| Research Article | ||
Open Vet. J.. 2025; 15(2): 994-1008 Open Veterinary Journal, (2025), Vol. 15(2): 994-1008 Research Article Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camelsAhmed Al-Montaser El-Sayed1, Ahmed Adel El-Sayed2*, Mohamed E. Ali1, Attia Eissa3, Ahmed Ragab Askar4 and Sabry Mousa51Department of Internal Medicine and Infectious Diseases, Faculty of Veterinary Medicine, Cairo University, Cairo, Egypt 2Department of Animal Health and Poultry, Animal and Poultry Production Division, Desert Research Center (DRC), Cairo, Egypt 3Department of Animal Medicine (Internal Medicine), Faculty of Veterinary Medicine, Arish University, Arish, Egypt 4Department of Animal and Poultry Nutrition, Animal and Poultry Production Division, Desert Research Center, Cairo, Egypt 5Departments of Veterinary Clinical Sciences, Faculty of Veterinary Medicine, Jordan University of Science and Technology, Irbid, Jordan *Corresponding Author: Ahmed Adel El-Sayed, Department of Animal Health and Poultry, Animal and Poultry Production Division, Desert Research Center (DRC), Cairo, Egypt. Email: decernes [at] yahoo.com Submitted: 31/12/2024 Accepted: 07/02/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
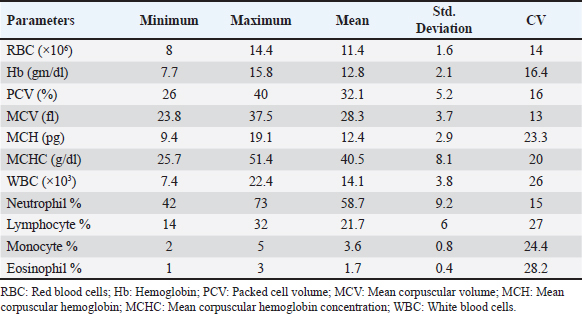
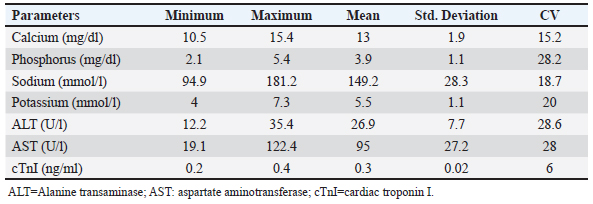
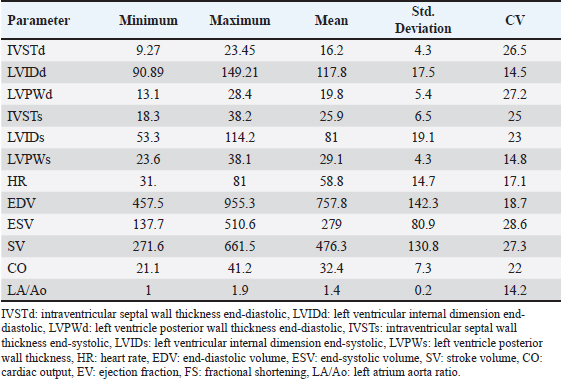
AbstractBackground: Pulsed-wave Doppler echocardiography (PWDE) is a practical, noninvasive imaging assessment method that has been used to reveal details about the characteristics, direction, timing, and velocity of blood flow of the heart under both healthy and diseased conditions. Aim: The purpose of this study was to investigate normal heart imaging and chamber measurements, hematobiochemical profile, and PWDE in racing camels. Methods: To accomplish this, blood samples, PWDE, and echocardiograms were obtained from 20 healthy racing camels aged 14–20 years and weighing 500–650 kg. Results: Red blood cell (RBCs), hemoglobin (Hb), packed cell volume (PCV), mean corpuscular volume (MCV), and white blood cell (WBCs) were recorded along with their mean values (11.4 (×106), 12.8 (gm/dl), 32.1%, 28.3 (fl), 14.1 (×103), respectively. The mean values of calcium (13 mg/dl), phosphorus (3.9 mg/dl), sodium (194.2 mmol/l), potassium (5.5 mmol/l), alanine transaminase (ALT) (26.6 U/l), aspartate aminotransferase (AST) (95 U/l), and cardiac troponin I (cTnI) (0.3 ng/ml). The interventricular septum, atria, and ventricles were viewed in a caudal long-axis four-chamber view in the 4th or 5th intercostal space (ICS) on both sides. The right ventricle, left ventricle, and interventricular septum were evident in the short-axis image of the cardiac ventricles acquired in the fourth ICS. The M mode was used to obtain cardiac measurements from every camel. Using the PWD at the left parasternal longitudinal axis, the mitral valve (Mv), tricuspid valve (Tv), and maximum aortic valve velocity were determined in all camels. The right panel is the pulmonary valve. Conclusion: Our findings showed that adult racing camels have normal heart dimensions and can acquire high-quality echocardiograms and PWDE. This study can serve as a reference for future research on racing camels with heart diseases. Keywords: Racing camels, Hematobiochemical variables, Echocardiography, Pulsed-wave Doppler echocardiography. IntroductionThe dromedary one-humped camel (Camelus dromedarius) is an important mammal that provides a variety of services, including labor, transportation, recreation, food (meat, milk), and race (Suliman et al., 2019; Swelum et al., 2020). Because of their variable core temperatures and ability to survive for long periods of time without water, these animals thrive in the dry, semi-arid, and tropical climates found in Asia, Africa, and Oceania (Faraz et al., 2019; Tibary and El Allali, 2020). A long-standing, conventional sport, camel racing is mostly practiced in Arab nations. In the past few decades, camel racing has gained more attention. In the United Arab Emirates and the nations surrounding the Arabian Gulf, camel racing has become more closely resembled thoroughbred horse racing. Depending on the camels’ age, breed, and sex, as well as the organizing committees’ decision-making, an official race’s length normally varies from 4 to 20 miles. A camel can move at about 9.5 minute/second on average during racing (Snow, 1992). Most camels gallop at the start of the race, and during the race, they alternate between pacing and galloping regularly. Remarkably, camels can pace at about the same speed as sprinters. When camels engage in vigorous exercise, their heart rates rise less than those of horses. The mean peak heart rate in horses is 7–8 times that of resting levels, but it is about four times that in camels (Evans et al., 1992). The training (Evans et al., 1992; Snow, 1992) and consumption (Holbrook et al., 2006) of camels have been the subjects of numerous research studies. Research programs similar to those used to enhance human and equine athletes have been prompted by the need to breed and train faster camels. Although camel physiological adaptations have been thoroughly examined, until recently, modifications linked to exercise were disregarded (Faraz et al., 2019). According to Abdel-Ghaffar et al. 2009; Fowler, 2011, Sarcocystosis, aortic and pulmonary transposition, persistent right aortic arch, persistent aortic trunk, septal abnormalities, and patent ductus arteriosus are congenital disorders that might affect camels. The majority of the heart conditions mentioned above are either unintentionally found during postmortem examinations or diagnosed at slaughterhouses (Fowler, 2011), demonstrating how difficult it may be to diagnose camel heart disease, particularly in cases where the patient does not exhibit the usual clinical symptoms of heart failure. These factors make auxiliary testing necessary for the evaluation of living animals. Animals should not start therapy too soon, and low-value patients should not make further therapeutic investments when the prognosis is poor. An excellent supplementary method for evaluating the heart of different ruminant species is echocardiography. It is an easy-to-use, noninvasive technique for evaluating the bovine heart (Hallowell et al., 2007). Due to the improved assessment of the disease scope and significance, this tool has been widely employed in cattle and may be used as a prognostic tool in certain cases (Buczinski, 2009; Mohamed and Buczinski, 2011). In horses, cattle, and small animals, this imaging method is frequently used to assess morphologic alterations, unusual wall