| Research Article | ||
Open Vet. J.. 2025; 15(2): 893-900 Open Veterinary Journal, (2025), Vol. 15(2): 893-900 Research Article Changes in blood serum protein profile in low-yielding ewes during the periparturient periodVeronika Glembová, Csilla Tóthová*, Veronika Oršuľaková and Oskar NagyClinic of Ruminants, University of Veterinary Medicine and Pharmacy in Košice, Košice, Slovak Republic *Corresponding Author: Csilla Tóthová. Clinic of Ruminants, University of Veterinary Medicine and Pharmacy in Košice, Košice, Slovak Republic. Email: csilla.tothova [at] uvlf.sk Submitted: 03/12/2024 Accepted: 09/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
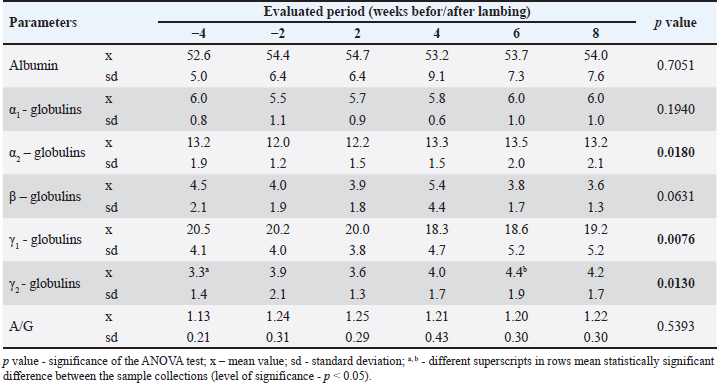
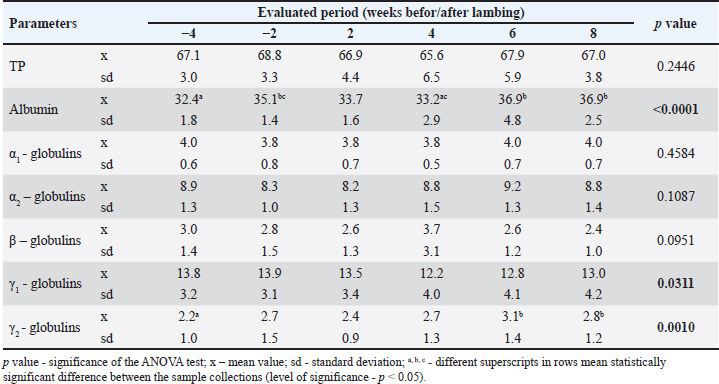
AbstractBackground: In high-yielding dairy ewes, metabolic and reproductive processes occurring around the time of parturition and during lactation initiation are accompanied by several changes in blood biochemical parameters, including those related to protein metabolism. However, little is known about the distribution of serum proteins throughout the peripartum period in ewes without higher economic productivity. Aim: This study aimed to evaluate changes in serum protein profile throughout the peripartal period in ewes with milk production only for the nutritional needs of lambs. Methods: Twelve clinically healthy ewes of Merino and Cigája crossbreeds at the late stage of pregnancy were included in the study. They were evaluated from 1 month before the expected lambing to 8 weeks after lambing. Blood samples were collected 4 and 2 weeks before the expected parturition and then 2, 4, 6, and 8 weeks postpartum. The serum samples were analyzed for the concentrations of total serum proteins and the main protein fractions separated by agarose gel electrophoresis. Results: Six protein fractions were identified on the serum protein electrophoretogram of the ewes, including albumin, α1-, α2-, β-, γ1, and γ2-globulins. The concentrations of total serum proteins did not significantly vary throughout the evaluated peripartal period but decreased only slightly around the time of parturition. Significant changes were observed in the relative values and distribution of α1-and γ-globulin fractions (p < 0.05 and p < 0.01). The absolute albumin concentrations were lowest 4 weeks before lambing and highest in the later postpartal period (p < 0.001). The γ1-globulin values showed a significant gradual decrease until 4 weeks after lambing (p < 0.05 and p < 0.01). Significant changes were observed in the concentrations of γ2-globulins (p < 0.01), with the highest values in the later postpartal period similar to γ1-globulins. Conclusion: The results suggest that changes in the concentration and distribution of serum protein fractions around the time of parturition also occur in ewes with low milk production and may help to better understand the physiological adaptations, that accompany the female’s organism during the transitional period. Keywords: Ewes, Peripartal period, Serum proteins, Electrophoresis, Protein fractions, Albumin, Globulins. IntroductionIn small ruminants, pregnancy and the initiation of lactation are critical physiological states accompanied by several intensive metabolic and reproductive processes. These are associated with the growth and development of the fetus, preparation of the urogenital tract for parturition, and the mammary gland for lactation, while the homeostasis of the mother should be maintained (King, 2000; Iriadam, 2007). All these processes require a great amount of energy, resulting in changes in blood biochemical parameters, which are related especially to the energetic profile (Saeed et al., 2009). Alterations may also occur in protein metabolism, with the synthesis of body proteins in the developing fetus during pregnancy, and mainly in its last period, leading to depletion of maternal serum proteins (Zvorc et al., 2000). Furthermore, free amino acids and proteins are also used for the synthesis of milk proteins in the mammary gland secretory cells (Karapehlivan et al., 2007). One of the laboratory methods available to evaluate changes in the serum protein profile is serum protein electrophoresis, which allows the separation and identification of protein fractions, each composed of several individual proteins with similar electrophoretic mobility (O’Connell et al., 2005). Serum protein electrophoresis can be used not only to identify abnormal proteins but also to assess the nature of dysproteinemia (predominantly hyperproteinemia or hyperglobulinemia) or to differentiate the type of