| Research Article | ||
Open Vet. J.. 2025; 15(2): 923-930 Open Veterinary Journal, (2025), Vol. 15(2): 923-930 Research Article Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus)Budiastuti Budiastuti1, Naomi Lan Noviana Thesia2, Hani Plumeriastuti3*, Mustofa Helmi Effendi4, Emmanuel Nnabuike Ugbo5, Aswin Rafif Khairullah6, Ikechukwu Benjamin Moses5, Katty Hendriana Priscilia Riwu7, Riza Zainuddin Ahmad6, Sheila Marty Yanestria8, Wasito Wasito6, Bima Putra Pratama9, Mo Awwanah10 and Agung Prasetyo111Study Program of Pharmacy Science, Faculty of Health Science, Universitas Muhammadiyah Surabaya, Surabaya, Indonesia 2Master Program of Veterinary Science and Public Health, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 3Division of Veterinary Pathology, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 4Division of Veterinary Public Health, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 5Department of Applied Microbiology, Faculty of Science, Ebonyi State University, Abakaliki, Nigeria 6Research Center for Veterinary Science, National Research and Innovation Agency (BRIN), Bogor, Indonesia 7Department of Veterinary Public Health, Faculty of Veterinary Medicine, Universitas Pendidikan Mandalika, Mataram, Indonesia 8Faculty of Veterinary Medicine, Universitas Wijaya Kusuma Surabaya, Surabaya, Indonesia 9Research Center for Agroindustry, National Research and Innovation Agency (BRIN), Bogor, Indonesia 10Research Center for Applied Botany, National Research and Innovation Agency (BRIN), Bogor, Indonesia 11Research Center for Estate Crops, National Research and Innovation Agency (BRIN), Bogor, Indonesia *Corresponding Author: Hani Plumeriastuti. Division of Veterinary Pathology, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: hani-p [at] fkh.unair.ac.id Submitted: 21/12/2024 Accepted: 22/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
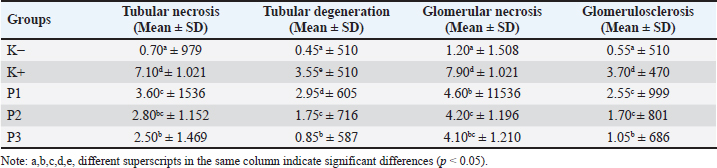
AbstractBackground: Chronic systemic disease known as diabetes mellitus is defined by elevated blood glucose levels and problems with fat, carbohydrate, and protein metabolism. In Indonesia, the cinnamon species found include Cinnamomum burmannii, which also has hypoglycemic activity. The primary antioxidant chemicals found in C. burmannii bark extract are polyphenols, which include tannins and flavonoids. Aim: The antidiabetic activity of cinnamon essential oil (C. burmannii) against streptozotocin (STZ) was examined in this study. Methods: STZ (45 mg/kg BW) was administered intraperitoneally as a single dose. Twenty male rats were employed in this investigation. The rats were divided into five groups: treatment 1 (P1) was administered with STZ and provided with 100 mg/kg BW; treatment 2 (P2) was administered with STZ and provided with 200 mg/kg BW; and treatment 3 (P3) was administered with STZ and provided with 400 mg/kg BW. The negative control group (K−) was not treated with STZ and was treated with 1% Tween 80. For 14 days, the medication was administered daily. Results: The essential oil of cinnamon may lessen glomerulosclerosis, tubular necrosis, tubular degeneration, and glomerular necrosis. The kidney P3 treatment (400 mg/kg BW) produced negligible effects. Conclusion: It is possible to create cinnamon essential oil as an herbal antidiabetic medication by lowering the degree of kidney cell damage. Keywords: Cinnamomum burmannii, Essential oil, Kidney, Streptozotocin, Antidiabetic. IntroductionChronic systemic disease known as diabetes mellitus (DM) is defined by elevated blood sugar levels and problems with the metabolism of fats, carbohydrates, and proteins (American Diabetes Association, 2018). DM occurs due to abnormalities in insulin secretion, insulin function, or both. Types 1 and 2 DM are the two categories into which DM is separated. Type 1 DM is caused by autoimmune disorders in the body that cause damage to pancreatic β cells, so insulin production is insufficient and the body experiences insulin deficiency. Type 2 DM is caused by the body’s inability to use insulin or insulin resistance (American Diabetes Association, 2010). High blood sugar levels increase the production of reactive oxygen species (ROS) because they impair natural antioxidant enzymes like superoxide dismutase and increase the levels of advanced glycation end products (Khalid et al., 2022). Oxidative stress plays a major role in the occurrence of diabetic nephropathy, which is one of the complications of hyperglycemia in the body. In people with DM, high blood glucose levels can cause progressive kidney damage, both in tubule cells and glomeruli, leading to chronic kidney disease (Krishan and Chakkarwar, 2011). Related research exploring active substances in plants has been conducted. Among them, several plant species have been found to have antidiabetic activity that can lower blood glucose levels or repair pancreatic β cells. In Indonesia, the cinnamon species found include Cinnamomum burmannii, which also has hypoglycemic activity (Pulungan and Pane, 2020). Cinnamon plants are species of the genus Cinnamomum and the Lauraceae family. They are woody plants commonly known as spices (Rao and Gan, 2014). Cinnamon contains low-fat content and has antibacterial, antifungal, antiviral, antioxidant, anticancer, and blood pressure-lowering properties (Nabavi et al., 2015). Eugenol and cinnamaldehyde compounds have potential as antidiabetic and antibiofilm (Olszewska et al., 2020). The primary antioxidant chemicals found in C. burmannii bark extract are polyphenols, which include tannins and flavonoids. These antioxidants can suppress the formation of ROS, which plays a major role in kidney tubule and glomerular damage (Ervina et al., 2016). However, despite the abundance of literature related to antidiabetes, the use of cinnamon essential oil has been found to be very little related to kidney damage prevention. Furthermore, the primary components of cinnamon essential oil are chemicals called cinnamaldehyde and eugenol, which may have the ability to influence the α-glucosidase enzyme and be developed into an antidiabetic agent (Stevens and Allred, 2022). Based on these findings, researchers aimed to investigate the effect of cinnamon essential oil on the histopathology of the kidneys of streptozotocin (STZ)-induced white rats. Disclosure of results from experimental animals will strengthen our understanding of the field of veterinary medicine, especially kidney histopathology. Materials and MethodsThe type of research is an experimental study (True experimental) with a post-test random control approach. The research was conducted at the Experimental Animal Laboratory, Faculty of Medicine, Airlangga University, Surabaya for the maintenance of experimental animals and treatment. The manufacture of cinnamon essential oil was carried out at PT Hiptasari, and the manufacture and observation of histopathology preparations of white rat kidneys were carried out at the Department of Veterinary Pathology, Faculty of Veterinary Medicine, Airlangga University. This research was conducted from August 2019 to April 2020. The research began with the production of cinnamon essential oil (C. burmannii) by steam distillation without washing. Adaptation of the white rats for 7 days was then induced with 0.5 ml of STZ at a dose of 45 mg/kg BW administered intraperitoneally. In this study, the essential oil doses used were 100, 200, and 400 mg/kg BW. Kidney organ removal was performed after 14 days of treatment, and preparation of kidney preparations (Ding et al., 2011). The examination results in the form of Tubular epithelial degeneration scores, tubular epithelial necrosis, glomerular necrosis, and glomerulosclerosis were tested using the Kruskal–Wallis test followed by the Mann–Whitney test. The statistical analysis in this study used the Statistical Program of Social Sciences (Ding et al., 2011). ResultIn this study, a semi-qualitative assessment (scoring) was carried out according to the modified Klopfleisch (2013) method. The results of the microscopic examination of the kidneys are presented in Table 1. Tubular hydropic degenerationIn the kidney preparation of group K, a normal picture of the renal tubules of white rats was observed, whereas, in group K+ induced with STZ, the histopathology of the kidneys of white rats experienced degeneration in the tubular epithelium, especially in the proximal area (Fig. 1). The statistical test, the Kruskal–Wallis test, was used to analyze renal tubular cell degeneration in STZ-induced white rats. The results indicated a significant difference (p < 0.05). Table 1. Sample examination results.
