| Research Article | ||
Open Vet. J.. 2025; 15(2): 977-982 Open Veterinary Journal, (2025), Vol. 15(2): 977-982 Research Article Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbitsAmer Al Ali1, Sara M. Salama2, Wageh Sobhy Darwish3* and Aaser M. Abdelazim11Department of Medical Laboratory Sciences, College of Applied Medical Sciences, University of Bisha, Bisha 67714, Saudi Arabia 2Department of Animal Production, Salhia Investment Development Company, Salhia, Egypt 3Food Hygiene, Safety and Technology Department, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt *Corresponding Author: Wageh Sobhy Darwish. Food Hygiene, Safety and Technology Department, Faculty of Veterinary Medicine, Zagazig University, Zagazig, Egypt. Email: wagehdarwish [at] gmail.com Submitted: 30/12/2024 Accepted: 07/02/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
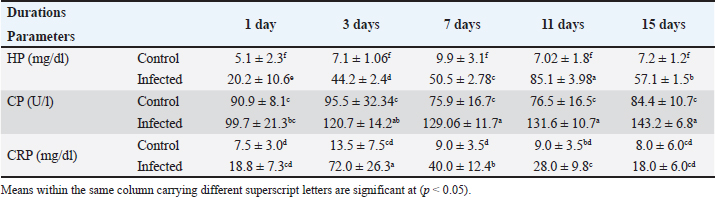
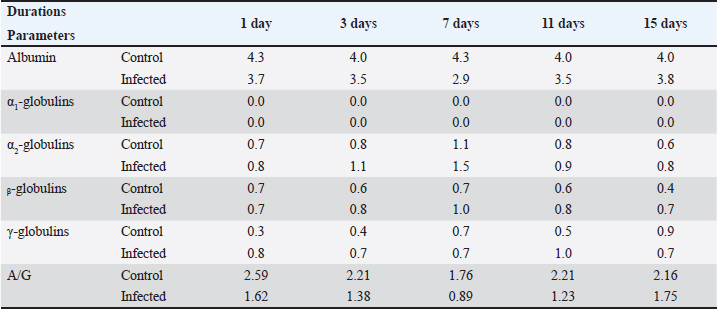
AbstractBackground: Recently, an increase in the synthesis of acute phase proteins (APPs) was associated with acute infections and inflammation in rabbits. Aim: The purpose of this study was to identify biochemical and molecular markers in rabbits that could be used for the early diagnosis of diarrhea. Method: Fifty male White New Zealand rabbits with an average weight of 450–650 g and an age of 6–8 weeks were chosen and then divided into two groups: group A, which served as a control and was not exposed to any infections, and group B, which was infected with Escherichia coli strain O26 through a single oral injection. After 1, 3, 7, 11, and 15 days, four rabbits from the control group and six rabbits from the infected group were slaughtered at various ages. For the purpose of determining the levels of hemoglobin (HP), C-reactive protein (CRP), and ceruloplasmin (CP) activity, as well as the electrophoresis patterns of proteins, the serum samples were taken. For the purpose of determining the level of mRNA expression of HP, livers were used. Results: The results revealed a significant increase in HP, CRP concentration, and CP activity in all infected rabbits. Electrophoresis patterns indicated a decline in serum albumin levels and an increase in α2, β, and γ-globulins levels post-infection. The mRNA expression of HP was significantly increased in infected rabbits. Conclusion: APP patterns and concentrations could be used as potent markers for the early detection of enteric disorders and diarrhea induced by E. coli infection in weaning rabbits. Keywords: Ceruloplasmin, C-reactive protein, Diarrhea, E. coli, Haptoglobin, Rabbits. IntroductionIt was approved that Escherichia coli could induce an outbreak of severe diarrhea and death in young rabbits many years ago (Prescott, 1978). In general, enteric diseases resulting from E. coli infection are considered the main causes of watery diarrhea and high mortality rates in rabbits (Penteado et al., 2002). Until now, there has been no perfect approach for the early detection of diarrhea in weaning rabbits. To a large extent, the diagnosis of enteric disorders in rabbits for a large extent depends on the traditional methods, such as history and signs (Marlier et al., 2006). The digestive tract is responsible for a variety of functions, including the absorption of nutrients and defense of the host. Due to the fact that bacteria are able to traverse the gut barrier and enter the epithelial surface. When it comes into contact with epithelial-derived antimicrobial peptides, cytokines, and acute phase proteins (APPs) (Clemente et al., 2003; Ozmen et al., 2009), it will interact with these substances. An increase in the production of APPs has been linked to acute infections and inflammations in rabbits, according to research published not too long ago (Baker and Long, 1990; Sheriff et al., 2021). According to Matson et al. (2012), haptoglobin (HP), which is a member of the APPs, is found in the plasma of all animals and is enhanced in response to inflammatory situations. Ceruloplasmin (CP) is an α2-glycoprotein that is considered to be one of the major positive