inflammation and stimulated humoral immune response (Keren et al., 1999; Vavricka et al., 2009). Even in cases with unchanged total serum protein concentrations, this laboratory technique may be useful for detecting possible abnormalities in the serum protein pattern and for the production of different protein fractions (Jania and Andraszek, 2016). Studies performed by Grünberg et al. (2011) and Tóthová et al. (2018) in periparturient cows showed a negative protein balance with lower concentrations of total serum proteins around calving compared with values outside the parturient period and in the following phases of lactation. This reflects the utilization of amino acids from the maternal circulation for protein synthesis in the fetal muscles, as well as the transfer of immunoglobulins and amino acids from the bloodstream to the mammary gland for the synthesis of colostrum and milk (Antunović et al., 2002, Roubies et al., 2006). Lower concentrations of total serum proteins were also found in periparturient goats and ewes than in later postpartum periods (Djuricic et al., 2011; Boudebza et al., 2016). In Comisana ewes, dynamic changes have also been observed in the serum protein electrophoretic pattern during pregnancy and lactation until the dry period (Piccione et al., 2012). The Comisana sheep is a very productive dairy breed with higher milk production and is suitable for twin pregnancy. However, there is limited information on the distribution of serum protein fractions during the peripartum period in ewes without higher economic productivity. Therefore, the aim of this study was to evaluate changes in serum protein profiles throughout the peripartal period in ewes not used for milk production but only for the production and rearing of lambs. At the same time, it was a goal to assess the changes in the distribution of serum protein fractions in the period including the late stage of pregnancy up to 2 months of lamb rearing. Materials and MethodsEthical statementHandling of the animals, sample collection, and preparation were conducted in accordance with the ethical standards and guidelines approved by the Committee of the University of Veterinary Medicine and Pharmacy in Košice on the protection of animals used for scientific purposes and complied with the institutional requirements of the Code of Ethics for Scientists (Directive 74/2019/UVLF). Animals and sample collectionFor the evaluation of the aim of the study, 12 clinically healthy ewes at a high stage of gestation were selected randomly from the university school farm. The selected ewes were transported from the farm to the Clinic of Ruminants of the University Veterinary Hospital where they were housed in straw-bedded box stalls, 2 to 3 animals were placed in one box. The ewes were crossbreeds of Merino and Cigája, aged between 2 and 4 years and weighing 49.3 ± 5.7 kg. Pregnancies were confirmed using ultrasound examination. They were included in the study 1 month before the expected parturition, and the evaluation lasted until 2 months after delivery. On the farm, the ewes were grazing in natural pasture until autumn and were housed in barns at night. In addition to grazing, the ewes were fed hay ad libitum and grain mixtures once a day with free access to water. The ewes on this farm were treated with antihelmintics against internal and external parasites, and their claws were treated regularly. At the clinic, the animals were fed native hay and concentrate mixtures and had free access to drinking water. The dams were continuously checked for their health status during pregnancy. Pregnancies were of normal length, and parturitions occurred under controlled conditions at the University Veterinary Hospital. The births were uncomplicated, and all newborns were clinically in good health condition. During the entire observation period, the ewes were clinically healthy, and after birth, the lambs were housed together with the mothers. The first blood samples from the ewes were collected 4 weeks before the expected parturition. Thereafter, blood was sampled 2 weeks before lambing and then 2, 4, 6, and 8 weeks postpartum. Blood samples were withdrawn from the jugular vein into 4.4-ml serum gel separator tubes containing no additives or anticoagulants (Sarstedt, Nümbrecht, Germany). After a 30-minute period of standing at room temperature, the tubes were centrifuged at 4,000 g for 15 minutes, and the separated sera were transferred into Eppendorf tubes. Aliquots of sera were frozen at 20°C and stored until further processing and laboratory analyses. Laboratory analysesThe serum