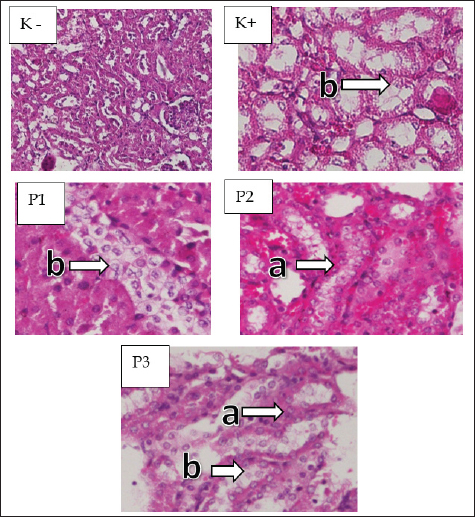
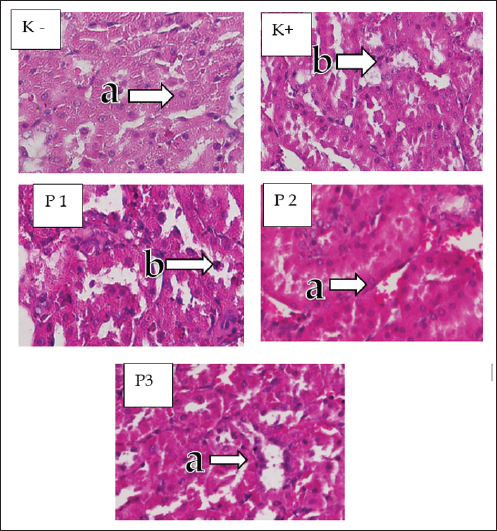
The highest hydropic degeneration score of tubular cells was observed in the K+ group (3.55 ± 510) and the lowest cell degeneration score was observed in the K group (0.45 ± 510). In the treatment group using cinnamon essential oil (C. burmannii), a P3 cinnamon essential oil dose of 400 mg/kg BW had the lowest tubular cell degeneration score (0.85 ± 587) followed by P2 (1.75 ± 716) and P1 (2.95 ± 605). Based on the Mann–Whitney test between groups presented in the table, each group was significantly different. Tubular cell necrosisKidney preparations in the K group showed normal renal tubules, whereas in the K+ group induced with STZ, the histopathology of the kidneys of white rats showed necrosis in the tubules, especially in the proximal area (Fig. 2). A statistical test of tubular necrosis score assessment using the Kruskal–Wallis test yielded significant differences (p < 0.05) in the data analysis. The highest score of renal cell tubular necrosis was obtained in the K+ group (7.10 ± 1.021). The smallest tubular necrosis score was obtained in the K− group (0.70 ± 979). P3 showed the lowest tubular necrosis value (2.50 ± 1.469) from the P2 (2.80 ± 1.152) and P1 (3.60 ± 1536) groups. Based on the Mann–Whitney test presented in Table 1, K− had a significant difference with groups P1, P2, P3, and K+. The K+ group exhibited a significant difference compared with groups P1, P2, P3, and K−. Group P1 differed significantly from P3, K−, and K + but not significantly from P2. Group P2 showed a significant difference between groups P1 and K, and K+ was not significantly different from group P3. Glomerular necrosisThe results of the K− group preparation showed normal glomeruli, whereas the K+ group showed necrosis in the glomeruli, which was because the K+ group was induced with STZ without administering Cinnamon essential oil (Fig. 3). A significant difference (p < 0.05) was found in the data analysis from the statistical test of the renal glomerular necrosis score assessment using the Kruskal–Wallis test. The K+ group had the highest glomerular necrosis score (7.90 ± 1.021), whereas the K group had the lowest glomerular necrosis score (1.20 ± 1.508). In the cinnamon essential oil treatment group, P1 had the highest glomerular necrosis score (4.60 ± 11536), whereas P2 had a glomerular necrosis score (4.20 ± 1.196) and the lowest glomerular necrosis value in the cinnamon essential oil treatment group was P1 (4.10 ± 1.210). Based on the Mann–Whitney test analysis to determine the differences in each group, as presented in Table 1 shows that K− has a significant difference with P1, P2, P3, and K+. The K+ group was significantly different from the P1, P2, P3, and K groups. Group P2 differs considerably from K−, K+, and P1 but not significantly from P3, whereas treatment group P1 differs significantly from groups K−, K+, and P2 but not significantly from P3.
