gentamicin 10 g. Measurement of millimeter-sized inhibitory zones that formed around the discs allowed us to ascertain the antibiotic sensitivity. A sensitivity, moderate, or resistant classification was given to the zone diameters. Detection of S. aureus virulence genes by using polymerase chain reaction (PCR)DNA was extracted from the samples using a QIAamp DNA Mini Kit (Qiagen, Germany, GmbH) with minor modifications. For 10 minutes, 200 µl of the sample suspension was treated with 20 µl of proteinase K and 200 µl of lysis buffer at 56°C. After incubation, 200 µl of 100% ethanol were added to the lysate. The samples were washed and centrifuged according to the manufacturer’s instructions. Finally, 100 µl of the kit elution buffer was used to elute the nucleic acids. Table 1 lists Metabion (Germany) primers. The PCR reaction was conducted in a 25-µl volume with 12.5 µl EmeraldAmp Max PCR Master Mix (Takara, Japan), 1 µl of each primer (20 pmol), 5.5 µl water, and 5 µl DNA template. The reaction was performed on an Applied Biosystems 2720 thermal cycler. The PCR products were electrophoresed on a 1.5% agarose gel (Applichem, Germany, GmbH) in 1x Tris-Borate-EDTA Buffer (TBE) buffer at room temperature with a 5 V/cm gradient. For analysis, 20 µl of uniplex PCR products and 30 µl of multiplex products were loaded into each gel slot. Fragment sizes were determined using a Fermentas 100-bp gene ruler ladder (Thermo, Germany). The gel data were analyzed by computer software from Alpha Innotech and the Biometra gel documentation system. Moringa oleifera ethanolic extractMoringa oleifera leaves were shade-dried and ground into a powder. The hydroethanolic extract was prepared by dissolving 500 g of this powder in 2 L of 80% ethanol at room temperature with occasional shaking. After three days, the mixture was filtered using sterile filter paper (No. 1. The ethanolic extract was concentrated and evaporated below 40°C to form a semisolid mass. This extract was reconstituted in sterile phosphate-buffered saline (PBS, pH 7.4) at a concentration of 1-g extract per 10 ml of PBS. The reconstituted extract was filtered through a 0.22 µm membrane (Millipore, India) and stored in a sterile vial under refrigeration for intramammary infusion. The dosage was determined based on prior research findings (Sharma and Sharma, 2022). Pharmaceutical compounds in Moringa oleifera ethanolic extractAssessing the total flavonoid concentration Following Marinova et al. 2005, a modified AlCl3 colorimetric method was used to measure flavonoid content. In a 10-ml volumetric flask, 1 ml of the extract was dissolved in 2 ml of methanol. Solutions of 5% NaNO3, 5% NaOH, and 7% AlCl3 were prepared in a 25 ml flask. Then, 200 µl of the extract was placed in a sealed glass vial, mixed with 75 µl of 5% NaNO3, and left for 5 minutes at room temperature. Subsequently, 1.25 ml of AlCl3 and 0.5 ml of NaOH were added to each vial, followed by sonication and incubation for 5 minutes. With the use of a quercetin standard calibration curve, the flavonoid content was quantified as micrograms of quercetin equivalent per gram of dry extract. The absorbance of each solution was measured at 510 nm. Total phenolic contentBy employing a colorimetric technique with the Folin-Ciocalteu reagent, the total phenolic content of the extracts was determined. One milliliter of the extract was dissolved in two milliliters of methanol. Subsequently, 2.5 ml of 75 g/l sodium carbonate and 500 µl of Folin-Ciocalteu reagent diluted 10 times were mixed with 2.5 ml of this solution. Before allowing the mixture to sit for 2 hours at 25°C, it was vortexed for 10 seconds. The absorbance was measured at 765 nm compared with a blank reagent after incubation. Gallic acid equivalent milligrams per gram of dry extract was used as the unit of expression for the results Marinova et al. 2005. Total alkaloid contentFollowing (Shamsa et al., 2008), to prepare the bromocresol green (BCG) solution, 69.8 mg of BCG was dissolved in 3 ml of 2 N NaOH and 5 ml of distilled water, and then brought to a volume of 1,000 ml with distilled water. A phosphate buffer (pH 4.7) was created by adjusting the pH of a 2M sodium phosphate solution with citric acid. The atropine standard solution was prepared by dissolving 10 mg of atropine in 10 ml of distilled water. Aliquots (5–60 µg/ml) of this solution were mixed with 5 ml of phosphate buffer and 5 ml of BCG solution, then shaken with chloroform in increments. The extracted solutions were collected and diluted with chloroform, and their absorbance was recorded at 470 nm. For plant extraction, 5 g of the plant material was finely ground and extracted with methanol at 26°C over 24 hours, followed by concentration under vacuum at 45°C. A portion of the residue was dissolved in 2N HCl, filtered, neutralized with NaOH, and then combined with 5 ml of BCG and 5 ml of buffer. The solution was shaken and extracted with chloroform, and its absorbance was measured at 470 nm. Determination of total saponins content (TSC)Following (Le et al. (2018), a standard curve for aescin was prepared by dissolving 150 mg of aescin in 10 ml of methanol (15 mg/ml stock) and performing serial dilutions in triplicate using methanol as the blank. For extraction, 1 g of powder was mixed with 30 ml of ethanol, left for 30 minutes at room temperature, cooled in an ice bath, and filtered through a 0.45 µm membrane. This clear extract was used to determine TSC. Samples were heated at 65°C to evaporate methanol, treated with 0.5 ml of 4% vanillin and 2.5 ml of 72% H2SO4, incubated at 60°C for 15 minutes, and cooled. The absorbance at 560 nm was measured, and the values were plotted for the standard curve. TSC was calculated as: text{Total saponin content}=frac{text{weight of saponins in extract (mg)}}{text{weight of powder (g)} times (1 - frac{text{moisture %}}{100})}and expressed as (mg AE/g). Table 1. Primer sequences, target genes, amplicon sizes, and cycling conditions.
