| Case Report | ||
Open Vet. J.. 2025; 15(2): 1056-1063 Open Veterinary Journal, (2025), Vol. 15(2): 1056-1063 Case Report Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspectsIago Martins Oliveira1*, Wanessa Patrícia Rodrigues da Silva1, Mariana Moreira Lopes2 and Naida Cristina Borges31Programa de Pós-graduação em Ciência Animal, Escola de Veterinária e Zootecnia, Universidade Federal de Goiás, Goiânia, Brazil 2Escola de Ciências Médicas e da Vida, Pontifícia Universidade Católica de Goiás, Goiânia, Brazil 3Departamento de Medicina Veterinária, Escola de Veterinária e Zootecnia, Universidade Federal de Goiás, Goiânia, Brazil *Corresponding Author: Iago Martins Oliveira. Programa de Pós-graduação em Ciência Animal, Escola de Veterinária e Zootecnia, Universidade Federal de Goiás, Goiânia, Brasil. Email: iago.vetufg [at] gmail.com Submitted: 12/11/2024 Accepted: 09/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
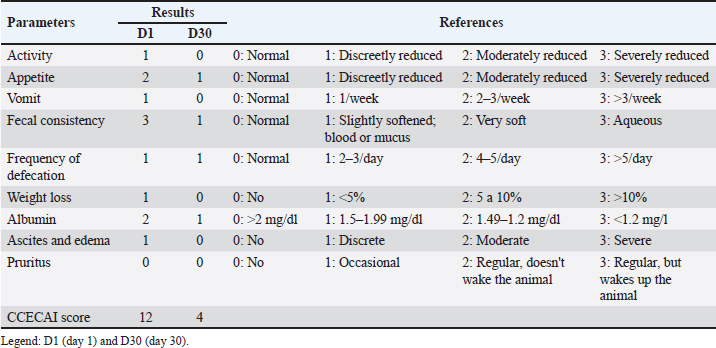
AbstractBackground: Primary intestinal lymphangiectasia in dogs is a condition characterized by dilation of the lymphatic vessels in the intestine. It has serious consequences, such as protein loss. Early diagnosis and appropriate treatment are essential for the quality of life of affected animals. Case Description: A 10-month-old female Border collie with persistent diarrhea for 3 months associated with weight loss, emesis, steatorrhea, and anorexia. During the investigation, the patient showed signs of dehydration; based on inspection and bone and muscle palpation, there was a reduction in the body condition score, as well as limb edema. An abdominal ultrasound revealed intestinal wall thickening and signs indicative of lymphangiectasia. The strain elastography revealed changes in intestinal rigidity parameters. After upper digestive endoscopy and histopathological examination, the female patient was diagnosed with intestinal lymphangiectasia. The female patient was treated with a low-fat diet and immunosuppressive drugs, which resulted in clinical and laboratory improvement and a reduction in the hardness rate on elastography. Conclusion: This case highlights the clinical, ultrasonographic, endoscopic, and strain elastographic findings of intestinal lymphangiectasia in a dog, demonstrating elastography’s potential as a diagnostic and monitoring tool while confirming the disease’s responsiveness to immunosuppressive therapy and dietary management. Keywords: Canine, Elasticity, Enteropathy, Intestinal mucosa. IntroductionCanine intestinal lymphangiectasia is a disease of the gastrointestinal tract that is characterized by the pathological dilation and rupture of lymphatic vessels, resulting in the leakage of lymph into the submucosa, lamina propria, and intestinal lumen (Louvet and Denis, 2004). The loss of lymphatic function leads to hypoalbuminemia, hypoglobulinemia, hypocholesterolemia, lymphopenia, and electrolyte disorders. Although commonly associated with nonspecific clinical signs, it can manifest in more serious ways, such as ascites, pleural effusion, anasarca, epileptic seizures, and thromboembolism (Jablonski, 2022). The clinicopathological and biochemical abnormalities observed in the disease are common to other conditions, such as gastroenteritis, liver failure, protein-losing nephropathy, and nephrotic syndrome, requiring the use of complementary tests to obtain a diagnosis (Lee et al., 2021). According to Okanishi et al. (2014), a definitive diagnosis is made through biopsy, which is performed surgically or through endoscopy and allows for the histopathological assessment of intestinal tissue samples. Endoscopic evaluation shows lesions that give the intestinal mucosa a whitish color and may present as multifocal granular foci and the presence of intraluminal lymphatic fluid. Histological analysis shows inflammatory infiltration with ectasia of intestinal lymphatic vessels (Larson et al., 2012). However, endoscopy is a supportive diagnostic method that should be used in association with other techniques because its specificity and sensitivity for the diagnosis of intestinal lymphangiectasia are only 48% and 68%, respectively (Lee et al., 2021). In addition, noninvasive diagnostic techniques, such as ultrasound and elastography, are