APPs. It plays a significant role as a ferroxidase, possessing a powerful antioxidant activity that safeguards host tissues against the release of toxic oxygen metabolites from phagocytic cells in an inflammatory state is present (Bakhautdin et al., 2014). A great consensus has been reached that both CP and HP can be used to monitor infections and inflammation in rabbits (Freitas et al., 2011). On the other hand, high levels of serum C-reactive protein (CRP) correlate with inflammatory conditions in mammals (Sheriff et al., 2021). It is elevated in many microbial infections-induced inflammatory conditions (Nathan and Scheld, 2002; Cazanga et al., 2024). In the present study, we aimed to evaluate the use of APPs as a potent early marker for detecting diarrhea induced by E. coli infection in rabbits. Materials and MethodsProtocolThe following study used 50 male New Zealand rabbits (Oryctolagus cuniculus) with an average weight of 450–650 g and an age of 6–8 weeks at the beginning of the experiment. The animals were divided into two main groups: group A, which served as a control (N=20), did not experience any infection, and were allowed to have unrestricted access to food and water; and group B, which was infected (N=30) and received a single oral dose of E. coli infection. After 1, 3, 7, 11, and 15 days of infection, four rabbits from the control group and six rabbits from the infected group were slaughtered. One week prior to the start of the experiment, the rabbits were maintained under observation for the duration of the experiment. All of the rabbits in the group were provided with the standard rabbit diet and were allowed unrestricted access to clean water. After all, the diet did not include any antimicrobial ingredients. A thorough examination of the fecal samples collected from each rabbit was performed prior to the commencement of the experiment to verify the absence of coccidia (Eimeria oocysts) and E. coli infection. Induction of diarrhea Resistant E. coli O26 strain was prepared using a protocol designed according to Peeters et al., 1988. The inocula for challenge were prepared for 24-hour nutrient broth cultures of the E. coli strain. The challenge dose was adjusted to be 2 × 107 CFU of E. coli. The challenge dose of E. coli was orally administered once to all rabbits in the infected group. Biochemical and molecular markers were determined in all groups at 1st, 3rd, 7th, 11th, and 15th days of infection. Sampling After 1, 7, 11, and 15 days from the commencement of the experiment, blood samples were collected from each animal. The serum was separated in centrifuge tubes that were clean, dry, sterilized, and labeled. None of the samples were immediately inspected; instead, they were held at a temperature of 20°C until biochemical examinations were performed. For the purpose of RNA extraction, liver tissue samples were collected from all animals immediately after slaughter. Until further research was conducted, tissue samples were immediately frozen in liquid nitrogen (Thielen et al., 2007). Biochemical analysisHP concentration was determined according to a method prescribed by Dati et al., 1996, CP activity and CRP concentration were determined according to a manual laboratory Kit manufactured by Invitrogen™, and protein electrophoresis was performed by (Helena, Laboratories, Beaumont, TX). Molecular analysisUsing an RNA extraction Kit (Fermentas, Life Science, USA), total RNA was isolated from liver tissues in accordance with the methodology outlined by the manufacturer, which was described by Boom et al. (1990). The synthesis of first-strand cDNA was performed with the help of the RevertAidTM H Minus First-strand cDNA synthesis kit (Fermentas, Life Science, USA) in accordance with the findings of Wiame et al. (2000). Using a thermal cycler (Biosystems, USA), the amplification was carried out under the following conditions: 25°C for 10 minutes (denaturation), 42°C for 90 minutes (annealing), 95°C for 3 minutes (extension), and then refrigerated in ice for 5 minutes. Reverse transcription-polymerase chain reaction (RT-PCR) was performed with the help of a set of HP-specific oligonucleotide primers, and the housekeeping gene was glyceraldehyde 3-phosphate dehydrogenase (GAPDH). According to Herrler et al. (2004), the primer pairs constructed for amplification were as follows: for high-performance (HP) primers, the forward primer was 5’-TGTGAAGCAGTATGTGGAAAGC-3’, and the reverse primer was 3’-TATAATCCTTTGAAGGTAGGCA-5’. The forward position of GAPDH is 5-CCCGTAGACAAAATGGTGAAGGTC-3, and the reverse position is 5-GCCAAAGTTGTCATGGATGACC-3. The product sizes at these two positions are 386 and 215, respectively. The PCR reaction was performed using the following cycling conditions: initial denaturation at 94°C for 3 minutes, followed by 35 cycles of denaturation at 94°C for 30 seconds, annealing at 58°C for 30 seconds, extension at 72°C for 45 seconds, and final extension at 72°C for 7 minutes. Subsequently, the amplified PCR products were electrophoresed on a 1.5% Agarose gel in a running buffer consisting of 1X Tris-acetate-EDTA (1 × TAE) at 100 V for 40 minutes. Through the use of the UV transilluminator T2621BS (BioRad, USA), bands were observed via ethidium bromide staining. Statistical analysisThe statistical package for social science (SPSS Inc., Chicago, Illinois, version 13) was used to process the data. The mean plus or minus the SD is used to express all results. Student’s t-test (unpaired) and one-way analysis of variance were used to compare the groups. Duncan’s test was used to examine homogeneity between the groups. Our statistical significance was determined to be p < 0.05. ResultsThe concentrations of HP (mg/dl), CRP (mg/dl), and CP activity (U/l) were determined in all rabbits. The data are summarized in (Table 1). Serum HP level (mg/dl)HP levels were elevated in the serum of the infected group at all experimental durations compared with the control group at the same durations. Serum CP activity (U/l)The activity of CP was gradually increased in the serum of the infected group from the 1st day and reached its maximum level at the 15th day post-infection when compared with the control group for the same duration (Table 1). CRP concentration (mg/dl)The level of CRP was increased in the infected group, reaching its maximum level at 3rd day post-infection, while it tended to decline again at 7th day, and the level at the 15th day nearly equals the level at 1st day of infection. Serum protein electrophoresis patternsThe serum protein electrophoresis patterns are summarized in Table 2. Albumin levels were lower in infected rabbits than in their controls at all duration of the experiment. Although α-2, ᵦ, and γ-globulins fractions were increased in the infected groups, α-1 fraction did not be detected at all. Table 1. Effects of E. coli infection on HP, CRP, and CP levels in rabbit's serum.
Table 2. Effect of E. coli infection on the protein electrophoresis fractions (g/dl) in rabbit's serum.
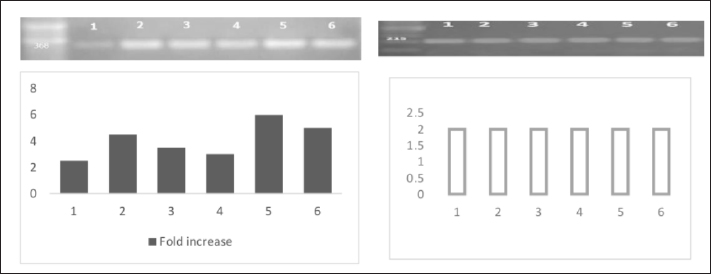
Fig. 1. HP and GPDH gene mRNA expression in liver tissues of infected rabbits, and gel picture analysis using ImageJ software to detect fold increases in expression levels between different groups. 1; Control, 2; 1st day post-infection, 3; 3rd day post-infection, 4; 7th day post-infection, 5; 11th day post-infection, and 6; 15th day post-infection. Expression level of mRNA HP geneThe level of mRNA expression of the HP gene was significantly high in the infected group livers than its expression level in the control group all over the durations of the experiment (Fig. 1). DiscussionIn the present study, we evaluated the use of APPs as markers for the early detection of diarrhea in weaning rabbits. Usually, the clinical signs of diarrhea appeared 4–9 days after bacterial infection (Swennes et al., 2012), and these showed profuse watery diarrhea with mucoid or bloody contents, accompanied by dehydration, anorexia, and lethargy, and usually these rabbits died after 1 week (Heczko et al., 2000). In our study, the clinical signs of diarrhea first appeared in two rabbits on the 5th day post-E. coli infection. On