Total tannins assessmentTannin content was measured using tannic acid as a reference. For this, 400 µL of extract was mixed with 3 ml of 4% vanillin solution in methanol and 1.5 ml of concentrated hydrochloric acid. After 15 min of incubation, absorbance was recorded at 500 nm (Broadhurst and Jones, 1978). Preparation of Selenium nanoparticlesOne gram of sodium selenite was dissolved in 100 ml of double-distilled water until a clear solution was obtained. Then, 0.5 ml of hydrazine hydrochloride was added as a reducing reagent. The mixture solution was left to react by stirring for 1 hour, then heated for 1 hour, and a red precipitate formed. The Se nanoparticles were centrifuged, washed, and dried. The characterization of Se nanoparticles was performed using various techniques, such as X-Ray diffraction (XRD), scanning electron microscopy, and Zeta potential (Chudobova et al., 2014; Fig. 1). Experimental designThe experimental design was completely randomized using three intramammary treatments. Ten subclinically dairy cows selected based on CMT, SCC, and S. aureus examination were involved as three groups (Group 1: n=3 for M. oleifera ethanolic extract; Group 2: n=3 Se nanoparticles and Group 3: n=4 for Ciprofloxacin antibiotic treatment according to in vitro sensitivity test). Concerning Group 1, an ethanolic extract of M. oleifera (1 g/10 ml of PBS) was prepared according to Sharma and Sharma (2022) and was infused in the intramammary once daily for 3 days; the Nano-Se-treated group cows (Group 2) were infused in the intramammary with nano-Se at a concentration of (500 µg/ml) according to Hassan et al. (2019). Furthermore, the third group (antibiotic) commercial CIP (Ciprolac® 100 mg intramammary antimicrobial syringe; Ourofino Animal Health) was chosen for this study after passing an in vitro sensitivity test using the gold standard—the disc diffusion method since 100 mg intramammary was infused once daily for 3 days. Milk samples were collected before treatment as a control group and on the 1st, 2nd, and 3rd days after therapy with M. oleifera extract, Se nanoparticles, and Ciprofloxacin. Bacteriological examination of milk samples before and after treatment was performed for the control and three treatment groups by counting the number of S. aureus colonies. A colony counter was used to count the SCC, and the values were expressed as Colony-forming units (CFU)/ml.
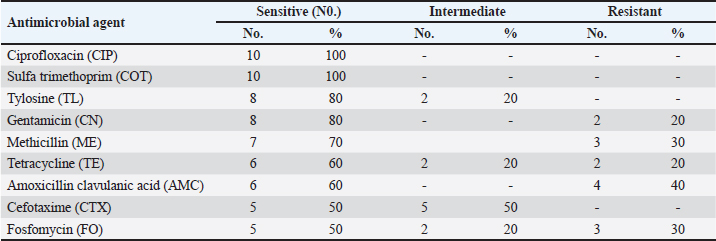
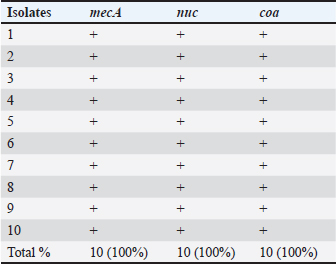
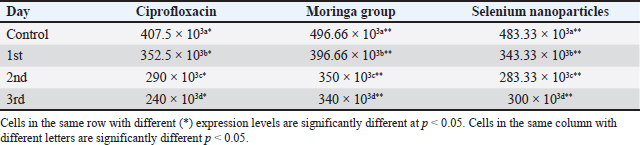
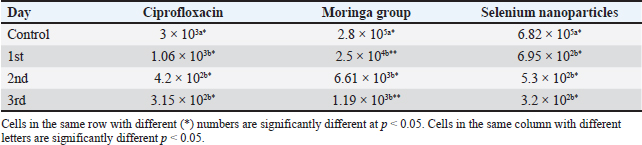
Fig. 1. Shows the Se nanoparticles’ scanning electron microscopy (SEM). The size and morphology of Selenium (Se) nanoparticles were studied by placing a small sample on a holder and measuring the average particle size of the synthesized Se nanorods. The high-resolution SEM images of the fabricated Se nanoparticles were acquired at different magnifications to perform the measurements. The Se nanostructure appeared nanospherical in the images, with an almost uniform morphology and width of approximately 30 nm. Statistical analysisTwo-way repeated measures analysis of variance was conducted to evaluate the effects of different treatment groups across various examination days on somatic cell counts and CFUs per ml. The statistical analysis was performed using IBM SPSS Statistics 23 (IBM, Armonk, NY). ResultsPrevalence of S. aureus in Bovine subclinical mastitis samplesA total of 110 milk samples were obtained after investigation of subclinical mastitis cases using somatic cell count (≥200 000 cells/ml), pH (≥ 6.8), and positive CMT, yielding 10 S. aureus-positive isolation samples. Antibiotic susceptibility analyses of the S. aureus isolates showed that the most susceptible to Ciprofloxacin CIP and COT (100%) were followed by tylosin and gentamicin (80%) (Table 2). Only three out of ten isolates were identified as MRSA based on the methicillin-resistant disk diffusion method and confirmed by the presence of mecA gene, although the remaining seven isolates were negative for methicillin-resistant disk diffusion but carried mecA gene as presented in Tables 2 and 3 and Fig. 2. All of the ten isolates were carrying nuc and coa genes as presented in Table 3 and Figs. 2, 3, and 4. The effect of M. oleifera ethanolic extract, Se nanoparticles, and Ciprofloxacin antibiotics on reducing somatic cell count cells/ml, as shown in Table 5, the highest significant decrease in somatic cell counting at p < 0.05 was achieved in the CIP antibiotic-treated group. The other two groups did not show a significant difference at p < 0.05. Furthermore, there was a substantial difference at p < 0.05 in the somatic cell counts between days of treatment, where the highest number of cells was in control a and the lowest was in the 3rd day of treatment in all 3 treatments. The effects of M. oleifera ethanolic extract, Se nanoparticles, and Ciprofloxacin antibiotics on S. aureus counting are shown in Tables 5 and 6, which showed that the highest significant decrease in S. aureus counting at p < 0.05 was achieved in the Se nanoparticle-treated group and Ciprofloxacin antibiotic-treated group than in the