samples were analyzed for the concentrations of total serum proteins (TP, g/l) and electrophoretic patterns of serum proteins. The biuret method was applied to measure the TP concentrations using commercially available diagnostic kits (Randox, Crumlin, United Kingdom) and an automated chemistry analyzer Alizé (Lisabio, Poully en Auxois, France). The separation and distribution of serum protein fractions were performed by zone electrophoresis on agarose gel using an automated electrophoresis system (Hydrasys) with commercial diagnostic kits Hydragel 7 Proteine, Sebia Corporate, Lisses, Evry Cedex, France) according to the application instructions of the manufacturer. After gel separation and staining, the electrophoretic curves were displayed. The staining intensity of individual protein bands was quantified using a densitometer (Epson Perfection V700, Epson America Inc., CA, USA) and the image analysis software Phoresis version 5.50 (Sebia Corporate, France). The protein fractions were expressed as relative values (%) according to the optical density, and their absolute concentrations (g/l) were quantified from the TP concentrations. Albumin:globulin ratios (A/G) were also calculated. Statistical analysesData were submitted to descriptive statistics using GraphPad Prism V5.02 (GraphPad Software Inc., CA, USA). All results are presented as means and standard deviations (SD). Kolmogorov-Smirnov test for normality was applied to evaluate data distribution. The overall variances among the results were estimated using repeated measures ANOVA. The Tukey multiple comparison test was applied to test the significance of differences between the sample collections. Differences were considered significant when the p value was less than 0.05. ResultsResults of the serum protein electrophoresis in ewes during the peripartal period are presented in Tables 1 and 2. Six protein fractions were identified on the serum protein electrophoretogram of the ewes, including albumin, α1-, α2-, β-, γ1, and γ2-globulins (Fig. 1a–f). Of the monitored serum protein fractions, significant changes in values (p < 0.05 and p < 0.01) were recorded for relative concentrations of α2-, γ1-, and γ2-globulins (Table 1). The relative concentrations of α2-globulins were lower around the time of parturition (2 weeks before and 2 weeks after) than during the other monitored periods. A similar but insignificant trend of changes in values was also noted for α1- and β-globulin fractions In the relative concentrations of γ1-globulins a gradual decrease in concentrations was recorded until week 4 after lambing. In the following period, an increase in the concentration of this globulin fraction was observed. The opposite trend was found in the relative concentrations of γ2-globulins. The lowest mean value of this fraction was recorded 4 weeks before lambing, and the highest mean values were observed in the later postpartal period. The analysis of the A/G ratios showed no significant changes during the monitored period. The lowest mean value was found 4 weeks before parturition and the highest 2 weeks after parturition. The total serum protein concentrations did not vary significantly in the ewes during the evaluation period (Table 2). The lowest means were recorded in the period 2 and 4 weeks after parturition. Significant changes in the absolute albumin concentrations were observed (p < 0.001). The lowest mean values were observed 4 weeks before and 2 and 4 weeks after lambing. The mean concentrations were highest in the later postpartum period. In the absolute concentrations of α1-, α2-, and β-globulins, no significant changes were observed in ewes during the evaluated peripartum period. The absolute mean concentrations of α2-globulins found in week 2 before and in week 2 after parturition were lower compared to the later postpartum period. Similar trends in mean values in the peripartum period were also recorded for α1- and β-globulins. However, significant changes compared with these protein fractions were observed in the absolute concentrations of γ1- and γ2-globulins (p < 0.05 and p < 0.01, respectively). The concentrations of these fractions changed during the peripartum period in the opposite direction. Although the mean concentrations of γ1-globulins were lower than before delivery, the changes found for γ2-globulins were characterized by higher mean values in the postpartum period, and their concentrations were the lowest 4 weeks before parturition. Table 1. Changes in the relative values of analyzed protein fractions (%) and A/G ratios in ewes during the peripartum period.