frequently used. Ultrasound can identify alterations such as segmental or focal thickening of the jejunum and duodenum, loss of stratification, hyperechogenicity of intestinal tissue, abdominal effusion, lymphadenomegaly, and hypermotility of the small intestine (Pollard et al., 2013). Elastography is a modern diagnostic technique that can involve static assessments, which aim to identify the displacement of the sonographic wave due to a slight pressure exerted on the tissue, and dynamic assessments, which determine tissue elasticity based on the propagation speed of transverse waves. The use of this technique has been described in the assessment of mammary, testicular, prostatic, hepatic, and splenic lesions, but currently, studies aim to apply the technique to the identification of alterations in organs of the gastrointestinal tract in dogs (Spużak et al., 2019). To our knowledge, there is no description in the literature about intestinal elastography in dogs with intestinal lymphangiectasia. Therefore, this case report is a pioneering work. Our hypothesis was that semiquantitative data from strain elastography would increase, indicating that the greater hardness of the intestinal mucosa and that this type of examination could be a means of monitoring and prognosis of the disease. The aim of this study is to report a case of canine intestinal lymphangiectasia, with emphasis on the clinical and diagnostic approach using ultrasound, endoscopy, and, above all, elastography. Case DetailsA 10-month-old female Border collie with a main complaint of profuse liquid diarrhea. During anamnesis, it was reported that fecal changes had been occurring for 3 months. The patient also described emesis, steatorrhea, inappetence, and weight loss. The physical examination revealed a reduced body condition score (4/9) (Chun et al., 2019), tenderness to deep abdominal palpation, slight edema of the limbs, increased intestinal borborygmus on auscultation, hypocolored mucous membranes, and a capillary refill time of more than 2 seconds. The other clinical parameters were within the reference values of the species. Based on the obtained information, the suspected diagnosis was infectious, inflammatory, parasitic, or alimentary enteropathy, or secondary to a systemic condition. Given the chronicity of the clinical picture, chronic enteropathy was graded using the canine chronic enteropathy activity index (CCECAI) (Allenspach et al., 2007). The index used behavior, appetite, vomiting frequency, stool consistency, defecation frequency, weight loss, serum albumin, presence of ascites, peripheral edema, and pruritus as parameters. These criteria were evaluated on a scale of 0–3, and the final score was obtained by adding up the data. The total score can vary from 0 to 27. Based on these values, chronic intestinal disease is classified as clinically insignificant (0–3 points), mild (4–5 points), moderate (6–8 points), severe (9–11 points), or very severe (≥12 points). The patient in this report underwent CCECAI 12, and the detailed results are presented in Table 1. Blood count, creatinine, alanine aminotransferase (ALT), alkaline phosphatase (ALP), albumin, total cholesterol, cobalamin, folate, C-reactive protein (CRP), basal bile acids, urinalysis, and urinary protein:creatinine ratio were performed. For the differential diagnosis of atypical canine hypoadrenocorticism and exocrine pancreatic insufficiency, basal cortisol was measured and trypsin-like immunoreactivity (TLI), respectively. The blood count showed lymphopenia and thrombocytosis. In addition, hypoalbuminemia, hypocholesterolemia, hypocobalaminemia, serum folate reduction, and increased CRP levels were observed. The other laboratory tests were within the reference values of the species. The results of the laboratory parameters are presented in Table 2. For fecal evaluation, fecal parasitology was performed using three serially collected samples, using direct and Faust techniques, as well as an immunochromatographic test to detect anti-Giardia duodenalis fecal antigens. However, no evidence of intestinal parasitism was found. Table 1. Canine chronic enteropathy activity index of a canine patient with intestinal lymphangiectasia at initial diagnosis and after 30 days of treatment.