moringa-treated group. However, there was a significant difference between day 0 and the other 3 days of treatment in the Ciprofloxacin, moringa extract, and Se nanoparticle groups. The findings presented in Table 6 demonstrate the antibacterial effects of M. oleifera ethanolic extract, Se nanoparticles, and Ciprofloxacin against S. aureus counts (CFU/ml) over the treatment period. In the control group, the initial S. aureus counts were 3 × 10³ CFU/ml, 2.8 × 105 CFU/ml, and 6.82 × 105 CFU/ml for the Ciprofloxacin, M. oleifera, and Se nanoparticle groups, respectively, highlighting the severity of infection. CIP showed the most consistent reduction in bacterial counts, decreasing significantly to 3.15 × 10² CFU/ml by the third day. Selenium nanoparticles demonstrated comparable efficacy, reducing bacterial counts to 3.2 × 10² CFU/ml by the third day, suggesting that they are an effective alternative to Ciprofloxacin. Moringa oleifera extract also significantly reduced S. aureus counts, but the reduction (to 1.19 × 10³ CFU/ml by the third day) was less pronounced compared with the other treatments. The statistical analysis revealed significant differences in bacterial reduction between treatments (p < 0.05) and across treatment days (p < 0.05), emphasizing the superior effectiveness of Ciprofloxacin and Se nanoparticles. These results suggest that Se nanoparticles represent a promising alternative therapy for managing S. aureus infections in subclinical mastitis. Table 2. Antimicrobial susceptibility test for S. aureus isolates (no=10).
Table 3. Prevalence of mecA, nuc, and coa viral genes in S. aureus isolates from subclinical mastitis cases.
DiscussionSubclinical mastitis is responsible for substantial monetary losses in dairy farming, and S. aureus is considered the most frequent bacterial species responsible for both clinical and subclinical types of mastitis in cattle: clinical and subclinical (Neelam et al., 2022). Moreover, MRSA is a type of S. aureus infection and is considered an emerging problem in dairy farms worldwide, including Egypt (Ewida, 2009). MRSA is resistant to commonly used antimicrobials, and it possesses virulent factors, such as enterotoxins and hemolysins, which have public health consequences. This highlights the importance of continuous surveillance for estimating the extent of the problem in dairy farms, controlling antimicrobial resistance, and prescribing appropriate antimicrobial agents in both veterinary holdings and public health hospitals. In the existing study, upon conducting a bacteriological examination of the subclinical milk samples, we found that S. aureus is responsible for almost 10% of SCM in dairy farms in the study area. In contrast, a study by (Ewida and Al-Hosary, 2020) reported higher results, with S. aureus detected in 46 out of 48 samples (95.83%) that were positive for CMT. Moreover, our findings demonstrated a lower prevalence than previous research in Benha Province, which found S. aureus in 90.4% of milk samples, and in Dakahlia Province, it was found in 81.3% (Adwan and Isayed, 2018). The high prevalence of S. aureus in these studies may be ascribed to inadequate hygiene before and after milking and the prevailing environmental conditions. Antimicrobial resistance poses a significant global challenge. Therefore, the susceptibility of the recovered S. aureus isolates to commonly used antimicrobials was examined. Table 2 shows that the isolates were vulnerable to the antimicrobials tested. The isolates displayed 100% sensitivity to Ciprofloxacin and COT. Moreover, the strains exhibited 80% sensitivity to tylosin and gentamicin, 40% were resistant to Amoxicillin clavulanic acid (AMC), and 30% were resistant to Methicillin (ME). They also found similar results (El-Azzouny et al., 2022), who observed that S. aureus isolates showed sensitivity to Ciprofloxacin (96%) and gentamicin (92%), and by (Saidi et al., 2019), who found that S. aureus isolates exhibited sensitivity to Tetracycline (TE) (65.71%) and Amoxicillin clavulanic acid (AMC) (51.42%). This study identified a high rate of MRSA discrepancies, with phenotypically MRSA-negative isolates testing positive for mecA by PCR. These findings suggest that reliance solely on mecA expression alone is insufficient for accurately distinguishing MRSA or MSSA. Another gene, similar to mecA, also contributes to methicillin resistance (Paterson et al., 2014). Another study reported that a group of genes located on the staphylococcal chromosomal cassette mec, which enhances antibiotic resistance, can lead to inaccurate results in disc diffusion tests (Aqib et al., 2018). The occurrence of MRSA in dairy farms is likely due to the widespread use of antimicrobials, which has driven the development of antimicrobial resistance in bacterial pathogens. Antibiotic administration is strongly associated with the emergence and persistence of resistance traits in pathogenic strains. These traits are encoded by specific genes located on the bacterial chromosome, plasmids, transposons, or gene cassettes within integrons (Daka et al., 2012). Consequently, isolates can easily transfer these genes (Hassan et al., 2016).