Table 2. Changes in the absolute concentration of total proteins (TPs) and the analyzed protein fractions in ewes during the peripartum period (g/l).
DiscussionLate pregnancy, parturition, and the initiation of lactation are critical physiological phases in small ruminants, which are accompanied by several adaptation mechanisms and metabolic disturbances (Bremmer et al., 2000; Antunović et al., 2011). All these processes are associated with maximum energy need, resulting in changes in biochemical parameters related to energy metabolism (Block et al., 2001). Changes have also been observed in the protein profile parameters, with decreased concentrations of total serum proteins in late gestation and early lactation compared with the dry period (Karapehlivan et al., 2007; Piccione et al., 2009; Boudebza et al., 2016). The decrease in serum protein concentrations during late pregnancy may be attributed to the synthesis of proteins by the fetus from the amino acids of the mother (Antunović et al., 2002). In dairy goats and cows, decreased total protein values were obtained at the end of gestation and at parturition, especially because of the migration of globulins to the mammary gland for colostrum synthesis, with the return to physiological values post partum (Piccione et al., 2011; Elzein et al., 2016; Soares et al., 2018). On the other hand, Baumgartner and Pernthaner (1994) did not find a significant effect of the reproduction stage on total serum protein concentrations in Karakul sheep. In ewes included in our study, the total protein concentrations obtained after lambing were slightly lower than those recorded during late gestation. This may be related to lower milk production in this type of sheep than in high-yielding dairy ewes and goats. In our sheep, the concentration of TP started to slightly increase from week 6 after lambing, in dairy ewes and goats more markedly increasing total protein values were observed with the progress of lactation (Krajnicakova et al., 1993; Piccione et al., 2009; Janků et al., 2011). In albumin concentrations, lower values were found in the period after lambing, whereas the values obtained shortly before lambing and from week 6 after lambing were higher. Boudebza et al. (2016) recorded lower albumin concentrations in ewes postpartum during early lactation, but the results presented by Piccione et al. (2012) and El-Sherif and Assad (2001) showed that serum albumin concentrations were highest in ewes during lactation. Similarly, Janků et al. (2011) reported the highest albumin concentrations in dairy goats during lactation from day 28 after parturition. In dairy cows, a general decrease in albumin concentrations was observed after parturition (Park et al., 2010; Grünberg et al., 2011). However, according to Yokus et al. (2006), because of substantial changes in plasma volume during the most critical period of pregnancy, albumin concentrations are difficult to interpret. In the concentrations of α-globulins, no significant changes were observed during the peripartal period, but the values were slightly higher in the later postpartal period. In dairy goats, slightly higher concentrations of α1-globulins were obtained by Janků et al. (2011) 7 days after parturition, while the α2-globulins were significantly higher shortly after parturition, as well as 28 days after parturition, when compared with those before parturition. In dairy cows, increasing values of α-globulins were found postpartum as reported by Piccione et al. (2011). Their findings were related to inflammatory processes occurring in the urogenital system after calving. The alterations in the concentrations of α-globulin in ewes throughout the peripartal period are not completely understood. This fraction is composed of many important inflammatory proteins, from which serum amyloid A, haptoglobin, and fibrinogen are diagnostically the most valuable in sheep. Greguła-Kania et al. (2020) found an increase in the concentrations of serum amyloid A and fibrinogen and a decrease in haptoglobin values in ewes in the peripartal period. Our results indicate that the possible increases in the concentrations of acute phase proteins in ewes after lambing are not marked enough to induce observable alterations in the serum protein electrophoretic patterns, and it appears that the regulatory mechanisms of peripartal processes in ewes differ from those in dairy cows. Furthermore, intensive lactation represents a type of metabolic stress that may also induce inflammatory processes and increase the production of inflammatory proteins in high-yielding female animals. However, the ewes included in our study were not high-yielding and only produced milk for their newborns. Therefore, they were not markedly affected by the metabolic stress caused by lactation. Differences in the distribution of protein fractions have been observed previously by Miglio et al. (2015) between Lacaune and Sarda sheep, and they stated that the serum protein electrophoretic pattern varies according to breed as well as metabolic peculiarities. It follows from the above that further studies are needed to describe the possible changes in individual serum proteins throughout the peripartal period.