thickness, chamber dimensions, and valvular appearance and purpose (Buczinski, 2009). Accurate and reproducible measurements require pictures captured from imaging planes that are properly oriented (Schiller et al., 1989). The noninvasive imaging diagnostic tool known as Doppler ultrasonography has been applied in the field of veterinary medicine for the purpose of determining the characteristics, direction, timing, and velocity of localized blood flow in both healthy and ill animals (Masoudifard et al., 2020). Researchers can learn much about an animal’s nutritional state, overall health, and overall well-being by studying its hematobiochemical composition (Faraz et al., 2019). Determining the extent of host tissue damage and infection severity may be possible by monitoring specific blood parameters for deviations from their typical boundaries; this could also serve as a tool for differential diagnosis (Van Saun, 2000). Whether exercise affects an animal’s physiological parameters is dependent on strength and duration dependent (Burton et al., 2004). Camel physiological markers and plasma biochemical components can change as a result of this specific activity (Tharwat et al., 2013). For the most part, ultrasound has only been used to monitor the reproductive system of camels. (Skidmore and Billah, 2011). The authors were unable to find any previous research that detailed the echocardiogram or Doppler echocardiography findings of a normal racing camel’s heart, as well as its internal measurements. This research aimed to identify the results of a potential examination in adult racing camels (Camelus dromedarius) by reporting pulsed-wave Doppler echocardiographic findings, regular cardiac look, chamber dimensions, and hematobiochemical profiles. We reasoned that these data could help veterinarians identify cardiac issues in racing camels. Materials and methodsAnimalsTwenty racing camels, all of which appeared to be in excellent condition, were used in the present investigation. Their ages ranged from 14 to 20 years (mean ± SD: 17.1 ± 1.5) and their weights were from 500 to 600 kg (mean ± SD: 567.5 ± 58.17). The South Sinai Research Station, Desert Research Center, Egypt, was the site of the experiment, which ran from April to December 2023. For these animals to be considered, a number of criteria had to be satisfied. These included continuous electrocardiogram recording, normal cardiac auscultation, normal complete blood cell counts, normal biochemistry profiles, and echocardiography (EDAN ACCLARIX AX8, 1-5 MHZ PHASED ARRAY PROBE) (Tharwat et al., 2012). Within an open yard, the camels were kept and given 3 kg of concentrated mixture that included 55% corn, 15% soybean meal, 10% cottonseed meal, 15% wheat bran, 2.5% limestone, 1.5% salt, 0.5% sodium bicarbonate, 0.1% yeast, 0.1% antitoxins, and 0.3% premix. They were also given free access to water and Egyptian clover hay (Trifolium alexandrinum). Permission number Vet CU: 25122023882 was obtained in compliance with all processes, which were conducted in line with the protocols of the Desert Research Center (Egypt) and the (IACUC). Blood samplingThrough jugular vein puncture, 10 ml of blood were drawn from each camel. To obtain whole blood or serum, the samples were collected in vacutainer tubes that contained anticoagulant (EDTA or sodium fluoride) or did not contain anticoagulant. The blood samples in plain tubes were centrifuged at 3,000 rpm for 15 minutes, and the EDTA blood was used for CBC. Additional biochemical, inflammatory, and oxidative stress/antioxidant parameter determinations were performed on the isolated serum, which was then kept at −80°C. Hematological analysisAn automated blood cell counter (Exigo eos veterinary Hematology system, Boule Medical AB, Sweden) was used for the hematological analysis. Biochemical analysesSerum biochemical analysis was performed using an Indian ROBONIC biochemistry analyzer to measure phosphate, calcium, sodium, potassium, alanine transaminase (ALT), and aspartate aminotransferase (AST). Using a FLUOstar Omega enzyme-linked immunosorbent assay (ELISA) reader and a specialized kit, a commercial ELISA test was conducted to measure cTnI. EchocardiographyEach camel’s forelimb was first bent and secured at the carpal joint using a rope. After that, the animal was prodded, and its head held until it was in a sternal recumbency. Next, a rope was wound around the hock and carpal joints of the fore and hind limbs, respectively. Every echocardiography test was performed on the standing and recumbent camels. An ultrasonic device called EDAN ACCLARIX AX8, which uses a 1–5 MHZ PHASED ARRAY PROBE, was used to conduct echocardiographic examinations. The intercostal gaps (third to sixth) on each side of the thorax were cut, trimmed, and cleaned with alcohol to eliminate excess oil prior to applying coupling gel to prepare the echocardiogram. Ultrasound was used to scan the third, fourth, and fifth intercostal gaps in the heart area on both sides of the thorax. To improve the connection between the probe and the intercostal space, the thoracic legs were shifted cranially. Within the heart’s chambers, the primary blood vessels, valves, and the heart itself were visualized. This study adapted the heart images reported for camels and used them in its analysis (Tharwat et al., 2012) and for cattle (Braun et al., 2001; Hallowell et al., 2007; Buczinski, 2009). The same operator performed both M-mode and 2D comprehensive real-time echocardiograms. For more accurate M-mode recordings and easier probe placement, two-dimensional images of the patient’s left and right chest were used. The right side of the image shows the results of a short-axis M-mode echocardiogram that measures various parameters, such as intraventricular septal wall thickness (IVSTd), left ventricular internal dimension end-diastolic (LVIDd), left ventricle posterior wall thickness (LVPWs), intraventricular septal wall thickness (IVSTs) during systole, heart rate (HR), stroke volume (SV), ejection fraction (EV), cardiac output (CO), left ventricular internal dimension end-systolic (LVIDs) during diastole (LVIDs), and fractional shortening (FS). The LA/Ao ratio was determined using the LA and Ao diameters acquired from the right parasternal two-dimensional short-axis images of the heart’s base in the first frame. On the left side, you can see a four-chamber image of the heart, which is called the caudal long-axis view. The right side had a caudal long-axis view of the ventricles, a caudal short-axis view of the left ventricular outflow tract (LVOT), and a caudal long-axis view of the right and left