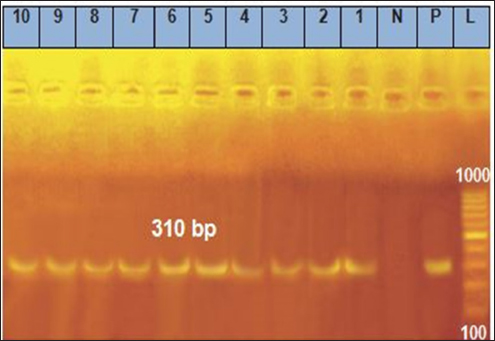
Fig. 2. Agarose gel electrophoresis of the polymerase chain reaction result for mecA virulence. Lanes 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10: Positive for mecA gene with 310 bp band.
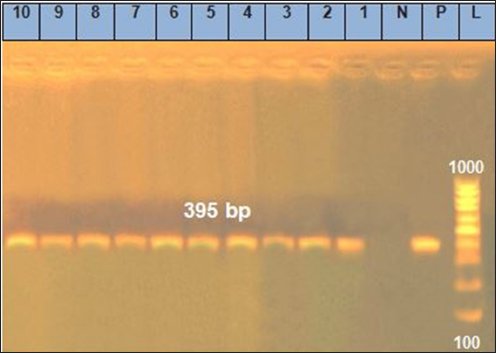
Fig. 3. Agarose gel electrophoresis of the polymerase chain reaction result for nuc virulence. Lanes 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10: Positive for nuc gene with 395 bp band.
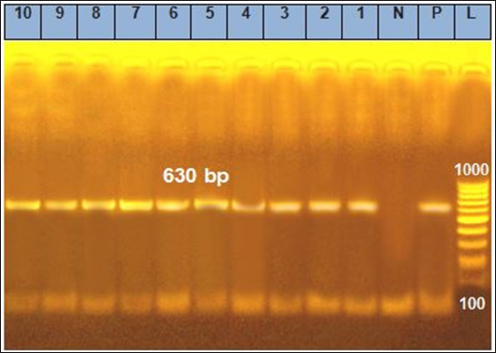
Fig. 4. Agarose gel electrophoresis of the polymerase chain reaction result for coa virulence gene. Lanes 1, 2, 3, 4, 5, 6, 7, 8, 9, and 10: Positive for coa gene with 630 bp band. Table 4. Determination of total phenolic, flavonoid, and alkaloid content, total saponin content, total alkaloid content, and total tannin content of Moringa oleifera (ethanolic extract) (mean ± SE).
Table 5. Effects of Moringa oleifera, Selenium nanoparticles, and Ciprofloxacin on the mean Somatic cell count cells ml.
Table 6. Effects of Moringa oleifera, Selenium nanoparticles, and Ciprofloxacin antibiotics on S. aureus count: CFUs per ml.