the 13th day of infection, diarrhea was observed in all rabbits. According to Ceron and Martinez-Subiela (2004), measures of APPs have been used extensively for monitoring the evolution of infectious diseases, as well as for diagnosing and evaluating infectious diseases. In the current investigation, weaning rabbits were chosen because this is the age at which rabbits experience the majority of issues and losses associated with this age group (El-Ashram et al., 2020; Wang et al., 2020). Our results revealed that the infection of rabbits by E. coli O26 staining at a dose of (6 × 107cfu) increased the levels of HP and CRP in the serum of infected rabbits at different periods as well as CP activity compared with their control in the same periods. The high level of measured APPs in infected animals was due to the inflammatory condition induced by E. coli, which was approved by Petersen et al., 2004. Usually, APPs could be increased 1,000-fold in response to injuries, inflammatory conditions, and infections (Baker and Long, 1990; Costa et al., 2013). HP and CRP levels were significantly increased, while albumin was decreased in serum during an acute phase response in the same observations of Lu et al., 2019; Sheriff et al., 2021. In the same line, the electrophoresis patterns indicated high globulin levels. In general, CRP responses are generally stronger than those of HP, while elevated HP remained for 2 weeks. These observations were in accordance with the results obtained by Gabay and Kushner, 1999. Although the records of previous studies showed high levels of APPs 24–48 hours, after the initiation of infection (Ballou and Kushner, 1992), our results showed elevated levels of serum HP, CRP, and CP from the 1st day of infection, which means that they started to be elevated even before the appearance of clinical signs which enforced its use as potent markers for early detection of diarrhea in weaning rabbits. In contrast to 15099494, Our results revealed a dramatic increase in serum HP levels from day 3 to 7 and then began to decrease in the 11th day post-E. coli inoculation in rabbits. On the other hand, CP levels showed a less dramatic pattern than that of HP, indicating that CP would be considered a sensitive transient marker of acute inflammation in rabbits (Cray, 2012). Our results showed high significant mRNA expression of HP in the liver tissue of infected rabbits compared with the control group. The expression level started to be elevated from the 1st day post-infection and remained markedly elevated during the entire experimental period within maximal values detected on the 11th and 15th days post-infection. The results were in accordance with Rahman et al. 2010; Quinton et al. 2009. The electrophoresis of serum proteins was approved as an effective tool for disease screening in clinical chemistry many years ago (Bossuyt et al., 1998). It has shown that the level of serum albumin is considered a negative APP because hypoalbuminemia is an actual marker for inflammation and inversely correlates with serum CRP levels (Gabay and Kushner, 1999; Amaro et al., 2021). In the same direction, Ruot et al., 2000 found that plasma albumin levels decreased by 34% in infections. The specific profile of the globulins is determined by protein electrophoresis; three major groups have been identified: gamma globulins, beta globulins, and alpha-2 globulins, whereas alpha-1 globulins were not detected in our electrophoretic pattern. Our results reveal that there are increase of α2-globulins after infection in rabbits compared with control rabbits. We can reveal the highest level of α2-globulins in infected groups to the high levels of HP and CP as they are major α2-globulin fractions. The increase in α-globulins in the infected groups may be a consequence of the increase in the APPs (Victoria Carapeto et al., 2006). In the same line of the present results, previous observations approved an increase in α2-globulins levels in acute and inflammatory conditions (Gori et al., 2022; Bellone et al., 2023). β-globulins levels were increased