ventricles. On the left side, two views were acquired: one of the LVOT and one of the heart (four-chamber view), both taken along the long caudal axis. Following each inspection, recordings were made that included information about the intercostal space and the angle of the probe used to obtain each image. The two-dimensional and multimode pictures yielded fifteen metrics. The parameters were measured as follows: IVSTd, LVIDd, LVIDs, HR, end-diastolic volume (EDV), end-systolic volume (ESV), SV, CO, EV, the ratio of the left atrium to the aorta, and FS. Doppler echocardiographyUsing a real-time B-mode ultrasound device, Doppler ultrasonography (EDAN ACCLARIX AX8 PHASED ARRAY PROBE 1–5 MHZ) was performed without the use of anesthesia. We looked at camels standing and lying down. Using the transducer—which stands for color flow, pulsed- and continuous-wave Doppler—the tricuspid, aortic, mitral, and pulmonary valve blood flows, as well as turbulent jets, were measured. Flows with positive and negative frequency shifts were detected. Image alignment with intracardiac markers was performed to establish the transducer position. With the goal of minimizing mistakes, every camel went through this procedure twice. The two-dimensional right parasternal short axis and parallel left parasternal longitudinal axis (LPSLAx) five-chamber views at the level of the aortic valve were used for measurement of the pulmonary valve maximum velocity. To assess the aortic valve (Av) Vmax, mitral valve (Mv) E/A, and tricuspid valve (Tv) E/A, pulsed-wave Doppler at LPSLAx was used. Statistical analysisWe used SPSS, version 20 from Inc is IBM SPSS Statistics in Chicago, USA, to conduct our statistical analyses. Every hematobiochemical, echocardiographic, and pulsed-wave Doppler echocardiographic parameter has been statistically described. The standard deviation, coefficient of variance, minimum, and maximum are the ways in which data are presented. ResultsClinical examinationThere was no history or indication of cardiac dysfunction in the camels used in this study. The clinical evaluation of any animal’s cardiothoracic systems revealed no anomalies, and cardiac auscultation revealed no murmurs or dysrhythmia. The results of the clinical evaluation showed that the mean heart rate was 36 ± 8 bpm (reference range: 28–40 bpm), the mean respiratory rate was 10 ± 4 bpm (reference range: 5–12 bpm), and the mean rectal temperature was 36.6°C ± 0.6°C (reference range: 36.0°C–38.0°C. Hematobiochemical profileTable 1 presents a summary of the hematological profile, and Table 2 presents the serum biochemical profile of adult racing camels. Hematological parameters such as Red blood cells (RBCs), hemoglobin (Hb), packed cell volume (PCV), mean corpuscular volume (MCV), MCH, MCHC, white blood cell (WBCs), neutrophils, lymphocytes, and monocytes were recorded along with their mean values ± standard deviation (11.4 ± 1.6 (×106), 12.8 ± 2.1 (gm/dl), 32.1% ± 5.2%, 28.3 ± 3.7 (fl), 12.4 ± 2.9 (pg), 40.5 ± 8.1 (g/dl), 14.1 ± 3.8 (×103), 58.7% ± 9.2%, 21.7% ± 6%, and 3.6% ± 0.8%), respectively (Table 1). The mean values ± standard deviation of the following biochemical parameters were noted: calcium (13 ± 1.9 mg/dl), phosphorus (3.9 ± 1.1 mg/dl), sodium (194.2 ± 28.3 mmol/l), potassium (5.5 ± 1.1 mmol/l), ALT (26.6 ± 7.7 U/l), AST (95 ± 27.2 U/l), and cardiac troponin I (cTnI) (0.3 ± 0.02 ng/ml) (Table 2). Echocardiographic measurementsTable 3 provides an overview of the echocardiographic and pulsed-wave Doppler echocardiographic serial measures in adult racing camels. The mean values ± standard deviation of the following echocardiographic parameters were recorded: HR, EDV, ESV, SV, CO, LVIDd, LVPWd, IVSTs, LVIDs, LVPWs, ESV, ESV, SV, and LA/Ao. Right parasternal ultrasonographyBy positioning the probe perpendicularly in the fourth intercostal space or longitudinally in the fifth space with a little clockwise rotation, the interventricular septum, ventricles, and atria could be observed in the caudal long-axis four-chamber view (Fig. 1). From this vantage point, all camels could be seen to have a left ventricle, mitral valve, interventricular septum, right ventricle, and tricuspid valve. Figure 2 shows that in the short-axis image of the cardiac ventricles obtained with a little clockwise rotation at the 4th ICS, only the right ventricle, interventricular septum, and left ventricle were visible. Here, the dimensions of all camel heart were measured in M mode. The right parasternal two-dimensional short-axis pictures of the heart’s base in the first frame were used to determine the LA/Ao ratio, by applying 2D and M mode (Figs. 3 and 4). Despite some difficulties, we successfully acquired a pulmonary image by attempting to acquire the right ventricular outflow tract for the right short axis at the level of the great vessels. Table 1. Hematological measurements of racing camels (n=20).
Table 2. Biochemical measurements of racing camels (n=20).
Table 3. Echocardiographic measurements of racing camels (n=20).
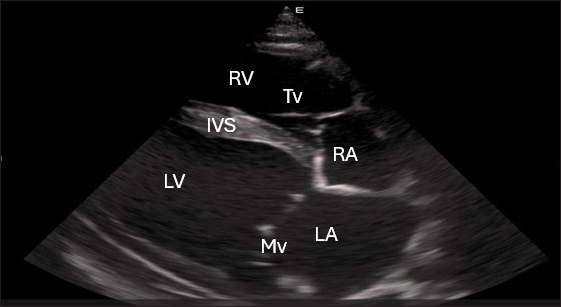
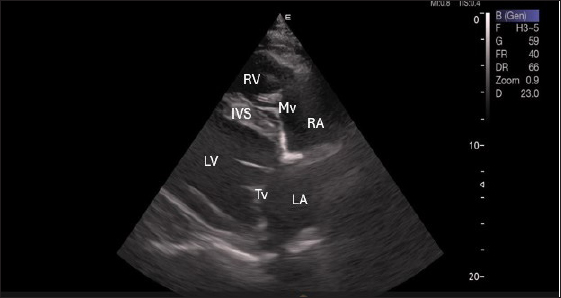
Fig. 1. Right parasternal longitudinal axis (systole) of 16 years racing camel showing Lv=left ventricle, Rv=right ventricle, Ivs=Inte3r ventricular septum, Mv=mitral valve, and Tv=tricuspid valve. Left parasternal ultrasonographyFigure 5 shows an image of the ventricles, atria, and interventricular septum in the caudal long-axis four-chamber view acquired by positioning the probe perpendicularly in the fourth intercostal space or longitudinally in the fifth intercostal space with a little clockwise rotation. Every camel had a perfectly normal heart anatomy, including ventricles, tricuspid valve, mitral valve, and atria on both sides. During the fourth ICS image of the LVOT, the probe was turned slightly further cranially and twisted somewhat counterclockwise (Fig. 6). No camel is complete without an oblique aortic cross-section.
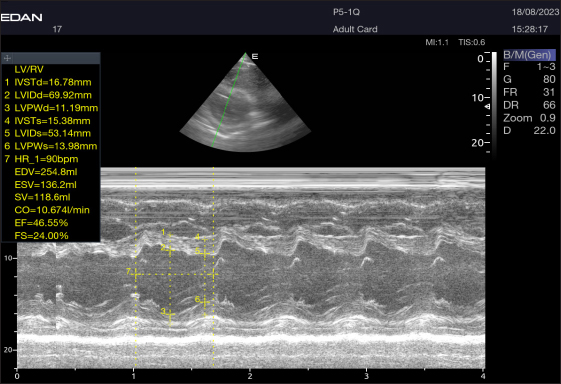
Fig. 2. Short-axis plane M mode (Mushroom shape) of a 16-year-old racing girl camel at level papillary muscle showing left ventricular dimensions (systole and diastole). IVSTd=intraventricular septal wall thickness end-diastolic, LVPWd=left ventricle posterior wall thickness end-diastolic, LVIDd=left ventricular internal dimension, LVIDs=left ventricular internal dimension end-systolic, LVPWs=left ventricle posterior wall thickness, IVSTs=intraventricular septal wall thickness end-systolic, HR=heart rate, EDV=end-diastolic volume, ESV=end-systolic volume, CO=cardiac output, EV=ejection fraction, SV=stroke volume, FS=fractional shortening.