Fig. 1. Microscopic image of degeneration of white rat kidney cells (H&E staining with 200x magnification) groups: K, K+, P1, P2, and P3. (a) normal cells (b) hydropic degeneration.
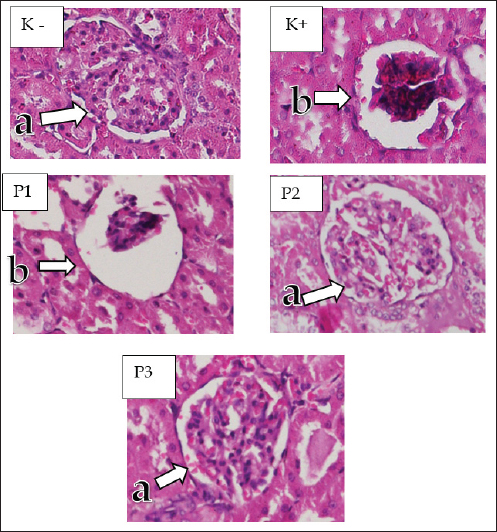
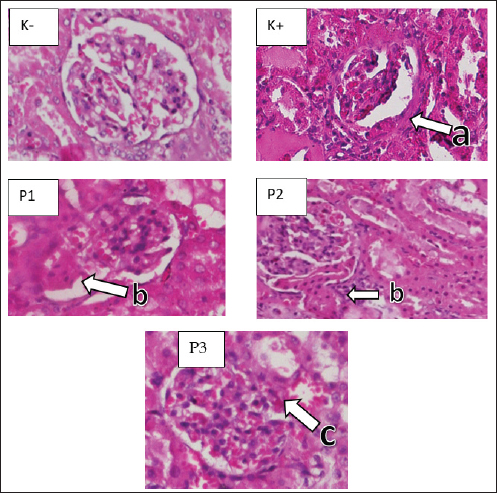
Fig. 2. Microscopic image of tubular cell necrosis. Renal cortex (H&E staining with 200x differed significantly frommagnification) K−, K+, P1, P2, and P3 groups. (a) normal peritubular cells (b) tubular cell necrosis. GlomerulosclerosisIn the kidney preparations of the K group, a normal picture of the kidney glomerulus was observed, whereas in the K+ group induced with STZ, a histopathological picture of the kidneys of white rats experiencing glomerulosclerosis was shown (Fig. 4). A significant difference (p < 0.05) was found in the data analysis from the statistical test of glomerulosclerosis score assessment using the Kruskal–Wallis test. The K+ group had the highest glomerulosclerosis necrosis score (3.70 ± 470), whereas the K group had the lowest glomerular necrosis score (0.55 ± 510). In the cinnamon essential oil treatment group, P1 had the highest glomerulosclerosis score (2.55 ± 999), whereas P2 had a glomerulosclerosis score (1.70 ± 801) and the lowest glomerulosclerosis value in the cinnamon essential oil treatment group was P3 (1.05 ± 686). Based on the Mann–Whitney test analysis to determine the differences in each group, as presented in Table 1, it shows that K has a significant difference with P1, P2, P3, and K+. There was a substantial difference between the K+ group and the P1, P2, P3, and K− groups. Group P3 was considerably different from K−, K+, P1, and P2, whereas the P1 treatment group was significantly different from groups K−, K+, and P2, but not from P2.