at the 3rd, 7th, 11th, and 15th post-infection by 1.3, 1.4, 1.3, and 1.7 folds, respectively, while its concentration on the 1st day remained as a control group. γ-globulins levels were increased at the 1st, 3rd, and 11th days post-infection by 2.6, 1.75, and 2 folds, while its concentration remained as the control group at the 7th day, then decreased at 15th day post-infection. We conclude that the drop in γ-globulins levels at the end of the experiment was due to the reduction in the number of T and B cells involved in the cellular and humeral immune response; the same concept is approved by Rodriguez-De Lara et al., 2008. ConclusionThe APP patterns and concentrations can be used as potent markers for the early detection of enteric disorders and diarrhea induced by E. coli infection in weaning rabbits. AcknowledgmentsThe authors are thankful to the Deanship of Graduate Studies and Scientific Research at the University of Bisha for supporting this work through the Fast-Track Research Support Program. Conflict of interestThe authors have no conflict of interest to declare. FundingThe deanship of Graduate Studies and Scientific Research at the University of Bisha supported this work through the Fast-Track Research Support Program. Authors’ contributionsAll authors contributed equally. Data availabilityData will be available upon reasonable request. ReferencesAmaro, E., Moore-Lotridge, S.N., Wessinger, B., Benvenuti, M.A., An, T.J., Oelsner, W.K., Polkowski, G.G. and Schoenecker, J.G. 2021. Albumin and the fibrinogen-to-albumin ratio: biomarkers for the acute phase response following total knee arthroplasty. PLoS One 16, e0247070. Baker, R.D. and Long, S. 1990. Acute phase proteins in neonatal rabbits: diminished C-reactive protein response. J. Pediatr. Gastroenterol. Nutr. 11, 534–541. Bakhautdin, B., Goksoy Bakhautdin, E. and Fox, P.L. 2014. Ceruloplasmin has two nearly identical sites that bind myeloperoxidase. Biochem. Biophys. Res. Commun. 453, 722–727. Ballou, S.P. and Kushner, I. 1992. C-reactive protein and the acute phase response. Adv. Intern. Med. 37, 313–336. Bellone, F., Morace, C., Impala, G., Viola, A., Lo Gullo, A., Cinquegrani, M., Fries, W., Sardella, A., Scolaro, M., Basile, G., Squadrito, G. and Mandraffino, G. 2023. Quality of life (QoL) in patients with chronic inflammatory bowel diseases: how much better with biological drugs? J. Pers. Med. 13, 947. Boom, R., Sol, C.J., Salimans, M.M., Jansen, C.L., Wertheim-Van Dillen, P.M. and Van Der Noordaa, J. 1990. Rapid and simple method for purification of nucleic acids. J. Clin. Microbiol. 28, 495–503. Bossuyt, X., Schiettekatte, G., Bogaerts, A. and Blanckaert, N. 1998. Serum protein electrophoresis by CZE 2000 clinical capillary electrophoresis system. Clin. Chem. 44, 749–759. Cazanga, V., Riquelme, J., Cornejo, D., Jeldres, J.A., Palma, C. and Perez-Fernandez, R. 2024. Influence of Escherichia coli lipopolysaccharide-induced endotoxemia on plasma and tissue disposition of florfenicol after intramuscular administration in rabbits. Res. Vet. Sci. 170, 105187. Ceron, J.J. and Martinez-Subiela, S. 2004. An automated spectrophotometric method for measuring canine ceruloplasmin in serum. Vet. Res. 35, 671–679. Clemente, M.G., De Virgiliis, S., Kang, J.S., Macatagney, R., Musu, M.P., Di Pierro, M.R., Drago, S., Congia, M. and Fasano, A. 2003. Early effects of gliadin on enterocyte intracellular signalling involved in intestinal barrier function. Gut, 52, 218–223. Costa, M.M., Dos Anjos Lopes, S.T., Franca, R.T., Da Silva, A.S., Paim, F.C., Palma, H.E., Maciel, R.M., Dornelles, G.L., De Azevedo, M.I., Tonin, A.A., Santurio, J.M., Duarte, M.M. and Monteiro, S.G. 2013. Role of acute phase proteins in the immune response of rabbits infected with Trypanosoma evansi. Res. Vet. Sci. 95, 182–188. Cray, C. 2012. Acute phase proteins in animals. Prog. Mol. Biol. Transl. Sci. 105, 113–150. Dati, F., Schumann, G., Thomas, L., Aguzzi, F., Baudner, S., Bienvenu, J., Blaabjerg, O., Blirup-Jensen, S., Carlstrom, A., Petersen, P.H., Johnson, A.M., Milford-Ward, A., Ritchie, R.F., Svendsen, P.J. and Whicher, J. 1996. Consensus of a group of professional societies and diagnostic companies on guidelines for interim reference ranges for 14 proteins in serum based on the standardization against the IFCC/BCR/CAP Reference Material (CRM 470). International Federation of Clinical Chemistry. Community Bureau of Reference of the Commission of the European Communities. College of American Pathologists. Eur. J. Clin. Chem. Clin. Biochem. 