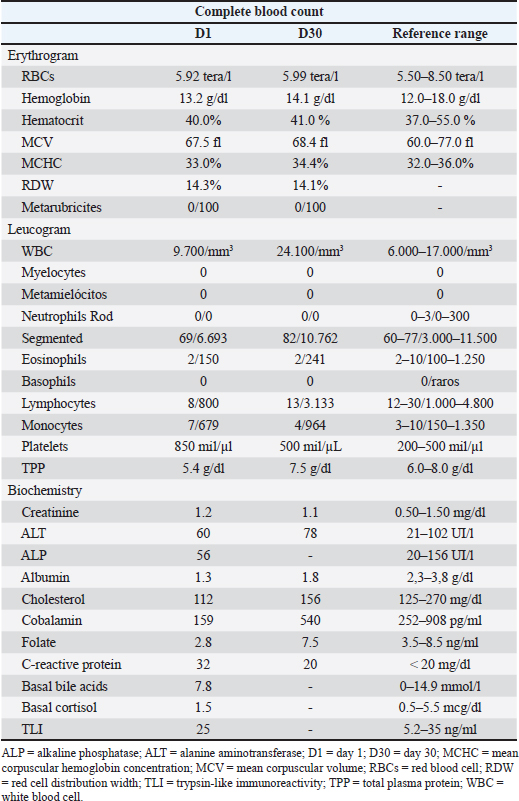
Table 2. Laboratory parameters of a dog with intestinal lymphangiectasia at the start of diagnosis and 30 days after treatment.
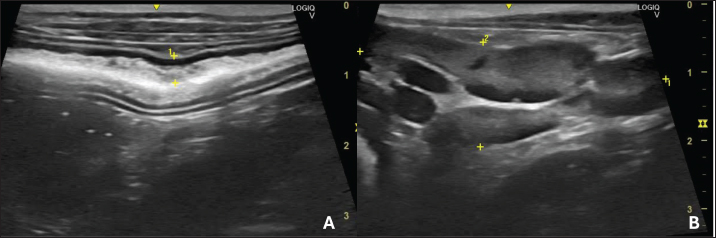
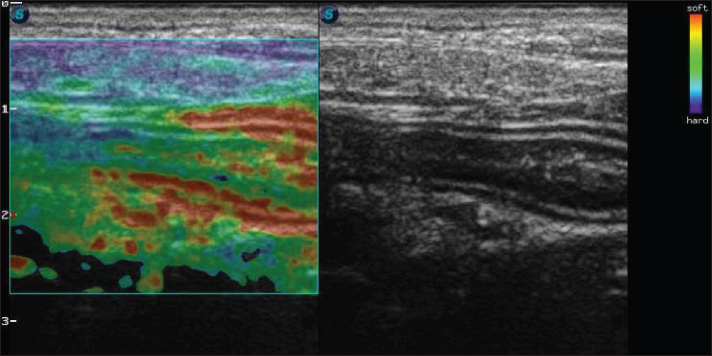
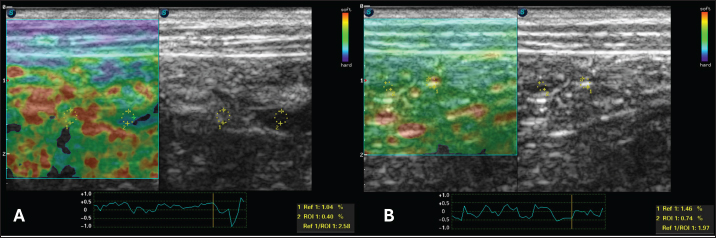
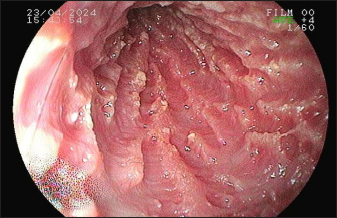
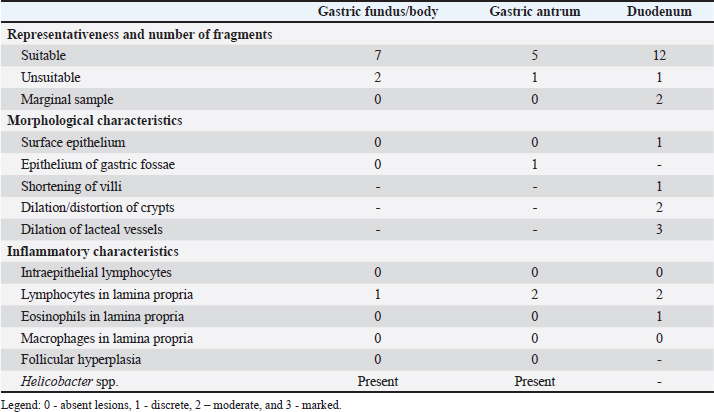
In view of the test results, fenbendazole (Fenzol Pet®, Agener União, São Paulo, SP, Brazil) 50 mg/kg every 24 hours for 3 days was prescribed, and the protocol was repeated after 15 days, as well as a commercial symbiotic. A restrictive hypoallergenic diet containing hydrolyzed protein is recommended for 30 consecutive days. There was no improvement in the clinical signs, and diarrhea persisted. An abdominal ultrasound was performed using SAEVO® FT422 equipment with an 8.0-MHzz multi-frequency microconvex transducer. Gastric thickening (0.71 cm) was observed in the accessible portions, with preserved stratification and a mucosal layer containing intramural gas, suggesting gastritis and gastric pneumatosis. The duodenum and jejunum were also thickened (0.56 and 0.40 cm, respectively), maintaining their layered pattern. The intestinal mucosa exhibited thickening and hyperechogenicity, with coalesced hyperechoic echoes on the luminal surface (Fig. 1A), indicating inflammation associated with lymphangiectasia. Additionally, an enlarged jejunal lymph node was noted, characterized by regular margins, homogeneous echotexture, preserved echogenicity, and dimensions of 4.63 × 1.53 cm (Fig. 1B). Strain elastography was performed using the same ultrasound machine with a linear transducer and a frequency of 10 MHz. The transducer was positioned on the animal’s abdomen, and then, five cycles of compression and decompression were performed. The color histogram was generated and superimposed on the duplicate B-mode image. This map generated images with a colorimetric scale ranging from blue to red for tissues with maximum stiffness, intermediate elasticity, and maximum elasticity. For the static qualitative assessment, the proximal segments of the duodenum and jejunum were analyzed in a longitudinal ultrasound section, and a