Fig. 3. Microscopic image of glomerular necrosis. Kidney cortex of white rats (H&E staining with 200x magnification) groups: K, K+, P1, P2, and P3 (a) Normal Glomerulus (b) Glomerular necrosis. DiscussionIn this study, administration of STZ 45 mg/kg BB resulted in a significant increase in blood sugar levels in the rats. The lowest fasting blood glucose levels were observed in the control group (normal), whereas the positive control and treatment groups had the highest fasting blood glucose levels. The findings of this investigation are consistent with those of Ghasemi and Jeddi’s (2023) research, and STZ administration to white rats (Rattus norvegicus) can cause hyperglycemia 84 hours after intraperitoneal injection. Rats are considered hyperglycemic if their blood glucose levels are >135 mg/dl (Ali and Ali, 2022). Hydropic degeneration occurs due to disruption of the cell membrane, resulting in fluid entering the cytoplasm and vacuoles. In this study, the average renal tubular hydropic degeneration was significantly different between the treatment and control groups. This indicates the presence of acute diabetic nephropathy in the renal tubules, which results in a decrease in the mechanism of kidney function. Accumulation of fluid in the cytoplasm occurs because of decreased cell function in balancing fluids. Hyperglycemia due to STZ administration increases intravascular ROS production, which affects cell permeability and leads to the formation of hydropic degeneration lesions in the renal tubules (Nakamoto et al., 2014). Necrosis is cell death due to injury. In this study, the average results of renal tubular necrosis showed a significant difference between the negative control group (normal) and positive control groups, indicating that the renal tubules and glomeruli are very susceptible to cell death due to high blood glucose. Hyperglycemia due to STZ induction can increase oxidative stress and cause inflammatory reactions that trigger the release of proinflammatory cytokines, such as tumor necrosis factor-alpha (TNF-α). TNF-α is the main inducer of renal microvascular inflammation and plays a role in the progression of tubular and glomerular damage (Ramseyer and Garvin, 2013). Tubular and renal glomerular necrosis results in blood unfiltration, which results in the body’s metabolic waste not being able to be removed. If cell death in the tubules and glomeruli continues, chronic disease and kidney failure will occur.
Fig. 4. Microscopic image of glomerulosclerosis. Renal cortex of white rats (H&E staining with 200x magnification) groups: K, K+, P1, P2, and P3 (a) fibrosis in the parietal layer (b) focal glomerulosclerosis (c) (synechiae) adhesions between the glomerulus and the parietal layer. Glomerulosclerosis in diabetic nephrotes is caused by the accumulation of extracellular matrix in the interstitial mesangial space of the renal glomerulus (Qian et al., 2008). The most abundant matrix proteins finglomerulosclerosis are collagen types I, III, IV, and fibronectin (Bülow and Boor, 2019). The results of the study showed that glomerulosclerosis was still in its early stages, and the glomerular nodules that formed were small. This result proves that STZ at 45 mg/kg BW can trigger the formation of glomerulosclerosis. Cinnamon is an herbal plant commonly used as a natural diabetes medication. Cinnamon has a flavonoid compound, namely methyhydroxy chalcone polymer (MHPC) (Qureshi et al., 2019). MHCP has the ability like insulin, namely to activate glycogen synthesis, increase glucose uptake, activate insulin receptor kinase, and inhibit insulin receptor dephosphorylation (Wang et al., 2022). Treatment group P1 cinnamon attrition oil dose of 100 mg/kg BW had the highest tubular