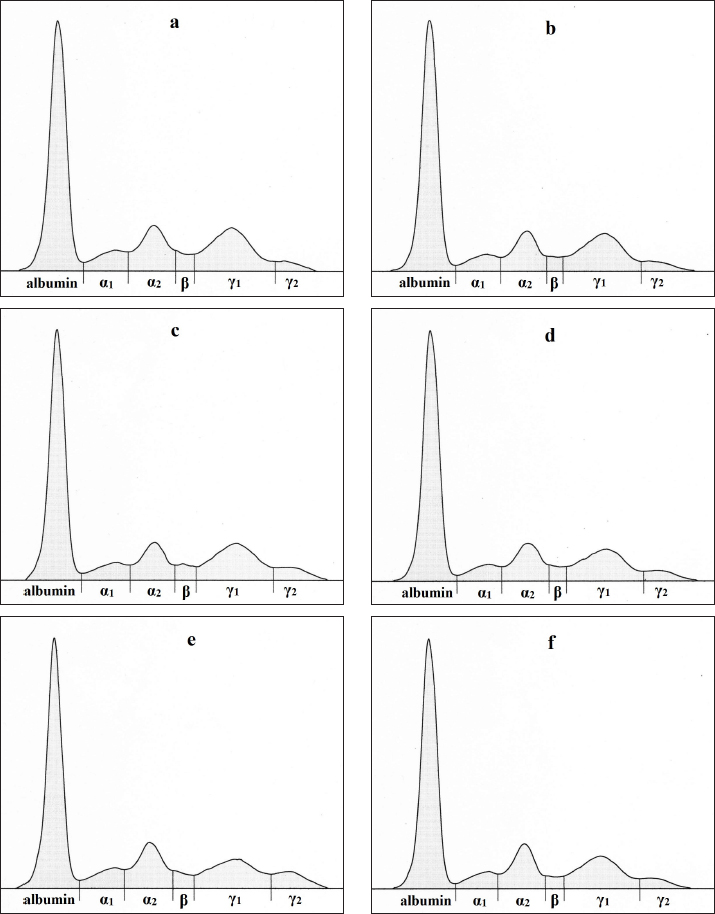
Fig. 1. (a-f): Representative electrophoretograms in ewes showing the protein fractionation of serum proteins into six bands: albumin, α1-, α2-, β-, γ1-, and γ1-globulins during the monitored period - 4 (a) and 2 weeks (b) before, 2 (c), 4 (d), 6 (e), and 8 (f) weeks after lambing. Similarly, the β-globulin concentrations have not been significantly affected by the reproductive stage in the sheep we monitored. Slightly lower values were obtained after lambing, probably due to the transfer of some immunoglobulins, which migrate into this fraction (especially IgA and IgM) from the bloodstream to the mammary gland for the production of colostrum. A more marked decrease after lambing was observed in the concentrations of γ1-globulins, seeing that this fraction contains the majority of immunoglobulins, especially IgG, which are transferred to the mammary gland for colostrum synthesis. Batavani et al. (2006) evaluated Makuii ewes during diestrus and pregnancy and observed a significant decrease in the concentrations of β-, γ1-, as well as γ2-globulins already on day 145 of gestation. In dairy goats, a significant decrease of γ-globulin concentrations was recorded by Janků et al. (2011) on the day of parturition, with a subsequent increase from day 7 after parturition to values higher than those measured 3 weeks before parturition. In ewes included in our study, the γ1-globulin concentrations started to increase from week 6 after lambing, but were still lower than the values obtained before lambing. The γ2-globulin concentrations started to increase from week 4 after lambing and reached higher values than before lambing. In dairy cows, the γ-globulin concentrations decreased and slightly increased from the third week of lactation (Tóthová et al., 2018). All these changes in the concentrations of A/G fractions resulted in changes in the A/G ratio, with the lowest mean value being 4 weeks before lambing and the highest being 2 weeks after lambing. Piccione et al. (2012) recorded an increase in the A/G ratio in ewes during early and mild lactation because of the drop in globulin fractions and higher albumin concentrations. In contrast, dairy cows had higher A/G ratios before parturition and a gradual decrease during early lactation (Tóthová et al., 2018). The results of our study showed significant changes in the distribution of several blood serum proteins in ewes with low milk production, raised especially for the production of lambs. Although the concentrations of total serum proteins did not vary significantly throughout the evaluated peripartal period, significant changes were observed in the concentrations of some protein fractions, especially the albumin and γ-globulin fractions. Although albumin concentrations were lower 4 weeks before lambing and higher in the later postpartal period, the γ1-globulin values showed a gradual decrease until 4 weeks after lambing. These results suggest that the changes observed in ewes around the time of parturition may differ according to the type and breed of sheep, as well as their productive performances, and may differ from the changes observed in dairy goats or cows. Furthermore, our results suggest that changes in the distribution of blood serum proteins found in ewes