heterogeneous distribution was found between the dorsal and ventral regions of the intestine, with patterns ranging from predominantly blue with some greenish parts and reddish foci (Fig. 2). To obtain the semiquantitative parameters, the region of interest (ROI) was drawn using the device’s software cursor. Initially, a reference, ROI 1 was obtained in the mesentery adjacent to the intestines, and ROI 2 was drawn in the dorsal region of the duodenum and jejunum. The drawings were similar and circular with the same depth and horizontal direction in the cross-section. The drawings were based on the diameter of the animal’s intestinal mucosa, and the force applied was guided by the elastographic compression wave. The strain ratio (SR) was obtained by calculating the ratio between the ROIs. The values for the duodenum were as follows: ROI 1=1.04 % and ROI=0.4%, with an SR of 2.58 (Fig. 3A). In the jejunum, ROI 1 measured 1.46 %, ROI 0.74 %, and SR 1.97 (Fig. 3B). Given the lack of response to treatment and the test results, the patient was referred for an upper digestive endoscopy. The examination revealed a smooth esophagus with hyperemic cardia mucosa. The stomach had anatomical folds in the body region, and the mucosa had erythematous spots. The pylorus presented a hyperplastic appearance, while the duodenum exhibited edematous mucosa and an irregular surface caused by increased whitish content, which was suggestive of lacteal dilation and duodenitis (Fig. 4). Materials were collected from 15 fragments of the gastric and duodenal mucosa for subsequent histopathological analysis. Histopathological analysis revealed different findings in the stomach and duodenum. In the stomach, slight lymphoplasmacytic infiltration was observed in the lamina propria. In the duodenum, there was no evidence of intraepithelial lymphocytes, but the lamina propria showed discrete eosinophilic infiltrate, moderate lymphoplasmacytic infiltrate along with dilation and distortion of the crypts, as well as dilation of the lacteal vessels. No specific etiological agents or lesions suggestive of neoplasia were identified. The histopathological score adapted from Allenspach et al. (2019) is shown in Table 3.