Molecular identification and detection of virulence genes were performed on the isolates. The screening revealed that all 10 S. aureus isolates carried mecA, nuc, and coa. In contrast, (Ewida and Al-Hosary, 2020) reported lower results, with the nuc virulence gene Confirmed in 33 (71.73%) of the milk samples examined, whereas mecA was not found in this study. These results are consistent with findings from a previous study conducted by (Abd El-Hamid and Bendary, 2013) in Egypt, which confirmed the presence of the nuc gene in all S. aureus strains and differed from the findings of (Al-Ashmawy et al., 2016), who detected only the mecA gene in 53% of the scanned samples, and (El-Tawab et al., 2017), who noticed that S. aureus bacteria taken from mastitic cows tested positive for the coa gene 50% of the time. Moringa plants contain a number of compounds. There are multiple possible medical uses for each plant. The values of total phenolics, flavonoid components, total saponin, total alkaloid, and total tannins in the ethanolic extract of M. oleifera are presented in Table 4. Similar results were reported by (Abadallah and Ali, 2019), who revealed that the leaves’ aqueous and ethanol extracts included alkaloids, saponins, flavonoids, reducing sugar, glycosides, quinones, tannin, and terpenoids. A vast class of nitrogenous chemicals known as alkaloids has been extensively applied as chemotherapeutic agents against cancer, as well as anesthetics and CNS stimulants (Madziga et al., 2010). Alkaloids regulate the growth and metabolism of living things (Edeoga et al., 2006). A study by (Salah et al., 1995) validated the antioxidant and antibacterial capabilities of flavonoids. Flavonoids have the potential to prevent tumor invasion and kill cancer cells through the processes they induce (Benavente-García et al., 1997). Also, (Arabski et al., 2012) noted that saponins possess noteworthy biological properties, including cytotoxic, hemolytic, molluscicidal, anti-inflammatory, antifungal, anti-yeast, antibacterial, and antiviral activities. In our study, Tables 5 and 6, we observed that using Ciprofloxacin followed by each Se nanoparticle and M. oleifera ethanolic extract decreased somatic cell counts. In contrast, Se nanoparticles and Ciprofloxacin antibiotics were used before the ethanolic extract of M. oleifera to decrease the S. aureus count. The difference in the results between the somatic cell count and S. aureus count may be attributed to the fact that the components of the somatic cell count are bacterial cells responsible for mastitis, such as S. aureus, as well as the immune cells responsible for defense (Ewida and Al-Hosary, 2020). In a laboratory setting, extracts of M. oleifera leaves in aqueous, ethanol, and methanol solutions were found to slow down the expansion of certain bacteria (Singh and Tafida, 2014). Different findings were informed by (Hassan et al., 2019), who reported that the ethanol and methanol extracts showed higher antibacterial activity than the antibiotic extract, with the ethanolic extract showing the strongest antimicrobial action against the bacteria tested. This suggests that plants could potentially be used to source new antibiotic compounds. Studies have shown that Se nanoparticles can interact with the surface of bacterial cell membranes, disrupting their permeability and respiratory function. They can also penetrate bacteria or fungi, making them a potential alternative to antibiotics and aiding in overcoming antibiotic resistance (Hassan et al., 2019). Proof of the potent suppressive effect of Se nanoparticles on S. aureus in vitro growth, with a declining percentage of viable bacteria detected in the presence of Se nanoparticles at different doses and over time (Tran and Webster, 2011). ConclusionIn conclusion, Staph aureus is a significant causative agent of subclinical bovine mastitis. Antimicrobial susceptibility testing revealed that Ciprofloxacin and COT are effective against S. aureus. Furthermore, all 10 isolates carried the virulence genes mecA, nuc, and coa, while three isolates were methicillin-resistant using the disk diffusion method, which has implications for public health. MRSA and the potential pollution of milk with this bacterium pose a severe risk to public health, necessitating sustained efforts to control the infection. Finally, Se nanoparticles and Ciprofloxacin had a better effect on S. aureus counting than M. oleifera ethanolic extracts. In contrast, Ciprofloxacin, followed by M. oleifera ethanolic extracts and Se nanoparticles, effectively reduced somatic cell counts. Based on these findings, we recommend further investigation into the use of Se nanoparticles as antibacterial because they show promise in preventing and treating S. aureus subclinical mastitis. AcknowledgmentsNot applicable. Conflict of interestThe authors declare