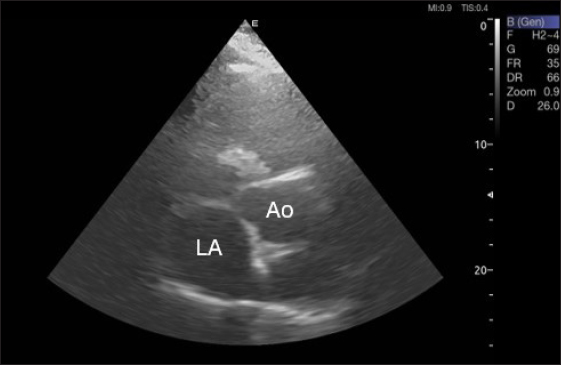
Fig. 3. Right parasternal short-axis view of 16 she-camel at great vessel level showing Ao=aorta and LA=left atrium.
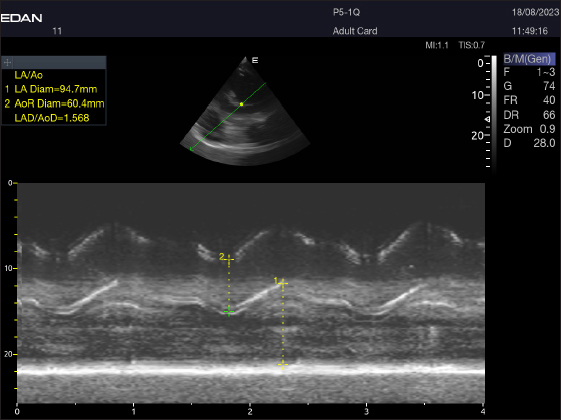
Fig. 4. LA/Ao ratios for the right parasternal short axis of 18 racing camels at the great vessel level. Through the anatomical M mode.
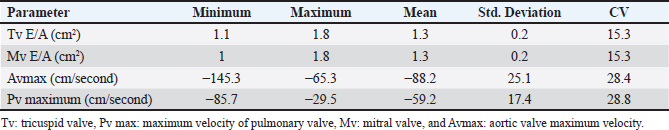
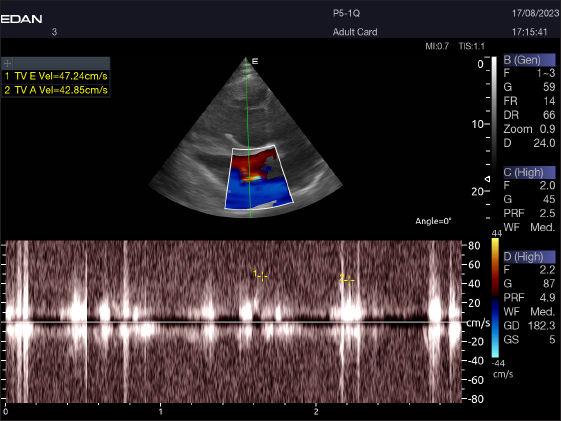
Fig. 5. Left parasternal longitudinal axis (Systole) of 16 years racing camel showing Lv=left ventricle, Rv=right ventricle, Ivs=interventricular septum, Mv=mitral valve, and Tv=tricuspid valve. Doppler echocardiographyTable 4 provides an overview of pulsed-wave Doppler echocardiographic serial measures in adult racing camels. The pulsed-wave Doppler echocardiographic measurements’ mean values ± standard deviation were recorded for Tv E/A (1.3 ± 0.2 cm2), Mv E/A (1.3 ± 0.2 cm2), Av max (−88.2 ± 25.1 cm/second), and Pv max (−59.2467 ± 17.4 cm/second), as shown in Table 4.
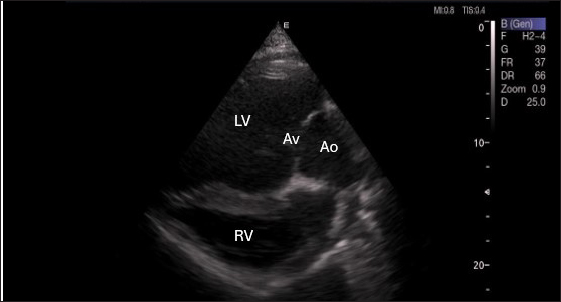
Fig. 6. Left parasternal long axis of 18 racing camel showing LOVT=left ventricular outflow tract. LV=left ventricle, Av=aortic valve, Ao=aortic artery, Rv=right ventricle. Table 4. Pulsed-wave Doppler echocardiographic measurements of racing camels (n=20).
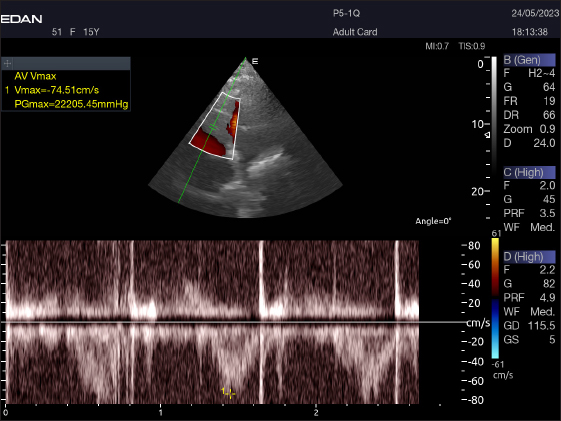
Right parasternal ultrasonographyThere was difficulty in measuring the pulmonary valve from the right short parasternal view in most cases (58.4%) (Fig. 7). Left parasternal ultrasonographyThe Mv E/A (Fig. 8), Tv E/A (Fig. 9), and maximum velocity of the aortic valve (Av Vmax) (Fig. 10) were all measured using pulsed-wave Doppler at LPSLAx in all camels. Two cases were observed with mild mitral regurgitation detected through continuous-wave Doppler, but the regurgitation was not considered significant (Fig. 11). DiscussionThis study is the first to assess echocardiographic and pulsed-wave Doppler echocardiographic measurements in racing camels, for which data from previous reports are still scarce (Hallowell et al., 2007; Tharwat et al., 2012). This study demonstrated that adult racing camels can provide accurate heart pictures, measures of internal cardiac structures, and pulsed-wave Doppler echocardiographic data. Understanding the normal appearance of the heart, cardiac dimensions, and pulsed-wave Doppler echocardiography can help identify, quantify, and evaluate cardiac disease in this population. It may also make it possible to diagnose and treat cardiac disorders earlier in the disease course. The hematological results were consistent with those published by Elgioushy et al. 2020, with the exception of RBCs and WBCs, which were higher than the respective values of 9.82 ± 0.28 (×106) and 13.22 ± 0.52 (×103), while the Hb value and neutrophil % were lower than the 13.47 ± 0.52 (gm/dl) and 70% reported by that author in 10 female racing camels (Camelus dromedarius). Conversely, our findings were not the same as those of Tharwat, 2021, whose values for RBCs, Hb, PCV, MCV, MCH, WBCs, and monocyte % [9.2 ± 1.0 (×106) and 11.1 ± 1.1 (gm/dl), 23.7 ± 4.5 (%), 26.0 ± 1.3 (fl), 11.6 ± 1.7 (pg), 12.9 ± 3.5 (×103) and 2.3%], respectively, were lower than ours. On the other hand, the MCHC value and lymphocyte percentage were lower than the values of 44.5 ± 7.1 (g/dl) and 36.2%, respectively, recorded by that author in 23 healthy racing camels (Camelus dromedarius). Unlike to our results, in a study by Martín-Barrasa et al., 2023, the values for RBCs, PCV, MCV, WBCs, and neutrophils [10.56 ± 1.58 (×106), 26.62 ± 3.70 (%), 25.23 ± 4.12 (fl), 12.21 ± 3.20 (×103) and 52.9%], respectively, were lower than ours. On the other hand, the MCHC value and lymphocyte and monocyte percentage were lower than the values of 48.59 ± 4.84 (g/dl) and 21.2% and 3.6%, respectively, recorded by that author in 114 clinically healthy dromedary camels (Camelus dromedarius). Differences in breed, physical and climatic conditions, analytical methods, and sampling strategies can all be blamed for variations in different erythrocytic indicators (Farooq et al., 2011).