Fig. 1. B-mode ultrasound of a dog with intestinal lymphangiectasia. A: Duodenum with hyperechogenic mucosa at the dorsal and ventral margins. B-Jejunal lymph node with regular margins, homogeneous echotexture, and preserved echogenicity.
Fig. 2. Color elastography in double projection with a B-mode ultrasound image of a longitudinal scan of the proximal jejunum. The submucosa, muscularis, ventral, and dorsal serosa are red-green; the ventral and dorsal mucosa are blue-green.
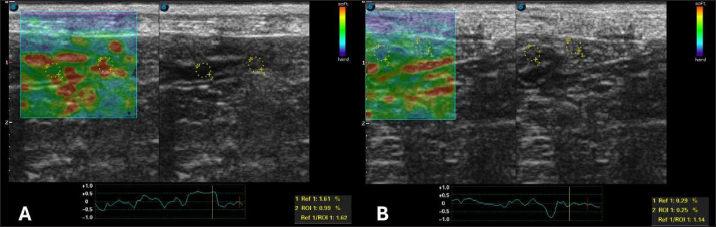
Fig. 3. Semiquantitative cross-sectional deformation elastography of the intestine of a dog with intestinal lymphangiectasia. The SR was obtained by evaluating ROI 1 of the mesenteric and ROI 2 of the dorsal region of the intestinal segment. A duodenum with an SR of 2.58 B-Jejunum with an SR of 1.97 Based on the obtained information, the definitive diagnosis was primary intestinal lymphangiectasia. As a result, a low-fat diet was prescribed (Gastrointestinal low fat, Royal Canin®, Descalvado, São Paulo, SP, Brazil), prednisolone (Predsim®, Mantecorp Farmasa, São Paulo, SP, Brazil) 1 mg/kg orally every 24 hours, chlorambucil (Leukeran®, Aspen Pharma, Rio de Janeiro, RJ, Brazil) 4 mg/m2 orally every 48 hours, and clopidogrel (Manipulado®, Drogavet, Goiânia, GO, Brazil) 1 mg/kg every 24 hours. After 30 days of treatment, there were no more laboratory changes (Table 2), the CCECAI was reduced to four (Table 1), and the patient no longer had diarrhea and gained weight. In addition, hyperechogenicity of the intestinal mucosa was reduced, and the values for elastography of the duodenum were as follows: ROI 1 1.61 % and ROI 0.99 %, with an SR of 1.62 (Fig. 5A). In the jejunum, ROI 1 measured 0.29 %, ROI 0.25%, and SR 1.14 (Fig. 5B). The drugs were withdrawn, and only the corticosteroid, which was reduced to 0.5 mg/kg every 24 hours, was administered. Follow-up was recommended every 2 months and, in cases of recurrence, clinical treatment. DiscussionAfter clinical, laboratory, and dietary screening, the patient was diagnosed with intestinal lymphangiectasia. This result corroborates a recent classification of canine chronic enteropathy, which included intestinal inflammation associated with protein loss (Dupouy-Manescau et al., 2024). The authors believe that this type of enteropathy can be further fragmented into diet-responsive, immunosuppressant-responsive, and non-responsive. We believe that the animals described in this report have immunosuppressant-responsive disease even though the treatment protocol included a low-fat diet. However, we recognize that this case report describes a short period of therapeutic intervention that does not allow differentiation of the type of protein-losing enteropathy (PLE).