around parturition are not always a result of pathological processes but may be related to physiological processes occurring around the time of parturition. The data obtained may help to better understand the physiological adaptations that occur in female organisms during the transitional period. Conflict of interestThe authors declare no conflict of interest. FundingThis work was financially supported by the Ministry of Education, Science, Research, and Sport of the Slovak Republic (grant no. VEGA 1/0177/22 and VEGA 1/0569/24). Author’s contributionsVG: Methodology, Resources. CT: Investigation, Methodology, Writing–Original Draft Preparation. VO: Methodology, Resources. ON: Conceptualization, Project Administration, Validation, and Writing—Review and Editing. All authors have read and approved the final version of the manuscript. Data availabilityAll data pertaining to the current study are available from the corresponding authors upon reasonable request. ReferencesAntunović, Z., Novoselec, J., Šperanda, M., Vegara, M., Pavić, V., Mioč, B. and Djidara, M. 2011. Changes in biochemical and hematological parameters and metabolic hormones in Tsigai ewes blood in the first third of lactation. Arch. Anim. Breed. 54, 535–545. Antunović, Z., Senčić, Đ., Šperanda, M. and Liker, B. 2002. Influence of the season and the reproductive status of ewes on blood parameters. Small Rumin. Res. 45, 39–44. Batavani, R.A., Ansari, M.H. and Asri, S. 2006. Concentrations of serum total protein and protein fractions during diestrus and pregnancy in Makuii ewes. Com. Clin. Pathol. 15, 227–230. Baumgartner, W. and Pernthaner, A. 1994. Influence of age, season, and pregnancy upon blood parameters in Austrian Karakul sheep. Small Rumin. Res. 13, 147–151. Block, S.S., Butler, W.R., Ehrhardt, R.A., Bell, A.W., Van Amburgh, M.E. and Boisclair, Y.R. 2001. Decreased concentration of plasma leptin in periparturient dairy cows is caused by negative energy balance. J. Endocrin. 171, 339–348. Boudebza, A., Arzour-Lakhel, N., Abdeldjelil, M.C., Dib, A.L., Lakhdara, N., Benazzouz, H. and Benlatreche, C. 2016. Blood biochemical parameters in Ouled Djellal ewes in the periparturient period. Pharm. Chem. 8, 406–410. Bremmer, D.R., Bertics, S.J., Besong, S.A. and Grummer, R.R. 2000. Changes in hepatic microsomal triglyceride transfer protein and triglyceride in periparturient dairy cattle. J. Dairy Sci. 83, 2252–2260. Djuricic, D., Dobranic, T., Grizelj, J., Gracner, D., Harapin, I., Stanin, D., Folnozic, I., Getz, I., Cvitkovic, D. and Samardzija, M. 2011. Concentrations of total proteins and albumins, and AST, AP, CK and GGT activities in the blood serum Boer and Saanen goats during puerperium. Reprod. Domest. Anim. 46, 674–677. El-Sherif, M.M.A. and Assad, F. 2001. Changes in some blood constituents of Barki ewes during pregnancy and lactation under semi arid conditions. Small Rumin. Res. 40, 269–277. Elzein, E., Osman, I. and Omer, S.A. 2016. Effect of physiological status in some haematological and biochemical parameters in desert goats. Int. J. Vet. Sci. 5, 95–98. Greguła-Kania, M., Kosior-Korzecka, U., Patkowski, K., Juszczuk-Kubiak, E., Plewik, M. and Gruszecki, T.M. 2020. Acute-phase proteins, cortisol and haematological parameters in ewes during the periparturient