degeneration, tubular necrosis, glomerular necrosis, and glomerulosclerosis values compared with groups P2 and P3. This is because the amount of antioxidants at that dose cannot balance the amount of ROS in the kidneys of streptozootocin-induced white mice. In group P3, we observed low values of tubular degeneration, tubular necrosis, glomerular necrosis, and glomerulosclerosis compared with group P1 and no significant difference with group P2, so the use of cinnamon essential oil at a dose of 400 and 200 mg/kg BW has antioxidant properties that can inhibit the formation of oxidative stress in the kidneys, but the higher the dose of cinnamon essential oil given, the higher the antioxidant properties produced to ward off free radicals. The primary components of cinnamon essential oil are eugenol and cinnamaldehyde, which have antibacterial and antioxidant properties (Behbahani et al., 2020). According to research by Hayward et al. (2019), the molecule cinnamaldehyde, which was extracted from cinnamon oil, has an IC50 value of 27.96 ppm against the α-glucosidase enzyme, indicating that it has great potential as an antidiabetic compound that inhibits the activity of the α-glucosidase enzyme. The presence of cinnamaldehyde molecules in cinnamon essential oil has a preventive effect on damage to rat kidney cells. This study was supported by research conducted by Hayward et al. (2019). In the histopathological description of the kidneys, the structure of the kidney cells at P2 and P3 appears normal. Antioxidant therapy inhibits the production of intracellular free radicals and increases the ability of defense enzymes to prevent the emergence of oxidative stress and vascular complications related to diabetes. Cinnamon essential oil can treat kidney damage caused by oxidative stress and free radical buildup in the kidneys. The antioxidants in cinnamon essential oil work by donating their electrons to unstable cells. ConclusionThe results of the study indicate that the histological images of the kidneys of white rats (R. norvegicus) generated by STZ are altered by the administration of cinnamon essential oil (C. burmannii). Cinnamomum burmannii essential oil has an effective dosage of 400 mg/kg BW. The current study has some limitations that only focused on the histopathology of rat kidneys. Therefore, further research on the pancreatic organ as a producer of insulin is needed. AcknowledgmentsThe author is grateful to Universitas Airlangga. Conflict of interestThe authors declare no conflict of interest. FundingThis study was partly supported by The International Research Consortium, Lembaga Penelitian dan Pengabdian Masyarakat, Universitas Airlangga, Surabaya, Indonesia, in 2024 (grant number: 171/UN3.LPPM/PT.01.03/2024). Author’s contributionsBB, SMY, and HP: Conceptualization and design. NLNT and KHPR: Data acquisition. MHE, RZA, and IBM: Formal analysis and interpretation of data. ARK, AP, and MA: Writing-original draft preparation. ENU, BPP, and WW: Writing-review and editing. All authors have read and agreed to the publication of the final version of the manuscript. Data availabilityAll data are available in the manuscript. ReferencesAli, S.K. and Ali, R.H. 2022. Effects of antidiabetic agents on Alzheimer’s disease biomarkers in an experimentally induced hyperglycemic rat model by streptozocin. PLoS One 17(7), e0271138. American Diabetes Association. 2010. Diagnosis and classification of diabetes mellitus. Diabetes Care. 33(Suppl 1), S62–S69. American Diabetes Association. 