no competing interests relevant to the content of this article. Competing interestsThe authors declare that they have no competing. Author’s contributionConceptualization: Mayada A.M. Abou Zeid, Lamiaa Okasha, Yamen Hegazy, and Mohamed Abdelmegeid. Data curation and validation: Yamen Hegazy and Mohamed Abdelmegeid. Writing-original draft: Mayada A.M. Abou Zeid and Lamiaa Okasha. Writing-review and editing: Mayada A.M. Abou Zeid, Lamiaa Okasha, and Mohamed Abdelmegeid. Investigation and visualization: Mayada A.M. Abou Zeid, Lamiaa Okasha, and Mohamed Abdelmegeid. Supervision: : Mayada A.M. Abou Zeid, Lamiaa Okasha, Yamen Hegazy, and Mohamed Abdelmegeid. Project administration: Mohamed Abdelmegeid. FundingNo funding, grants, or other support was received. Ethical approvalThis study was conducted according to research ethics approved by the committee on research of the College of Veterinary Medicine, Kafr-Elsheikh University, Kafr El-Shaikh, Egypt (KFS-IACUC/95/2019). Data availabilityAvailable upon request from the authors. ReferencesAamir, S., Ghulam, M. and Muhammad, A. 2009. Mastitis in buffaloes Pakistan. J. Zool. Suppl. (9), 479–490. Abadallah, M.S. and Ali, M. 2019. Antibacterial activity of Moringa oleifera leaf extract against bacteria isolated from patients attending General Sani Abacha Specialist Hospital. J. Allied Pharm. Sci. 1, 61–66. Abd El-Hamid, M. and Bendary, M.M. 2013. Association between agr alleles and toxin gene profiles of S. aureus isolates from human and animal sources in Egypt. Int. J. Adv. Res. 1, 133–144. Adwan, G. and Isayed, H. 2018. Prevalence and characterization of S. aureus isolated from bulk tank milk dairy cow farms in West Bank-Palestine. Microbiol. Res. J. Int. 23, 1–13. Ahangari, Z., Ghorbanpoor, M., Shapouri, M.R.S., Gharibi, D. and Ghazvini, K. 2017. Methicillin resistance and selective genetic determinants of Staphylococcus aureus isolates with bovine mastitis milk origin. Iran. J. Microbiol. 9, 152. Al-Ashmawy, M.A., Sallam, K.I., Abd-Elghany, S.M., Elhadidy, M. and Tamura, T. 2016. Prevalence, molecular characterization, and antimicrobial susceptibility of methicillin-resistant Staphylococcus aureus isolated from milk and dairy products. Foodborne Pathog. Dis. 13, 156–162. Al Emon, A., Hossain, H., Chowdhury, M.S.R., Rahman, M.A., Tanni, F.Y., Asha, M.N., Akter, H., Hossain, M.M., Islam, M.R. and Rahman, M.M. 2024. Prevalence and antimicrobial susceptibility profiles of subclinical bovine mastitis pathogens in Bangladesh. Heliyon 10(14), e34567. Algharib, S.A., Dawood, A. and Xie, S. 2020. Nanoparticles for treating bovine Staphylococcus aureus mastitis. Drug Deliv. 27, 292–308. Aqib, A.I., Ijaz, M., Farooqi, S.H., Ahmed, R., Shoaib, M., Ali, M.M., Mehmood, K. and Zhang, H. 2018. Emerging discrepancies in the conventional and molecular epidemiology of methicillin-resistant Staphylococcus aureus isolated from bovine milk. Microb. Pathog. 116, 38–43. Arabski, M., Węgierek-Ciuk, A., Czerwonka, G., Lankoff, A. and Kaca, W. 2012. Effects of saponins against clinical E. coli strains and eukaryotic cell lines. BioMed Res. Int. 2012, 286216. Baird-Parker, A. 1969. The use of Baird-Parker’s medium for the isolation and enumeration of Staphylococcus aureus. In Isolation methods for microbiologists. Shapton, D.A. and Gould, G.W. (Eds.). Academic Press, London, UK, vol. 3, pp 1–8. Benavente-García, O., Castillo, J., Marin, F.R., Ortuño, A. and Del Río, J.A. 1997. Uses and properties of citrus flavonoids. J. Agric. Food Chem. 45, 4505–4515. Bhatt, V., Ahir, V., Koringa, P., Jakhesara, S., Rank, D., Nauriyal, D., Kunjadia, A. and Joshi, C. 2012. Milk microbiome signatures of subclinical mastitis-affected cattle analyzed by shotgun sequencing. J. Appl. Microbiol. 112, 639–650. Broadhurst, R.B. and Jones, W.T. 1978. Analysis of condensed tannins using acidified vanillin. J. Sci. Food Agric. 29, 788–794. Bugno, A., Nicoletti, M.A., Almodóvar, A.A., Pereira, T.C. and Auricchio, M.T. 2007. The antimicrobial efficacy of Curcuma zedoaria extract compared with commercial mouthrinses. Braz. J. Microbiol. 38, 440–445. Cheesbrough, M. 2006. District laboratory practices in tropical countries, part 2. Cambridge, UK: Cambridge University Press. Chudobova, D., Cihalova, K., Dostalova, S., Ruttkay-Nedecky, B., Merlos Rodrigo, M.A., Tmejova, K., Kopel, P., Nejdl, L., Kudr, J. and Gumulec, J. 2014. Comparison of the effects of silver phosphate and selenium nanoparticles on Staphylococcus aureus growth revealed the potential for selenium particles to prevent infection. FEMS Microbiol. Lett. 351, 195–201. Constable, P.D., Hinchcliff, K.W., Done, S.H. and Grünberg, W. 2016. Veterinary medicine: a textbook on the diseases of cattle, horses, sheep, pigs, and goats. Edinburgh, UK: Elsevier Health Sciences. Dahiru, D., Onubiyi, J. and Umaru, H.A. 2006. Phytochemical screening and antiulcerogenic effects of Moringa oleifera aqueous leaf extract. Afr. J. Tradit. Complement. Altern. Med. 3, 70–75. Daka, D., G/silassie, S. and Yihdego, D. 2012. Antibiotic-resistant Staphylococcus aureus isolated from cows’ milk in the Hawassa area, South Ethiopia. Ann. Clin. Microbiol. Antimicrob. 