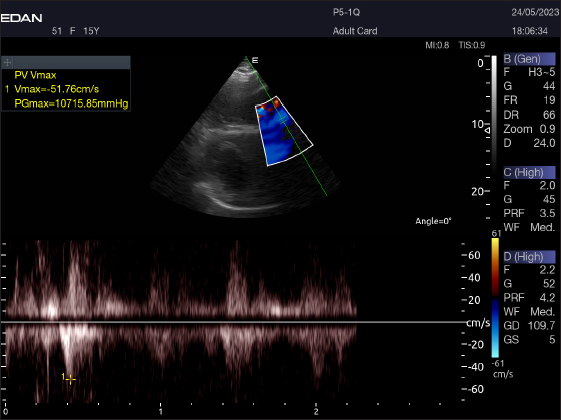
Fig. 7. Right parasternal short axis of a 17-year racing she-camel showing pulsed wave Doppler of pulmonary valve.
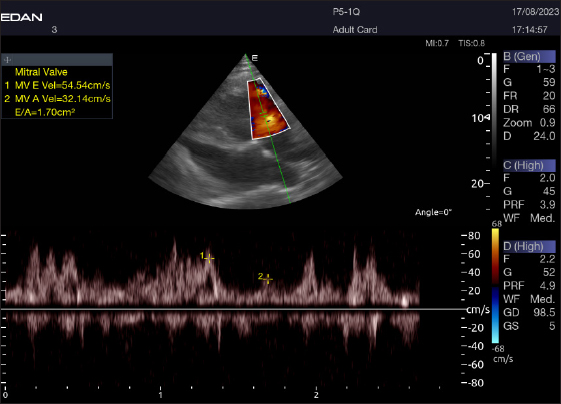
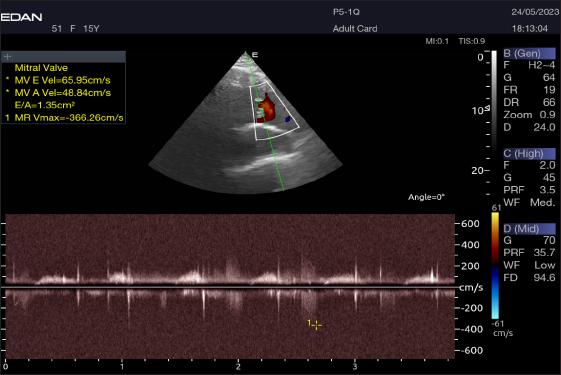
Fig. 8. Left parasternal longitudinal axis of a 16-year-old racing camel showing pulsed wave Doppler over the mitral valve (1=E wave and 2=A wave).
Fig. 9. Left parasternal longitudinal axis of an 18-year-old racing camel showing pulsed wave Doppler over the tricuspid valve (1=E wave and 2=A wave).
Fig. 10. Normal flow of the aortic valve is demonstrated by left caudal–parasternal spectral PWD Doppler in a 20 years old camel.
Fig. 11. Left parasternal longitudinal axis of a 20-year racing she-camel showing mild mitral valve regurgitation by continuous-wave Doppler. Within animals, calcium aids in bone development, balances excessive acidity in the rumen, and permeates bodily fluids such as blood and secretions such as hormones and milk. The serum of camels under examination had Ca values ranging from 10.5 to 15.4 mg/dl (mean 13 ± 1.9). These values were higher than those reported by Mohri et al. 2008 in UAE camels (9.5–11.5 mg/dl), Al-Busadah, 2007 in Saudi camels (7.6–13.1 mg/dl), Faye et al. 1995 in temperate camels (11.5 mg/dl), Eltahir et al. 2016 in the Omani racing Arabian camels, and Elgioushy et al. 2020 in racing camels (11.27 ± 0.9). Approximately 80% of the phosphorus in an animal’s body is required for coprecipitated calcium to maintain and support the skeleton, which is one of the body’s many metabolic processes. In addition to being a vital component of cells, membranes, and bodily fluids, phosphorus is also crucial for energy production. In addition, it is a buffer that is essential for transferring phospholipids, fatty acids, amino acids, and DNA and RNA. Results showed that the tested racing camel P serum levels ranging from 2.1 to 5.4 mg/dl with an average of 3.9 ± 1.1 were lower than those reported by Seleim et al. 2003 in Egyptian camels (6.8 mg/dl), Eltahir et al. 2016 in the Omani racing Arabian camels (Camelus dromedarius) (5.5–8), Mohamed and Hussein, 1999 in Kuwaiti camels, and Osman and Al-Busadah, 2003 in Saudi camels (3.8 mg/dl). The p values found in this investigation were greater than those found in the Bactrian camel (1.65–2.01 mg•dl-1) in the United Arab Emirates by Wernery et al. (1999). Sodium is a mineral that the body and its cells need to function. Camels used in racing depend significantly on it because of the high levels of stress they experience during training and competition. The serum of racing camels had Na concentrations ranging from 94.9 to 181.2 mmol/l, with an average of 149.2 ± 28.3 mmol/l. Our conclusions aligned with the findings of the following by Eltahir et al. 2016 in racing Arabian camels from Oman, Zongping, 2003 in adult Bactrian camels (Camelus bactrianus) in China, Ayoub and Saleh 1998; Wernery et al. 1999, in Emirati camels, Al-Busadah, 2007 in Saudi camels, and Al-Shami, 2009 in the Saudi and Nigerian populations. According to research, Sudanese camels have a greater range of 300–390 mmol/l (Hassan et al., 1968). Compared with studies that included male camels during rutting and those that did not, the sodium levels in this study were marginally reduced (Ahmad et al., 2007). Potassium is the principal ion found inside the cells of all known animal species. Maintaining a healthy fluid and electrolyte balance in the body of an animal depends on it. The nervous system and other animal cells use sodium and potassium to generate electrical potentials. Animals with potassium deficiency display a spectrum of neurological abnormalities. The average K values of the racing camels were tested as 5.5 ± 1.1 mmol/l (ranging from 4 to 7.3 mmol/l), which were similar to the levels discovered in Sudanese camels (Omer et al., 2008); Nigerian camels (Mohammed et al., 2007); Egyptian camels (Seleim et al., 2003); Kuwaiti camels (Mohamed and Hussein, 1999); and Omani racing Arabian camels (Eltahir et al., 2016). Based on the liver function tests, liver enzyme measurements reveal issues with hepatocyte membrane integrity, necrosis of the biliary or hepatocyte epithelium, cholestasis, or induction phenomena. By measuring the length and size of transaminase activities in a sequential manner, one can approximately estimate the number of cells involved, as well as forecast the severity and activity of the disease. Several tissues, including the liver, contain significant amounts of the aminotransferases AST and ALT. The skeletal muscles, kidneys, and heart have greater AST activity than the liver. In the animals under examination, the mean ALT value was 26.9 ± 7.7 U/l, with a range of 12.2–35.4. This seems to be greater than the ALT levels in the blood of Saudi camels (17.2 U/l and 8.0 U/l, respectively) (Osman and Al-Busadah, 2003; Al-Busadah, 2007), Egyptian camels (19.9 U/l) (Seleim et al., 2003), Omani racing Arabian camels (13.3 U/l) (Eltahir et al., 2016), and racing camels (Camelus dromedarius) (8.60 ± 2.06 U/l) (Elgioushy et al., 2020). The average AST level in the racing camels studied was 95 ± 27.2 U/l, ranging from 19.1 to 122.4 U/l. This seems to be greater than the AST levels in the blood of Saudi camels, which were recorded to range between 24.1 and 35.1 U/l (Al-Busadah, 2007), Egyptian camels (31.4 U/l) (Seleim et al., 2003), Omani racing Arabian camels (57 U/l) (Eltahir et al., 2016), racing camels (Camelus dromedarius) (37.7 U/l) (Elgioushy et al., 2020), and race camels (85.5 U/l) (Tharwat, 2021). However, it is less than the average AST value in Saudi camels (164.6 U/l) described by Osman and Al-Busadah (2003). To ascertain whether the variations in AST between Omani camels and camels from other regions of the world are genetic or clinical, more research is required. The cardiac troponins, particularly cardiac troponin I (cTnI), are the most effective cardiac indicators of myocardial damage. The tropomyosin and actin on the thin filaments of striated muscle fibers are bound by the troponin complex subunit cTnI (Hastings, 1997). The myocardium expresses only cTnI (Lan and Bell, 2019). After acute myocardial injury, leakage from destroyed myocardial cells causes a rise in the blood cTnI level (Gupta et al., 2018). Racing camel serum had a cTnI concentration ranging from 0.2 to 0.4, with an average of 0.3 ± 0.02 ng/ml. These levels were higher than