Fig. 4. Duodenoscopy of a dog with intestinal lymphangiectasia. Lacteal dilatation with a raised, whitish appearance in the duodenal mucosa. The CCECAI score was used in dogs with PLE in a correlation study with the expression of a marker associated with lymphatic dilation, with an average score of eight points (Nagahara et al., 2021). Despite the similarity between the definitive diagnosis of this clinical case and the study, there was a discrepancy between the averages of the animals and the value described here, which was initially 12. This indicates the difference in the severity of the clinical signs of this enteropathy. However, this clinical index was important for monitoring therapy because it was reduced after 30 days of treatment. This finding corroborates the report by Resende et al. (2023), who observed a reduction in the clinical activity score in a dog with lymphangiectasia after the use of immunosuppressants, supportive therapy, and diet. An abdominal ultrasound can show intestinal wall thickening and the presence of hyperechogenic striations in the mucosa (Sutherland-Smith et al., 2007) Figure 1A. However, in our patient, the echogenic echoes coalesced in the mucosa, which we believe was associated with greater lymphatic dilation. These results are therefore in line with Malacuns (2021), who found a greater increase in mucosal echogenicity in dogs with lacteal dilatation confirmed by digestive endoscopy. Despite the suggestion of greater severity, our patient’s findings differ from the description (De Magistris et al., 2023), which states that in cases of severe PLE, there may be the formation of lipogranuloma. Table 3. Severity score of histopathologic findings of endoscopic biopsy samples from the stomach and duodenum of a dog with intestinal lymphangiectasia.
Fig. 5. Semiquantitative elastography of deformation in a cross-section of the intestines of a dog with intestinal lymphangiectasia after 30 days of treatment. The deformation ratio (SR) was obtained by evaluating ROI 1 of the mesentery and ROI 2 of the dorsal portion of the intestinal segment. A duodenum with an SR of 1.62 B-Jejunum with SR=1.14. Endoscopic evaluation by upper gastrointestinal endoscopy can reveal isolated, punctuated, or coalesced white spots related to dilation of the lymphatic vessels with or without the presence of rupture and extravasation of lymph (Jablonski, 2022). These findings were observed via duodenoscopy in this case, but it was not possible to determine whether lymphatic rupture was present. In addition, there was a correlation between the macroscopic findings and histopathology, which showed distortion of the crypts and lacteal dilatation. This correlation was also found in the literature (García-Sancho et al., 2011). Regarding elastographic aspects, no directly comparable veterinary studies have been conducted because there is no description of the strain elastography technique in dogs with this disease. A single analogous study provided reference data on the healthy jejunal mucosa of 60 dogs using shear wave elastography (Spużak et al., 2019). In this study, the mean values of the healthy animals were lower than those of the dogs with PLE. We therefore conclude that the stiffness of the intestinal mucosa is likely to increase, regardless of the elastography technique used. We suggest that this report serves as a basis for further research. In human medicine, the results are similar to those observed in this report. Intestinal inflammation leads to an increase in the hardness rates seen on strain elastography, and after treatment, these parameters are reduced (Gabbiadini et al., 2021). Therefore, they are used for follow-up and as predictors of fibrosis. Despite the similarity, we did not reveal fibrosis in the duodenal mucosa. The most frequently identified biochemical abnormalities are hypoalbuminemia, hypogammaglobulinemia, lymphopenia, anemia, and ionized hypocalcemia (Huber et al., 2020). These results were partially verified in the tests performed on the dogs in this study. It should be noted that the differentials of hypoalbuminemia were ruled out by excluding liver failure, malnutrition, and protein-losing glomerulopathy. Treatment was based on