11, 1–6. Deurenberg, R.H., Vink, C., Kalenic, S., Friedrich, A., Bruggeman, C. and Stobberingh, E. 2007. Molecular evolution of methicillin-resistant Staphylococcus aureus. Clin. Microbiol. Infect. 13, 222–235. Ebrahimie, E., Ebrahimi, F., Ebrahimi, M., Tomlinson, J., South, D. and Petrovski, K.R. 2018. A large-scale study of indicators of subclinical mastitis in dairy cattle by weighting analysis of milk composition features: highlighting the predictive power of lactose and electrical conductivity. J. Dairy Res. 85, 193–200. Edeoga, H., Omosun, G. and Uche, L. 2006. Chemical composition of Hyptis suaveolens and Ocimum gratissimum hybrids from Nigeria. Afr. J. Biotechnol. 5, 1–5. El-Azzouny, M.M., Khater, S.I., Adli, S.H. and Abou-Khadra, S.H. 2022. Antibacterial activities of selenium nanoparticles against multidrug-resistant Staphylococcus aureus and Escherichia coli isolated from mastitic milk. J. Adv. Vet. Res. 12, 768–772. El-Tawab, A., Ammar, A., Hofy, F., Mohamed, S. and Abubakr, H. 2017. Virulence genes of Staphylococcus aureus isolated from mastitic cows. Benha J. Appl. Sci. 2, 89–93. Elshall, S. 2019. Enterotoxins and enterotoxigenic staphylococci in milk and other milk products. Ph.D. Thesis, Assiut University, Faculty of Veterinary Medicine, Assiut, Egypt. Ewida, R. 2009. Some studies on S. aureus in milk and milk products sold in Assiut City with special reference to antibiotic-resistant S. aureus. Ph.D. Thesis, Faculty of Veterinary Medicine, Assiut University, Assiut, Egypt. Ewida, R.M. and Al-Hosary, A.A. 2020. The prevalence of enterotoxins and other virulence genes associated with S. aureus-induced subclinical mastitis in dairy cows. Vet. World 13, 1193. Farabi, A.A., Hossain, H., Brishty, K.A., Rahman, M.H., Rahman, M., Siddiqui, M.S.I., Atikuzzaman, M., Saleh, A., Binsuwaidan, R. and Selim, H.M.R.M. 2024. Prevalence, risk factors, and antimicrobial resistance of Staphylococcus and Streptococcus species isolated from subclinical bovine mastitis. Foodborne Pathog. Dis. PubMed PMID: 39479784; doi:10.1089/fpd.2024.0097. Gao, J., Ferreri, M., Liu, X.Q., Chen, L.B., Su, J.L. and Han, B. 2011. Development of multiplex polymerase chain reaction assay for rapid detection of Staphylococcus aureus and selected antibiotic resistance genes in bovine mastitic milk samples. J. Vet. Diagn. Investig. 23, 894–901. Hassan, A., Mansour, M.K., Nahed, M., Shawky, R.M. and El Ahl, S. 2019. Influence of selenium nanoparticles on the bacterial and fungal causes of mastitis in buffaloes. Anim. Health Res. 7, 76–94. Hassan, F.A., Bayoumi, H.M., El-Gawad, M., Enab, A. and Youssef, Y. 2016. Utilization of Moringa oleifera leaf powder in production of yoghurt. Int. J. Dairy Sci. 11, 69–74. Hoque, M., Azal, M., Chowdhury, E., Hossain, H., Imranuzzaman, M., Islam, M. and Ahad, A. 2023. Prevalence and antibiotic susceptibility profile of S. aureus in clinical and subclinical mastitis milk samples. Bangladesh J. Vet. Med. (BJVM) 21, 27–37. Iyer, A.P. and Kumosani, T.A. 2011. PCR based detection of nosocomial infection causing MRSA (Methicillin resistant Staphylococcus aureus). 2nd International Conference on Biotechnology and Food Science IPCBEE, 7. IACSIT Press, Singapore. Khazaie, F. and Ahmadi, E. 2021. Bovine subclinical mastitis-associated methicillin-resistant Staphylococcus aureus, selective genotyping, and antimicrobial susceptibility profile of the isolates in Kurdistan Province of Iran. Iran. J. Microbiol. 13, 65. Le, V.A., Parks, S.E., Nguyen, H.M. and Roach, P.D. 2018. Improving the vanillin-sulfuric acid method for quantifying total saponins. Technologies 6, 84. Madziga, H., Sanni, S. and Sandabe, U. 2010. Phytochemical and elemental analysis of Acalypha wilkesiana leaf. J. Am. Sci. 6, 510–514. Modh, R., Nauriyal, M.I.D., Modi, R. and Wadhwani, K. 2019. Study on pH and somatic cell counts in subclinical mastitic cow milk in association with udder and teat shape. Indian J. Anim. Prod. Manag. 34, 75–9. McClure, J.A., Conly, J.M., Lau, V., Elsayed, S., Louie, T., Hutchins, W. and Zhang, K. 2006. Novel multiplex PCR assay for detection of the staphylococcal virulence marker Panton-Valentine leukocidin genes and simultaneous discrimination of methicillin-susceptible from -resistant staphylococci. J. Clin. Microbiol. 44, 1141–1114. Neelam, Jain, V., Singh, M., Joshi, V.G., Chhabra, R., Singh, K. and Rana, Y. 2022. Virulence and antimicrobial resistance gene profiles of Staphylococcus aureus associated with clinical mastitis in cattle. PLoS One 17, e0264762. Nisha, A. 2021. Master of veterinary science. Mathura: Pandit Deen Dayal Upadhyaya Pashu Chikitsa Vigyan Vishwavidyalaya. Paterson, G.K., Harrison, E.M. and Holmes, M.A. 2014. Emergence of mecC-resistant Staphylococcus aureus. Trends Microbiol. 