what was stated by Elgioushy et al. 2020 at 0.04 ± 0.02 ng/ml, Tharwat, 2021 at 0.03 ± 0.03 ng/ml, and Tharwat et al. 2013 at 0.01–0.07 ng/ml. The causes of the observed increases in cTnI concentrations are unclear. It seems improbable that these increases are connected to cross-reactivity with skeletal muscle because cardiac troponin I is said to have a very high selectivity for the myocardium (O’brien et al., 2006). Consequently, it is probable that the elevated cTnI of the camels was related to the racing camels employed in this study, which were clinically healthy and did not have any cardiac condition that might have caused the increases in cTnI. Elevated levels of cTnI have also been linked to high-intensity exercise in sled dogs, moderate-duration exercise in humans, and endurance exercise in horses (Wakshlag et al., 2010). Moderate hypoxia during exercise may alter the permeability of the myocardium, allowing macromolecules to seep into the bloodstream, according to one explanation explaining the rise in cTnI levels (Nostell and Häggström, 2008). The finding that brief exposure to hypoxia can cause the release of cTnI without causing cell death in rats supports this theory (Piper et al., 1984). Therefore, it is unclear whether this can have any clinical significance in spite of the large increase. Serum troponin levels can increase due to damage to cardiomyocytes caused by nonprimary cardiac disorders in people (Mahajan, 2006). An increased risk of sepsis, gastrointestinal bleeding, pulmonary embolism, diabetic ketoacidosis, COPD, and other conditions has been linked to elevated cTnI levels (Mahajan, 2006). Researchers found elevated cTnI levels in dogs and cats suffering from azotemia due to renal failure, noncardiac systemic diseases, gastric dilatation, and volvulus. This suggests the degeneration and necrosis of cardiomyocytes (Porciello et al., 2008). Postmortem investigations in cattle with noncardiac intrathoracic diseases have also failed to detect any substantial cardiac abnormalities, leading to similar outcomes (Mellanby et al., 2009). Although the precise process remains a mystery, most studies have suggested that the heart plays a supporting role in these activities, leading to an increase in cTnI. The camels in the present study may also fall into this category. Therefore, additional research is required to identify the causes of the increase in cTnI. To further understand the relationship between these factors and cTnI concentrations, more studies on racing-induced stress are required. Finally, to determine the link between high cTnI and adverse cardiac outcomes, investigations assessing the blood cTnI concentration of camels exhibiting indications of heart disease are necessary. Our study’s limitations compared with other published research stem from two main issues: first, the species we examined has received little attention in the field of animal science, and second, there has been a lack of consistency in the methods used to evaluate transportation stress. Echocardiography allows for the assessment of the heart’s dimensions as well as examination of its composition and operation. One of the most important ways to assess the severity and prognosis of cardiac disease in camels and cattle is to measure the ventricular chamber diameters (Holbrook et al., 2006; Buczinski, 2009; Tharwat et al., 2012). Dehghan et al. (2011) reported that the echocardiographic results of camels that were 1 year old were within the normal ranges. This is the first study to demonstrate the ability of adult racing camels to measure internal cardiac features and take heart images. As a result, it is better to conduct the camel’s ultrasonography examination when seated rather than standing (Tharwat et al., 2012). The short-axis view was technically simple, but Holbrook et al. 2006 claimed that it was technically challenging and had poor visualization because of pleural hyperechogenicity. In addition to probe size, compression of the thoracic intercostal spaces, and costal bone’s detrimental effect on the acquired images, other obstacles were encountered. Observed in cattle (Braun et al., 2001; Tharwat et al., 2012), the ultrasound images were of poor quality because the camel thoracic wall was thick, which attenuated the beam through absorption and reflection. All camels had little trouble obtaining the four-chamber view, which displays the atria, ventricles, and valves. However, because this study was conducted without sedation, unlike previous research (Braun et al., 2001; Tharwat et al., 2012), securing the camels required physical work. The cranial left and right parasternal long axis (five chamber view) is where the aortic valve is obtained, in contrast to Braun et al., 2001, who received the aorta from the right only. A right parasternal long-axis four-chamber picture was produced and aligned with Tharwat et al. 2012 after placing the probe in the fifth intercostal space. The pulsed-wave Doppler echocardiography of racing camels is not well documented. While all camels had their Mv E/A, Tv E/A, and maximum velocity of the Av Vmax recorded using pulsed-wave Doppler at (LPSLAx), the majority of cases (58.4%) had trouble measuring the pulmonary valve from the right short parasternal view. Using continuous-wave Doppler, minor mitral regurgitation was observed in two patients, but the regurgitation was not substantial. Some limitations of this study should be taken into account in future research. The present study only included a small sample of racing camels as starters. Consequently, a plethora of racing camels require further investigation. Second, to make a more accurate health judgment, this inquiry should also be conducted in camels that have heart disorders, and the pulsed-wave echocardiographic and echocardiographic measurements should be compared between the diseased and healthy camels. Third, only a few biochemical parameters were tested. Therefore, future studies must consider a variety of aspects. In addition, camels have thick skin and a huge chest cavity, which degrades the clarity of images. Fifth, because of the location of the heart and ribs, pulmonary flow at the right parasternal short axis was difficult. Finally, the quality of the Doppler echocardiogram is affected by the left-sided angulation of the heart location. ConclusionThis research presents data showing that mature racing camels can have good-quality