the use of immunosuppressive drugs and restriction of fat intake. This finding corroborates the literature stating that the disease can be responsive to diet and immunosuppressants (Dupouy-Manescau et al., 2024). More severe and refractory cases may require other therapies, such as the use of octreotide, a somatostatin analog. A recent study found that approximately 50% of PLEs do not respond to treatment, that is why the authors used this drug (Jablonski et al., 2024). Despite the possibility, in this study, the animal responded to conventional treatment, and this response was proven by clinical, laboratory, and elastographic improvement. ConclusionThis case report pioneers the use of intestinal elastography in a dog with lymphangiectasia, demonstrating increased mucosal stiffness that may not necessarily indicate fibrosis. The integration of clinical, ultrasonographic, endoscopic, and elastographic findings underscores the potential of semiquantitative strain elastography as a valuable tool for disease monitoring and prognostication. AcknowledgmentsTo the Veterinary Hospital of the Veterinary and Zootechnical School of the Federal University of Goiás. Conflicts of interestThe authors declare no conflicts of interest. FundingThis research did not receive any specific grant. Data availabilityAll data supporting the findings of this study are available in the manuscript. Authors’ contributionConceptualization I. M. O., N. C. B., W. P. R. S., and M. M. L., methodology I. M. O. and W. P. R. S., formal analysis N. C. B., M. M. L., data curation N. C. B., I. M. O., conducted the clinical case; W. P. R. S., performed the animal’s elastography; I. M. O., W. P. R. S., and M. M. L., drafted the manuscript; N. C. B., revised the manuscript and provided guidance on the clinical case; I. M. O., and N. C. B., translated the manuscript. All authors have read and agreed with the published version of the manuscript. ReferencesAllenspach, K.A., Mochel, J.P., Du, Y., Priestnall, S.L., Moore, F., Slayter, M., Rodrigues, A., Ackermann, M., Krockenberger, M., Mansell, J., Luckschander, N., Wang, C., Suchodolski, J., Berghoff, N. and Jergens, A.E. 2019. Correlating gastrointestinal histopathologic changes to clinical disease activity in dogs with idiopathic inflammatory bowel disease. Vet. Pathol. 56, 435–443. Allenspach, K., Wieland, B., Gröne, A. and Gaschen, F. 2007. Chronic enteropathies in dogs: evaluation of risk factors for negative outcome. J. Vet. Intern. Med. 21, 700–708. Chun, J.L., Bang, H.T., Ji, S.Y., Jeong, J.Y., Kim, M., Kim, B., Lee, S.D., Lee, Y.K., Reddy, K.E. and Kim, K.H. 2019. A simple method to evaluate body condition score to maintain the optimal body weight in dogs. J. Anim. Sci. Technol. 61, 366–370. De Magistris, A.V., Linta, N., Specchi, S., Procoli, F., Sabattini, S., Diana, A. and Donato, P.D. 2023. Ultrasonographic features of intestinal lipogranulomatous lymphangitis in 10 dogs. Vet. Radiol. Ultrasound 64, 973–981. Dupouy-Manescau, N., Méric, T., Sénécat, O., Drut, A., Valentin, S., Leal, R.O. and Hernandez, J. 2024. Updating the classification of chronic inflammatory enteropathies in dogs. Animals 14, 681–700. Gabbiadini, R., Zacharopoulou, E., Furfaro, F., Craviotto, V., Zilli, A., Gilardi, D. and Allocca, M. 2021. Application of ultrasound elastography for assessing intestinal fibrosis in inflammatory bowel disease: fiction or reality? Curr. Drug Targets 22, 347–355. García-Sancho, M., Sainz, A., Villaescusa, A., Rodríguez, A. and Rodríguez-Franco, F. 2011. Manchas brancas na superfície mucosa do duodeno em cães com enterite plasmocítica linfocítica. J. Vet. Sci. 12, 165–169. Huber, R., Semmler, G., Mayr, A., Offner, F. and Datz, C. 2020. Linfangiectasia intestinal primária em um paciente adulto: um relato de caso e revisão da literatura. World J. Gastroenterol. 