period. Reprod. Domest. Anim. 55, 393–400. Grünberg, W., Donkin, S.S. and Constable, P.D. 2011. Periparturient effects of feeding a low dietary cation-anion difference diet on acid-base, calcium, and phosphorus homeostasis and on intravenous glucose tolerance test in high-producing dairy cows. J. Dairy Sci. 94, 727–745. Iriadam, M. 2007. Variation in certain hematological and biochemical parameters during the peri-partum period in Kilis does. Small Rumin. Res. 73, 54–57. Jania, B. and Andraszek, K. 2016. Application of native agarose gel electrophoresis of serum proteins in veterinary diagnostics. J. Vet. Res. 60, 501–508. Janků, L., Pavlata, L., Mišurová, Ľ., Filípek, J., Pechová, A. and Dvořák, R. 2011. Levels of protein fractions in blood serum of periparturient goats. Acta Vet. Brno. 80, 185–190. Karapehlivan, M., Atakisi, E., Atakisi, O., Yucayurt, R. and Pancarci, S.M. 2007. Blood biochemical parameters during the lactation and dry period in Tuj ewes. Small Rumin. Res. 73, 267–271. Keren, D.F., Alexanian, R., Goeken, J.A., Gorevic, P.D., Kyle, R.A. and Tomar, R.H. 1999. Guidelines for clinical and laboratory evaluation of patients with monoclonal gammopathies. Arch. Pathol. Lab. Med. 123, 106–107. King, J.C. 2000. Physiology of pregnancy and nutrient metabolism. Am. J. Clin. Nutr. 71, 1218S–1225S. Krajnicakova, M., Bekeova, E., Hendrichovský, V. and Maracek, I. 1993. Levels of total lipids, cholesterol and progesterone during estrus synchronization and pregnancy in sheep. Vet. Med. Czech. 38, 349–357. Miglio, A., Antognoni, M. T., Maresca, C., Moncada, C., Riondato, F., Scoccia, E. and Mangili, V. 2015. Serum protein concentration and protein fractions in clinically healthy Lacaune and Sarda sheep using agarose gel electrophoresis. Vet. Clin. Pathol. 44, 564–569. O’Connell, T.X., Horita, T.J. and Kasravi, B. 2005. Understanding and interpreting serum protein electrophoresis. Am. Fam. Physician. 71, 105–112. Park, A.F., Shirley, J.E., Titgemeyer, E.C., Cochran, R.C., DeFrain, J.M., Wickersham, E.E. and Johnson, D.E. 2010. Characterization of plasma metabolites in Holstein dairy cows during the periparturient period. Int. J. Dairy Sci. 5, 253–263. Piccione, G., Alberghina, D., Marafioti, S., Giannetto, C., Casella, S., Assenza, A. and Fazio, F. 2012. Electrophoretic serum protein fraction profile during the different physiological phases in Comisana ewes. Reprod. Domest. Anim. 47, 591–595. Piccione, G., Caola, G., Giannetto, C., Grasso, F., Runzo, S.C., Zumbo, A. and Pennisi, P. 2009. Selected biochemical serum parameters in ewes during pregnancy, post-parturition, lactation and dry period. Anim. Sci. Pap. Rep. 27, 321–330. Piccione, G., Messina, V., Schembari, A., Casella, S., Giannetto, C. and Alberghina, D. 2011. Pattern of serum protein fractions in dairy cows during different stages of gestation and lactation. J. Dairy Res. 78, 421–425. Roubies, N., Panousis, N., Fytianou, A., Katsoulos, P. D., Giadinis, N. and Karatzias, H. 2006. Effects of age and reproductive stage on certain serum biochemical parameters of Chios sheep under Greek rearing conditions. J. Vet. Med. A. 53, 277–281. Saeed, A., Khan, I.A.A. and Hussein, M.M. 2009. Change in biochemical profile of pregnant camels (Camelus dromedarius) at term. Comp. Clin. Pathol. 