echocardiograms and pulsed-wave Doppler echocardiograms with normal heart dimensions, compared with sick animals. Therefore, our findings are pertinent for the early detection of heart diseases in camels used for racing. Although the method is quite simple, it does require some physical strength. It may be possible to confirm or refute the existence of heart illnesses in racing camels with cardiovascular disease by applying the normal ranges developed in this study. Furthermore, pulsed-wave Doppler echocardiography and echocardiography readings from healthy racing camels can be used to predict the prognosis of sick racing camels with cardiac conditions. Thus, it is important to compare the results of echocardiography and pulsed-wave Doppler echocardiography with the typical reference ranges that were discussed in this study. AcknowledgmentThe Desert Research Center Animal Health and Poultry Department employees in Egypt were acknowledged by the authors. Conflict of interestThe authors declare no conflicts of interest. FundingThe author(s) received no financial support for research, authorship, or publication of this article. Authors’ contributionsAhmed Adel El-Sayed and Sabry Mousa contributed to the main design of this study, supported the experiment, wrote, analyzed data, reviewed, and edited the manuscript. Ahmed Al-Montaser El-Sayed, Attia Eissa, and Ahmed Ragab Askar led the experiment, collected blood samples, analyzed serum metabolites, and reviewed and edited the manuscript. Mohamed East Ali performed echocardiography and pulsed-wave Doppler echocardiography, wrote part of the manuscript, and participated in critical checking of the manuscript. Data availabilityAll data used and/or analyzed during the current study are available in the manuscript. ReferencesAbdel-Ghaffar, F., Mehlhorn, H., Bashtar, A.R., Al-Rasheid, K., Sakran, T. and El-Fayoumi, H. 2009. Life cycle of Sarcocystis camelicanis infecting the camel (Camelus dromedarius) and the dog (Canis familiaris), light and electron microscopic study. Parasitol. Res. 106, 189–195. Ahmad, N., Bukhari, S.A., Akhtar, N. and Haq, I. 2007. Serum hormonal, electrolytes and trace element profiles in the rutting and non-rutting one-humped male camel (Camelus dromedarius). Anim. Reprod. Sci. 101(1-2), 172–178. Al-Busadah, K.A. 2007. Some biochemical and haematological indices in different breeds of camels in Saudi Arabia. Sci. J. King Faisal Univ (Basic and Applied Sciences). 8 (1), 1428. Al-Shami, S.A. 2009. Comparative determination of serobiochemical constituents in-door and free grazing camels. J. Anim. Vet. Adv. 12(2), 141–143. Ayoub, M. and Saleh, A. A comparative physiological study between camels and goats during water deprivation. Paper presented at the Proceedings of the 3rd annual meeting for animal production under arid conditions. UAE University, P. O. Box 17555, Al-Ain, United Arab Emirates. 1998, vol. 1, p 71–87. Braun, U., Schweizer, T. and Pusterla, N. 2001. Echocardiography of the normal bovine heart: technique and ultrasonographic appearance. Vet. Record. 148(2), 47–51. Buczinski, S. 2009. Cardiovascular ultrasonography in cattle. Vet. Rec. Food Anim. Pract. 25(3), 611–632. Burton, D.A., Stokes, K. and Hall, G.M. 2004. Physiological effects of exercise. Continuing Educ. Anaesthesia Crit. Care Pain. 4(6), 185–188. Dehghan, A., Rezakhani, A., Veshgini, A., Zadeh, M.N. and Bagheri, K. 2011. Normal echocardiographical parameters of healthy camel (Camelus dromedarius). J. Camel Pract. Res. 18(1), 69–72. Elgioushy, M., Noseer, E., Rizk, M.A. and El-Adl, M. 2020. Effect of racing on blood gases components and selected biochemical variables in racing camels (Camelus dromedarius). Asian J. Anim. Vet. Adv. 15(1), 38–44. Eltahir, Y.E., Ali, H.M., Hago, B. and Mahgoub, O. 2016. Serum biochemistry parameters in the Omani racing Arabian camels (Camelus dromedarius). J. Agric. Mar. Sci. 21, 65–76. Evans, D., Rose, R., Knight, P., Cluer, D. and Mannefield, G. 1992. Physiological responses during an incremental treadmill exercise test in the camel. Conference paper, UK, 0-9516604-4-6, Newmarket, Proceedings of the First International Camel Conference, Dubai. 223–227. Faraz, A., Waheed, A., Mirza, R., Ishaq, H. and Tariq, M. 2019. Socio economic status and associated constraints of camel production in desert Thal Punjab. Pakistan. J. Fish. Livest. Prod. 7(01), 288. Farooq, U., Samad, H., Khurshid, A. and Sajjad, S. 2011. Normal reference hematological values of one-humped camels (Camelus dromedarius) kept in Cholistan desert. J. Anim. Plant Sci. 21(2), 157–160. Faye, B., Ratovonanahary, M., Chacornac, J.P. and Soubre, P. 1995. Metabolic profiles and risks of diseases in camels in temperate conditions. Comparative Biochem. Physiol. Part A: Physiol. 112(1), 67–73. Fowler, M. 2011. Medicine and surgery of camelids. Hoboken, NJ: John Wiley & Sons. Gupta, K.K., Srivastava, M., Sudan, V., Singh, S.K., Choudhury, S. and Shanker, D. 2018. Variation in cardiac markers and electrocardiographic alterations in young calves naturally infected with bovine tropical theileriosis. Trop. Anim. Health Prod. 50(6), 1227–1230. Hallowell, G., Potter, T. and Bowen, I. 2007. Methods and normal values for echocardiography in adult dairy cattle. J. Vet. Cardiol. 9(2), 91–98. Hassan, Y., Hoeller, H. and Hassan, I. 1968. Observations on the blood constituents of camels in the Sudan. Sudan J. vet. Sci. anim. 9(1), 464–476. Hastings, K.E. 1997. Molecular evolution of the vertebrate troponin I gene family. Cell Struct. Funct. 22(1), 205–211. Holbrook, T., Birks, E., Sleeper, M. and Durando, M. 2006. Endurance exercise is associated with increased plasma cardiac troponin I in horses. Equine Vet. J. 38(S36), 27–31. Lan, N.S. and Bell, D.A. 2019. Revisiting the biological variability of cardiac troponin: implications for clinical practice. Clin. Biochem. Rev. 40(4), 201. Mahajan, A. 2006. Identification and estimation of regression models with misclassification. Econometrica. 