2018. Diagnosis and classification of diabetes mellitus. Diabetes Care. 32 (Suppl 1), S62–S67. Behbahani, B.A., Falah, F., Arab, F.L., Vasiee, M. and Yazdi, F.T. 2020. Chemical composition and antioxidant, antimicrobial, and antiproliferative activities of Cinnamomum zeylanicum bark essential oil. Based Complementary Altern. Med. 2020(1), 5190603. Bülow, R.D. and Boor, P. 2019. Extracellular matrix in kidney fibrosis: more than just a scaffold. J. Histochem. Cytochem. 67(9), 643–661. Ding, Y., Zou, J., Li, Z., Tian, J., Abdelalim, S., Du, F., She, R., Wang, D., Tan, C., Wang, H., Chen, W., Lv, D. and Chang, L. 2011. Study of histopathological and molecular changes of rat kidney under simulated weightlessness and resistance training protective effect. PLoS One 6(5), e20008. Ervina, M., Nawu, Y.E. and Esar, S.Y. 2016. Comparison of in vitro antioxidant activity of infusion, extract, and fractions of Indonesian cinnamon (Cinnamomum burmannii) Bark. Int. Food Res. J. 23(3), 1346–1350. Ghasemi, A. and Jeddi, S. 2023. Streptozotocin as a tool for induction of rat models of diabetes: a practical guide. EXCLI J. 22(1), 274–294. Hayward, N.J., McDougall, G.J., Farag, S., Allwood, J.W., Austin, C., Campbell, F., Horgan, G. and Ranawana, V. 2019. Cinnamon has species-specific antidiabetic properties: effects of enzyme activity inhibition and starch digestion. Plant Foods Hum. Nutr. 74(4), 544–552. Khalid, M., Petroianu, G. and Adem, A. 2022. Advanced glycation end products and diabetes mellitus: mechanisms and perspectives. Biomolecules 12(4), 542. Klopfleisch, R. 2013. Multiparametric and semiquantitative scoring systems for the evaluation of mouse model histopathology—a systematic review. BMC Vet. Res. 9(1), 123. Krishan, P. and Chakkarwar, V.A. 2011. Diabetic nephropathy: aggressive involvement of oxidative stress. J. Pharm. Edu. Res. 2(1), 35–41. Nabavi, S.F., Di Lorenzo, A., Izadi, M., Sobarzo-Sánchez, E., Dagliam, M. and Nabavim S.M. 2015. The antibacterial effects of cinnamon: from farm on food, cosmetic and pharmaceutical industries. Nutrients 7(9), 7729–7748. Nakamoto, H., Hamada, C., Shimaoka, T., Sekiguchi, Y., Io, H., Kaneko, K., Horikoshi, S. and Tomino, Y. 2014. Accumulation of advanced glycation end products and beta 2-microglobulin in fibrotic thickening of the peritoneum in long-term peritoneal dialysis patients. J. Artif. Organs 17(1), 60–68. Olszewska, M.A., Gędas, A. and Simões, M., 2020. The effects of eugenol, trans-cinnamaldehyde, citronellol, and terpineol on Escherichia coli biofilm control as assessed by culture-dependent and -independent methods. Molecules 25(11), 2641. Pulungan, A. and Pane, Y.S. 2020. The benefit of cinnamon (Cinnamomum burmannii) in lowering total cholesterol levels after consumption of high-fat-containing foods in white mice (Mus musculus) models. F1000Res 9(1), 168. Qian, Y., Feldman, E., Pennathur, S., Kretzler, M. and Brosius, F.C. 2008. From fibrosis to sclerosis: the mechanisms of glomerulosclerosis in diabetic nephropathy. Diabetes 57(6), 1439–1445. Qureshi, A.S. Ghaffor, J., Usman, M., Ehsan, N., Umar, Z. and Sarfraz, A. 2019. Effect of ethanolic preparations of cinnamon (Cinnamomum zeylanicum) extract on hematologic and histometric parameters of selected organs in Alloxan®-induced diabetic female albino rats. J. Diabetes Metab. Disord. 18(2), 505–512. Ramseyer, V.D. and Garvin, J.L. 2013. Tumor necrosis factor-α: regulation of renal function and blood pressure. Am. J. Physiol. Renal Physiol. 304(10), F1231–F1242. Rao, P.V. and Gan, S.H. 2014. Cinnamon: a multifaceted medicinal plant. J. Evid. Based Complementary Altern. Med. 2014(1), 642942. Stevens, N. and Allred, K. 2022. Antidiabetic potential of volatile cinnamon oil: a review and exploration of the mechanisms of in silico molecular docking simulations. Molecules 27(3), 853. Wang, L., Li, J. and Di, L.J. 2022. Glycogen synthesis and beyond, a comprehensive review of GSK3 as a key regulator of metabolic pathways and a therapeutic target for treating metabolic diseases. Med. Res. Rev. 42(2), 946–982. | ||