22, 42–47. Ramamurthy, C., Sampath, K., Arunkumar, P., Kumar, M.S., Sujatha, V., Premkumar, K. and Thirunavukkarasu, C. 2013. Green synthesis and characterization of selenium nanoparticles and their augmented cytotoxicity with doxorubicin on cancer cells. Bioprocess Biosyst. Eng. 36, 1131–1139. Marinova, D., Ribarova, F. and Atanassova, M. 2005. Total phenolics and total flavonoids in Bulgarian fruits and vegetables. J. Univ. Chem. Technol. Metall. 40, 255–260. Available via https://journal.uctm.edu/node/j2005-3/Marinova.pdf Saidi, R., Mimoune, N., Baazizi, R., Benaissa, M.H., Khelef, D. and Kaidi, R. 2019. Antibiotic susceptibility of staphylococci isolated from bovine mastitis in Algeria. J. Adv. Vet. Anim. Res. 6, 231. Salah, N., Miller, N.J., Paganga, G., Tijburg, L., Bolwell, G.P. and Rice-Evans, C. 1995. Polyphenolic flavanols act as scavengers of aqueous-phase radicals and chain-breaking antioxidants. Arch. Biochem. Biophys. 322, 339–346. Shamsa, F., Monsef, H., Ghamooshi, R. and Verdian-Rizi, M. 2008. Spectrophotometric determination of total alkaloids in some Iranian medicinal plants. Thai J. Pharm. Sci. 32, 17–20. Sharma, D.K. and Sharma, P. 2022. Alternative herbal medicines against bovine mastitis: a comprehensive review. Him. J. Agric. Res. 48, 145–150. Singh, K. and Tafida, G. 2014. Antibacterial activity of Moringa oleifera (Lam) leaves extracts against some selected bacteria. Int. J. Pharm. Pharm. Sci. 6, 52–54. Tran, P.A. and Webster, T.J. 2011. Selenium nanoparticles inhibit Staphylococcus aureus growth. Int. J. Nanomedicine 6, 1553–1558. Wang, H., Bi, C., Wang, Y., Sun, J., Meng, X. and Li, J. 2018. Selenium ameliorates Staphylococcus aureus-induced inflammation in bovine mammary epithelial cells by inhibiting the activation of the TLR2, NF-κB, and MAPK signaling pathways. BMC Vet. Res. 14, 1–8. Wayne, P. 2011. Clinical and Laboratory Standards Institute: performance standards for antimicrobial susceptibility testing, 21st information supplement, M100-S21, 2011. Wedley, A.L., Dawson, S., Maddox, T.W., Coyne, K.P., Pinchbeck, G.L., Clegg, P., Jamrozy, D., Fielder, M.D., Donovan, D. and Nuttall, T. 2014. Carriage of Staphylococcus species in a veterinary visiting dog population in mainland UK: molecular characterization of resistance and virulence. Vet. Microbiol. 170, 81–88. | ||
| How to Cite this Article |
| Pubmed Style Zeid MAA, Okasha L, Hegazy Y, Abdelmegeid M. Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and selenium nanoparticles. Open Vet. J.. 2025; 15(2): 835-846. doi:10.5455/OVJ.2025.v15.i2.33 Web Style Zeid MAA, Okasha L, Hegazy Y, Abdelmegeid M. Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and selenium nanoparticles. https://www.openveterinaryjournal.com/?mno=227988 [Access: September 02, 2025]. doi:10.5455/OVJ.2025.v15.i2.33 AMA (American Medical Association) Style Zeid MAA, Okasha L, Hegazy Y, Abdelmegeid M. Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and selenium nanoparticles. Open Vet. J.. 2025; 15(2): 835-846. doi:10.5455/OVJ.2025.v15.i2.33 Vancouver/ICMJE Style Zeid MAA, Okasha L, Hegazy Y, Abdelmegeid M. Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and selenium nanoparticles. Open Vet. J.. (2025), [cited September 02, 2025]; 15(2): 835-846. doi:10.5455/OVJ.2025.v15.i2.33 Harvard Style Zeid, M. A. A., Okasha, . L., Hegazy, . Y. & Abdelmegeid, . M. (2025) Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and selenium nanoparticles. Open Vet. J., 15 (2), 835-846. doi:10.5455/OVJ.2025.v15.i2.33 Turabian Style Zeid, Mayada A.m. Abou, Lamiaa Okasha, Yamen Hegazy, and Mohamed Abdelmegeid. 2025. Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and selenium nanoparticles. Open Veterinary Journal, 15 (2), 835-846. doi:10.5455/OVJ.2025.v15.i2.33 Chicago Style Zeid, Mayada A.m. Abou, Lamiaa Okasha, Yamen Hegazy, and Mohamed Abdelmegeid. "Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and selenium nanoparticles." Open Veterinary Journal 15 (2025), 835-846. doi:10.5455/OVJ.2025.v15.i2.33 MLA (The Modern Language Association) Style Zeid, Mayada A.m. Abou, Lamiaa Okasha, Yamen Hegazy, and Mohamed Abdelmegeid. "Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and selenium nanoparticles." Open Veterinary Journal 15.2 (2025), 835-846. Print. doi:10.5455/OVJ.2025.v15.i2.33 APA (American Psychological Association) Style Zeid, M. A. A., Okasha, . L., Hegazy, . Y. & Abdelmegeid, . M. (2025) Staphylococcus aureus in subclinical bovine mastitis: Prevalence and innovative treatment with Moringa oleifera and selenium nanoparticles. Open Veterinary Journal, 15 (2), 835-846. doi:10.5455/OVJ.2025.v15.i2.33 |