18, 139–143. Soares, G.S.L., Souto, R.J.C., Cajueiro, J.F.P., Afonso, J.A.B., Rego, R.O., Macêdo, A.T.M., Soares, P.C. and Mendonça, C.L. 2018. Adaptive changes in blood biochemical profile of dairy goats during the period of transition. Rev. Méd. Vet. 169, 65–75. Tóthová, C., Nagy, O., Nagyová, V. and Kováč, G. 2018. Serum protein electrophoretic pattern in dairy cows during the periparturient period. J. Appl. Anim. Res. 46, 33–38. Vavricka, S.R., Burri, E., Beglinger, C., Degen, L. and Manz, M. 2009. Serum protein electrophoresis: an underused but very useful test. Digestion 79, 203–210. Yokus, B., Cakir, D.U., Kanay, Z., Gulten, T. and Uysal, E. 2006. Effects of seasonal and physiological variations on the serum chemistry, vitamins and thyroid hormone concentrations in sheep. J. Vet. Med. A. 53, 271–276. Zvorc Z., Matijatko, V., Beer, B., Foršek, J., Bedrica, L. and Kučer, N. 2000. Blood serum proteinograms in pregnant and non-pregnant cows. Vet. Arh. 70, 21–30. | ||
| How to Cite this Article |
| Pubmed Style Glembová V, Tóthová C, Oršuľaková V, Nagy O. Changes in blood serum protein profile in low-yielding ewes during the periparturient period. Open Vet. J.. 2025; 15(2): 893-900. doi:10.5455/OVJ.2025.v15.i2.38 Web Style Glembová V, Tóthová C, Oršuľaková V, Nagy O. Changes in blood serum protein profile in low-yielding ewes during the periparturient period. https://www.openveterinaryjournal.com/?mno=231389 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.38 AMA (American Medical Association) Style Glembová V, Tóthová C, Oršuľaková V, Nagy O. Changes in blood serum protein profile in low-yielding ewes during the periparturient period. Open Vet. J.. 2025; 15(2): 893-900. doi:10.5455/OVJ.2025.v15.i2.38 Vancouver/ICMJE Style Glembová V, Tóthová C, Oršuľaková V, Nagy O. Changes in blood serum protein profile in low-yielding ewes during the periparturient period. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 893-900. doi:10.5455/OVJ.2025.v15.i2.38 Harvard Style Glembová, V., Tóthová, . C., Oršuľaková, . V. & Nagy, . O. (2025) Changes in blood serum protein profile in low-yielding ewes during the periparturient period. Open Vet. J., 15 (2), 893-900. doi:10.5455/OVJ.2025.v15.i2.38 Turabian Style Glembová, Veronika, Csilla Tóthová, Veronika Oršuľaková, and Oskar Nagy. 2025. Changes in blood serum protein profile in low-yielding ewes during the periparturient period. Open Veterinary Journal, 15 (2), 893-900. doi:10.5455/OVJ.2025.v15.i2.38 Chicago Style Glembová, Veronika, Csilla Tóthová, Veronika Oršuľaková, and Oskar Nagy. "Changes in blood serum protein profile in low-yielding ewes during the periparturient period." Open Veterinary Journal 15 (2025), 893-900. doi:10.5455/OVJ.2025.v15.i2.38 MLA (The Modern Language Association) Style Glembová, Veronika, Csilla Tóthová, Veronika Oršuľaková, and Oskar Nagy. "Changes in blood serum protein profile in low-yielding ewes during the periparturient period." Open Veterinary Journal 15.2 (2025), 893-900. Print. doi:10.5455/OVJ.2025.v15.i2.38 APA (American Psychological Association) Style Glembová, V., Tóthová, . C., Oršuľaková, . V. & Nagy, . O. (2025) Changes in blood serum protein profile in low-yielding ewes during the periparturient period. Open Veterinary Journal, 15 (2), 893-900. doi:10.5455/OVJ.2025.v15.i2.38 |