34, 517–520. El-Ashram, S., Aboelhadid, S.M., Abdel-Kafy, E.M., Hashem, S.A., Mahrous, L.N., Farghly, E.M. and Kamel, A.A. 2020. Investigation of pre- and post-weaning mortalities in rabbits bred in Egypt, with reference to parasitic and bacterial causes. Animals (Basel), 10, 537. Freitas, F.L., Yamamoto, B.L., Freitas, W.L., Fagliari, J.J., Almeida Kde, S., Machado, R.Z. and Machado, C.R. 2011. Systemic inflammatory response indicators in rabbits (Oryctolagus cuniculus) experimentally infected with sporulated oocysts of Eimeria stiedai (Apicomplexa: Eimeriidae). Rev. Bras. Parasitol. Vet. 20, 121–126. Gabay, C. and Kushner, I. 1999. Acute-phase proteins and other systemic responses to inflammation. N. Engl. J. Med. 340, 448–454. Gori, E., Pierini, A., Tulone, F., Abramo, F. and Marchetti, V. 2022. Serum protein electrophoresis in 26 dogs with chronic hepatitis. J. Vet. Diagn. Invest. 34, 738–741. Heczko, U., Abe, A. and Finlay, B.B. 2000. In vivo interactions of rabbit enteropathogenic Escherichia coli O103 with its host: an electron microscopic and histopathologic study. Microbes Infect. 2, 5–16. Herrler, A., Krusche, C.A., Muller-Schottle, F. and Beier, H.M. 2004. Haptoglobin expression and release by rabbit oviduct and endometrium, its localization in blastocyst extra-embryonic matrix and fluid during preimplantation time. Hum. Reprod. 19, 2730–2737. Lu, G., Wang, C., Wu, C., Yan, L. and Tang, J. 2019. Identification of early biomarkers in a rabbit model of primary Candida pneumonia. BMC Infect. Dis. 19, 698. Marlier, D., Dewree, R., Lassence, C., Licois, D., Mainil, J., Coudert, P., Meulemans, L., Ducatelle, R. and Vindevogel, H. 2006. Infectious agents associated with epizootic rabbit enteropathy: isolation and attempts to reproduce the syndrome. Vet. J. 172, 493–500. Matson, K.D., Horrocks, N.P., Versteegh, M.A. and Tieleman, B.I. 2012. Baseline haptoglobin concentrations are repeatable and predictive of certain aspects of a subsequent experimentally-induced inflammatory response. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 162, 7–15. Nathan, B.R. and Scheld, W.M. 2002. The potential roles of C-reactive protein and procalcitonin concentrations in the serum and cerebrospinal fluid in the diagnosis of bacterial meningitis. Curr. Clin. Top. Infect. Dis. 22, 155–165. Ozmen, M.M., Zulfikaroglu, B., Col, C., Cinel, I., Isman, F.K., Cinel, L. and Besler, T.H. 2009. Effect of increased abdominal pressure on cytokines (IL1 beta, IL6, TNFalpha), C-reactive protein (CRP), free radicals (NO, MDA), and histology. Surg. Laparosc. Endosc. Percutan. Tech. 19, 142–147. Peeters, J.E., Geeroms, R. and Orskov, F. 1988. Biotype, serotype, and pathogenicity of attaching and effacing enteropathogenic Escherichia coli strains isolated from diarrheic commercial rabbits. Infect. Immun. 56, 1442–1448. Penteado, A.S., Ugrinovich, L.A., Blanco, J., Blanco, M., Blanco, J.E., Mora, A., Andrade, J.R., Correa, S.S. and Pestana De Castro, A.F. 2002. Serobiotypes and virulence genes of Escherichia coli strains isolated from diarrheic and healthy rabbits in Brazil. Vet. Microbiol. 89, 41–51. Petersen, H.H., Nielsen, J.P. and Heegaard, P.M. 2004. Application of acute phase protein measurements in veterinary clinical chemistry. Vet. Res. 35, 163–187. Prescott, J.F. 1978. Escherichia coli and diarrhoea in the rabbit. Vet. Pathol. 15, 237–248. Quinton, L.J., Jones, M.R., Robson, B.E. and Mizgerd, J.P. 2009. Mechanisms of the hepatic acute-phase response during bacterial pneumonia. Infect. Immun. 