26, 7707–7718. Jablonski, S.A., Mazepa, A.S. and Tolbert, M.K. 2024. Use of octreotide for the treatment of protein-losing enteropathy in dogs: retrospective study of 18 cases. J. Vet. Intern. Med. 38, 145–151. Jablonski, S.A. 2022. Pathophysiology, diagnosis, and management of canine intestinal lymphangiectasia: a comparative review. Animals 12, 2791. Larson, R.N., Ginn, J.A., Bell, C.M., Davis, M.J., and Foy, D.S. 2012. Duodenal endoscopic findings and histopathologic confirmation of intestinal lymphangiectasia in dogs. J. Vet. Intern. Med. 26, 1087–1092. Lee, EJ., Chung, MJ. and Jeong, KS. 2021. Canine intestinal lymphangiectasia concomitant with renal cell carcinoma. Acta Vet. 71, 351–360. Louvet, A. and Denis, B. 2004. Ultrasonographic diagnosis - small bowel lymphangiectasia in a dog. Vet. Radiol. Ultrasound 45, 6–10. Malancus, R.N. 2021. Ultrasound and endoscopic findings in dogs with lymphangiectasia. Arq. Bras. Med. Vet. Zootec. 73, 49–54. Nagahara, T., Ohno, K., Nagao, I., Nakagawa, T., Goto-Koshino, Y., Tsuboi, M. and Tsujimoto, H. 2022. Association between intestinal lymphangiectasia and expression of inducible nitric oxide synthase in dogs with lymphoplasmacytic enteritis. J. Vet. Med. Sci. 84, 20–24. Okanishi, R., Yoshioka, R., Kagawa, Y. and Watari, T. 2014. The clinical efficacy of dietary fat restriction in treatment of dogs with intestinal lymphangiectasia. J. Vet. Intern. Med. 28, 809–817. Pollard, R.E., Johnson, E.G., Pesavento, P.A., Baker, T.W., Cannon, A.B., Kass, P.H. and Marks, S.L. 2013. Effects of corn oil administered orally on conspicuity of ultrasonographic small intestinal lesions in dogs with lymphangiectasia. Vet. Radiol. Ultrasound 54, 390–397. Resende, S.I.L., Bastos, K.O., Ribeiro, D., Ferreira, W.A., Ribeiro, C.B., Pereira, L.D.S. and Abreu, C.B. 2023. Inflammatory bowel disease associated with lymphangiectasia in a dachshund dog. Acta Sci. Vet. 51, 1–6. Spużak, J., Kubiak, K., Glińska-Suchocka, K., Jankowski, M., Borusewicz, P. and Kubiak-Nowak, D. 2019. Accuracy of real-time shear wave elastography in the assessment of normal small intestine mucosa in dogs. Pol. J. Vet. Sci. 22 3, 457–461. Sutherland-Smith, J., Penninck, D.G., Keating, J.H. and Webster, C.R. 2007. Ultrasonographic intestinal hyperechoic mucosal striations in dogs are associated with lacteal dilation. Vet. Radiol. Ultrasound 48, 51–57. | ||
| How to Cite this Article |
| Pubmed Style Oliveira IM, Silva WPRD, Lopes MM, Borges NC. Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspects. Open Vet. J.. 2025; 15(2): 1056-1063. doi:10.5455/OVJ.2025.v15.i2.54 Web Style Oliveira IM, Silva WPRD, Lopes MM, Borges NC. Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspects. https://www.openveterinaryjournal.com/?mno=228286 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.54 AMA (American Medical Association) Style Oliveira IM, Silva WPRD, Lopes MM, Borges NC. Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspects. Open Vet. J.. 2025; 15(2): 1056-1063. doi:10.5455/OVJ.2025.v15.i2.54 Vancouver/ICMJE Style Oliveira IM, Silva WPRD, Lopes MM, Borges NC. Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspects. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 1056-1063. doi:10.5455/OVJ.2025.v15.i2.54 Harvard Style Oliveira, I. M., Silva, . W. P. R. D., Lopes, . M. M. & Borges, . N. C. (2025) Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspects. Open Vet. J., 15 (2), 1056-1063. doi:10.5455/OVJ.2025.v15.i2.54 Turabian Style Oliveira, Iago Martins, Wanessa Patrícia Rodrigues Da Silva, Mariana Moreira Lopes, and Naida Cristina Borges. 2025. Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspects. Open Veterinary Journal, 15 (2), 1056-1063. doi:10.5455/OVJ.2025.v15.i2.54 Chicago Style Oliveira, Iago Martins, Wanessa Patrícia Rodrigues Da Silva, Mariana Moreira Lopes, and Naida Cristina Borges. "Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspects." Open Veterinary Journal 15 (2025), 1056-1063. doi:10.5455/OVJ.2025.v15.i2.54 MLA (The Modern Language Association) Style Oliveira, Iago Martins, Wanessa Patrícia Rodrigues Da Silva, Mariana Moreira Lopes, and Naida Cristina Borges. "Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspects." Open Veterinary Journal 15.2 (2025), 1056-1063. Print. doi:10.5455/OVJ.2025.v15.i2.54 APA (American Psychological Association) Style Oliveira, I. M., Silva, . W. P. R. D., Lopes, . M. M. & Borges, . N. C. (2025) Intestinal lymphangiectasia in a dog: Clinical, ultrasonographic, endoscopic, and strain elastographic aspects. Open Veterinary Journal, 15 (2), 1056-1063. doi:10.5455/OVJ.2025.v15.i2.54 |