| How to Cite this Article |
| Pubmed Style Budiastuti B, Thesia NLN, Plumeriastuti H, Effendi MH, Ugbo EN, Khairullah AR, Moses IB, Riwu KHP, Ahmad RZ, Yanestria SM, Wasito W, Pratama BP, Awwanah M, Prasetyo A. Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus). Open Vet. J.. 2025; 15(2): 923-930. doi:10.5455/OVJ.2025.v15.i2.41 Web Style Budiastuti B, Thesia NLN, Plumeriastuti H, Effendi MH, Ugbo EN, Khairullah AR, Moses IB, Riwu KHP, Ahmad RZ, Yanestria SM, Wasito W, Pratama BP, Awwanah M, Prasetyo A. Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus). https://www.openveterinaryjournal.com/?mno=234043 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.41 AMA (American Medical Association) Style Budiastuti B, Thesia NLN, Plumeriastuti H, Effendi MH, Ugbo EN, Khairullah AR, Moses IB, Riwu KHP, Ahmad RZ, Yanestria SM, Wasito W, Pratama BP, Awwanah M, Prasetyo A. Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus). Open Vet. J.. 2025; 15(2): 923-930. doi:10.5455/OVJ.2025.v15.i2.41 Vancouver/ICMJE Style Budiastuti B, Thesia NLN, Plumeriastuti H, Effendi MH, Ugbo EN, Khairullah AR, Moses IB, Riwu KHP, Ahmad RZ, Yanestria SM, Wasito W, Pratama BP, Awwanah M, Prasetyo A. Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus). Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 923-930. doi:10.5455/OVJ.2025.v15.i2.41 Harvard Style Budiastuti, B., Thesia, . N. L. N., Plumeriastuti, . H., Effendi, . M. H., Ugbo, . E. N., Khairullah, . A. R., Moses, . I. B., Riwu, . K. H. P., Ahmad, . R. Z., Yanestria, . S. M., Wasito, . W., Pratama, . B. P., Awwanah, . M. & Prasetyo, . A. (2025) Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus). Open Vet. J., 15 (2), 923-930. doi:10.5455/OVJ.2025.v15.i2.41 Turabian Style Budiastuti, Budiastuti, Naomi Lan Noviana Thesia, Hani Plumeriastuti, Mustofa Helmi Effendi, Emmanuel Nnabuike Ugbo, Aswin Rafif Khairullah, Ikechukwu Benjamin Moses, Katty Hendriana Priscilia Riwu, Riza Zainuddin Ahmad, Sheila Marty Yanestria, Wasito Wasito, Bima Putra Pratama, Mo Awwanah, and Agung Prasetyo. 2025. Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus). Open Veterinary Journal, 15 (2), 923-930. doi:10.5455/OVJ.2025.v15.i2.41 Chicago Style Budiastuti, Budiastuti, Naomi Lan Noviana Thesia, Hani Plumeriastuti, Mustofa Helmi Effendi, Emmanuel Nnabuike Ugbo, Aswin Rafif Khairullah, Ikechukwu Benjamin Moses, Katty Hendriana Priscilia Riwu, Riza Zainuddin Ahmad, Sheila Marty Yanestria, Wasito Wasito, Bima Putra Pratama, Mo Awwanah, and Agung Prasetyo. "Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus)." Open Veterinary Journal 15 (2025), 923-930. doi:10.5455/OVJ.2025.v15.i2.41 MLA (The Modern Language Association) Style Budiastuti, Budiastuti, Naomi Lan Noviana Thesia, Hani Plumeriastuti, Mustofa Helmi Effendi, Emmanuel Nnabuike Ugbo, Aswin Rafif Khairullah, Ikechukwu Benjamin Moses, Katty Hendriana Priscilia Riwu, Riza Zainuddin Ahmad, Sheila Marty Yanestria, Wasito Wasito, Bima Putra Pratama, Mo Awwanah, and Agung Prasetyo. "Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus)." Open Veterinary Journal 15.2 (2025), 923-930. Print. doi:10.5455/OVJ.2025.v15.i2.41 APA (American Psychological Association) Style Budiastuti, B., Thesia, . N. L. N., Plumeriastuti, . H., Effendi, . M. H., Ugbo, . E. N., Khairullah, . A. R., Moses, . I. B., Riwu, . K. H. P., Ahmad, . R. Z., Yanestria, . S. M., Wasito, . W., Pratama, . B. P., Awwanah, . M. & Prasetyo, . A. (2025) Effect of cinnamon oil (Cinnamomum burmannii) on the histological kidney of male diabetic rats (Rattus norvegicus). Open Veterinary Journal, 15 (2), 923-930. doi:10.5455/OVJ.2025.v15.i2.41 |