77, 2417–2426. Rahman, M.M., Lecchi, C., Avallone, G., Roccabianca, P., Sartorelli, P. and Ceciliani, F. 2010. Lipopolysaccharide-binding protein: local expression in bovine extrahepatic tissues. Vet. Immunol. Immunopathol. 137, 28–35. Rodriguez-De Lara, R., Cedillo-Pelaez, C., Constantino-Casas, F., Fallas-Lopez, M., Cobos-Peralta, M.A., Gutierrez-Olvera, C., Juarez-Acevedo, M. and Miranda-Romero, L.A. 2008. Studies on the evolution, pathology, and immunity of commercial fattening rabbits affected with epizootic outbreaks of diarrhoeas in Mexico: a case report. Res. Vet. Sci. 84, 257–268. Ruot, B., Breuille, D., Rambourdin, F., Bayle, G., Capitan, P. and Obled, C. 2000. Synthesis rate of plasma albumin is a good indicator of liver albumin synthesis in sepsis. Am. J. Physiol. Endocrinol. Metab. 279, E244–E251. Sheriff, A., Kayser, S., Brunner, P. and Vogt, B. 2021. C-reactive protein triggers cell death in ischemic cells. Front. Immunol. 12, 630430. Swennes, A.G., Buckley, E.M., Parry, N.M., Madden, C.M., Garcia, A., Morgan, P.B., Astrofsky, K.M. and Fox, J.G. 2012. Enzootic enteropathogenic Escherichia coli infection in laboratory rabbits. J. Clin. Microbiol. 50, 2353–2358. Thielen, M.A., Mielenz, M., Hiss, S., Zerbe, H., Petzl, W., Schuberth, H.J., Seyfert, H.M. and Sauerwein, H. 2007. Cellular localization of haptoglobin mRNA in the experimentally infected bovine mammary gland. J. Dairy Sci. 90, 1215–1219. Victoria Carapeto, M., Barrera, R., Cinta Mañe, M. and Zaragoza, C. 2006. Serum α-globulin fraction in horses is related to changes in the acute phase proteins. J. Equine Vet. Sci. 26, 120–127. Wang, J., Ni, X., Wen, B., Zhou, Y., Liu, L., Zeng, Y., Zhao, W., Khalique, A., Wang, P., Pan, K., Yu, Z., Jing, B., Liu, H. and Zeng, D. 2020. Bacillus strains improve growth performance via enhancing digestive function and anti-disease ability in young and weaning rex rabbits. Appl. Microbiol. Biotechnol. 104, 4493–4504. Wiame, I., Remy, S., Swennen, R. & Sagi, L. 2000. Irreversible heat inactivation of DNase I without RNA degradation. Biotechniques, 29, 252–256. | ||
| How to Cite this Article |
| Pubmed Style Ali AA, Salama SM, Darwish WS, Abdelazim AM. Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbits. Open Vet. J.. 2025; 15(2): 977-982. doi:10.5455/OVJ.2025.v15.i2.46 Web Style Ali AA, Salama SM, Darwish WS, Abdelazim AM. Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbits. https://www.openveterinaryjournal.com/?mno=240530 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.46 AMA (American Medical Association) Style Ali AA, Salama SM, Darwish WS, Abdelazim AM. Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbits. Open Vet. J.. 2025; 15(2): 977-982. doi:10.5455/OVJ.2025.v15.i2.46 Vancouver/ICMJE Style Ali AA, Salama SM, Darwish WS, Abdelazim AM. Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbits. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 977-982. doi:10.5455/OVJ.2025.v15.i2.46 Harvard Style Ali, A. A., Salama, . S. M., Darwish, . W. S. & Abdelazim, . A. M. (2025) Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbits. Open Vet. J., 15 (2), 977-982. doi:10.5455/OVJ.2025.v15.i2.46 Turabian Style Ali, Amer Al, Sara M. Salama, Wageh Sobhy Darwish, and Aaser M. Abdelazim. 2025. Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbits. Open Veterinary Journal, 15 (2), 977-982. doi:10.5455/OVJ.2025.v15.i2.46 Chicago Style Ali, Amer Al, Sara M. Salama, Wageh Sobhy Darwish, and Aaser M. Abdelazim. "Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbits." Open Veterinary Journal 15 (2025), 977-982. doi:10.5455/OVJ.2025.v15.i2.46 MLA (The Modern Language Association) Style Ali, Amer Al, Sara M. Salama, Wageh Sobhy Darwish, and Aaser M. Abdelazim. "Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbits." Open Veterinary Journal 15.2 (2025), 977-982. Print. doi:10.5455/OVJ.2025.v15.i2.46 APA (American Psychological Association) Style Ali, A. A., Salama, . S. M., Darwish, . W. S. & Abdelazim, . A. M. (2025) Acute phase proteins as potent early biomarkers for the detection of Escherichia coli infection in white New Zealand rabbits. Open Veterinary Journal, 15 (2), 977-982. doi:10.5455/OVJ.2025.v15.i2.46 |