74(3), 631–665. Martín-Barrasa, J.L., Tejedor-Junco, M.T., Cabrera, S., Morales, M., Melián, A. and Corbera, J. A. 2023. Haematological and biochemical blood reference values for Canary Island camels (Camelus dromedarius), an endangered dromedary species. Saudi J. Biol. Sci. 30(6), 103677. Masoudifard, M., Esmailinejad, M., Sakhaee, E., Azari, O., Vafaei, R. and Ghalekhani, N. 2020. Pulsed wave Doppler echocardiographic assessment after sedation by intravenous injection of medetomidine and xylazine hydrochloride on cardiac output and systolic time intervals in one-humped camel calves (Camelus dromedarious). Iran. J. Vet. Res. 21(4), 257. Mellanby, R.J., Thomas, D.C. and Lamb, J. 2009. Role of regulatory T-cells in autoimmunity. Clin Sci. 116(8), 639–649. Mohamed, H. and Hussein, A. 1999. Studies on normal haematological and serum biochemical values of the ‘Hijin’racing camels (Camelus dromedarius) in Kuwait. Vet. Res. Commun. 23, 241–248. Mohamed, T. and Buczinski, S. 2011. Clinicopathological findings and echocardiographic prediction of the localisation of bovine endocarditis. Vet. Rec. 169(7), 180. Mohammed, A., Sackey, A., Tekdek, L. and Gefu, J. 2007. Serum biochemical values of healthy adult one humped camel (Camelus dromedarius) introduced into a sub-humid climate in Shika-Zaria, Nigeria. Mohri, M., Moosavian, H. and Hadian, M. 2008. Plasma biochemistry of one-humped camel (Camelus dromedarius): effects of anticoagulants and comparison with serum. Res. J. Vet. Sci. 85(3), 554–558. Nostell, K. and Häggström, J. 2008. Resting concentrations of cardiac troponin I in fit horses and effect of racing. J. Vet. Cardiol. 10(2), 105–109. O’brien, P., Smith, D., Knechtel, T., Marchak, M., Pruimboom-Brees, I., Brees, D., Potter, A. 2006. Cardiac troponin I is a sensitive, specific biomarker of cardiac injury in laboratory animals. Lab. Anim. 40(2), 153–171. Omer, S., Agab, H., Samad, G. and Turki, I. 2008. Effect of feed type on some blood constituents of Sudanese growing camel (Camelus dromedarius) calves. Sud. J. Vet. Sci. Anim. Husb. 47, 107–115. Osman, T. and Al-Busadah, K. 2003. Normal concentrations of twenty serum biochemical parameters of she-camels, cows and ewes in Saudi Arabia. J. Mol. Cell. Cardiol. 6(14), 1253–1256. Piper, H., Schwartz, P., Spahr, R., Hütter, J. and Spieckermann, P. 1984. Early enzyme release from myocardial cells is not due to irreversible cell damage. J. Mol. Cell. Cardiol.16(4), 385–388. Porciello, F., Rishniw, M., Herndon, W., Birettoni, F., Antognoni, M.T. and Simpson, K. 2008. Cardiac troponin I is elevated in dogs and cats with azotaemia renal failure and in dogs with non-cardiac systemic disease. Aust. Vet. J. 86(10), 390–394. Schiller, N.B., Shah, P.M., Crawford, M., DeMaria, A., Devereux, R., Feigenbaum, H., Schnittger, I. 1989. Recommendations for quantitation of the left ventricle by two-dimensional echocardiography. J Am Soc Echocardiogr. 2(5), 358–367. Seleim, R., Amal, R., Sahar, R., Nada, H. and Gobran, R. 2003. ELISA and other tests in the diagnosis of Pasteurella multocida infection in camels. Deutscher Tropentag, International Research on Food Security, Natural Resource Management and Rural Development Georg-August-Universität Göttingen. Skidmore, J. and Billah, M. 2011. Embryo transfer in the dromedary camel (Camelus dromedarius) using non-ovulated and ovulated, asynchronous progesterone-treated recipients. Reprod. Fertil. Dev. 23(3), 438–443. Snow, D. An introduction to the racing camel. Proc. 1st Int Camel Conf, 1992, p 215–217. Suliman, G.M., Alowaimer, A.N., Hussein, E.O., Ali, H.S., Abdelnour, S.A., El-Hack, M.E. A. and Swelum, A.A. 2019. Chemical composition and quality characteristics of meat in three one-humped camel (Camelus dromedarius) breeds as affected by muscle type and post-mortem storage period. Animals. 9(10), 834. Swelum, A.A.A., Saadeldin, I.M., Abdelnour, S.A., Ba-Awadh, H., Abd El-Hack, M.E. and Sheiha, A.M. 2020. Relationship between concentrations of macro and trace elements in serum and follicular, oviductal, and uterine fluids of the dromedary camel (Camelus dromedarius). Trop Anim Health Prod. 52, 1315–1324. Tharwat, M. 2021. Influence of 8 Km Training On Cardiac Biomarkers Alongside Haematobiochemical Profiles In Race Camels. J. Camel Pract. Res. 28(1),79–84. Tharwat, M., Al-Sobayil, F., Ali, A. and Buczinski, S. 2012. Echocardiography of the normal camel (Camelus dromedaries) heart: technique and cardiac dimensions. BMC Vet Res. 8, 1–8. Tharwat, M., Al-Sobayil, F. and Buczinski, S. 2013. Cardiac biomarker changes in camels (Camelus dromedarius) secondary to road transportation. J. Vet. Cardiol. 15(1), 15–22. Tibary, A. and El Allali, K. 2020. Dromedary camel: a model of heat resistant livestock animal. Theriogenology 154, 203–211. Van Saun, R.J. Blood profiles as indicators of nutritional status. Paper presented at the InProc. 18th Annu. Western Canadian Dairy Seminar. Red Deer Alberta, Canada. 2000, 1–6. Wakshlag, J., Kraus, M., Gelzer, A., Downey, R. and Vacchani, P. 2010. The influence of high-intensity moderate duration exercise on cardiac troponin I and C-reactive protein in sled dogs. J. Vet. Intern. Med. 24(6), 1388–1392. Wernery, U., Fowler, M.E. and Wernery, R. 1999. Color atlas of camelid hematology. Blackwell Wissenschafts-Verlag, Blackwell. Berlin, Germany. Zongping, L. 2003. Studies on the haematology and trace element status of adult Bactrian camels (Camelus bactrianus) in China. Vet. Res. Commun. 27, 397–405. | ||
| How to Cite this Article |
| Pubmed Style El-sayed AA, El-sayed AA, Ali ME, Eissa A, Askar AR, Mousa S. Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camels. Open Vet. J.. 2025; 15(2): 994-1008. doi:10.5455/OVJ.2025.v15.i2.48 Web Style El-sayed AA, El-sayed AA, Ali ME, Eissa A, Askar AR, Mousa S. Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camels. https://www.openveterinaryjournal.com/?mno=240616 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.48 AMA (American Medical Association) Style El-sayed AA, El-sayed AA, Ali ME, Eissa A, Askar AR, Mousa S. Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camels. Open Vet. J.. 2025; 15(2): 994-1008. doi:10.5455/OVJ.2025.v15.i2.48 Vancouver/ICMJE Style El-sayed AA, El-sayed AA, Ali ME, Eissa A, Askar AR, Mousa S. Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camels. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 994-1008. doi:10.5455/OVJ.2025.v15.i2.48 Harvard Style El-sayed, A. A., El-sayed, . A. A., Ali, . M. E., Eissa, . A., Askar, . A. R. & Mousa, . S. (2025) Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camels. Open Vet. J., 15 (2), 994-1008. doi:10.5455/OVJ.2025.v15.i2.48 Turabian Style El-sayed, Ahmed Al-montaser, Ahmed Adel El-sayed, Mohamed E. Ali, Attia Eissa, Ahmed Ragab Askar, and Sabry Mousa. 2025. Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camels. Open Veterinary Journal, 15 (2), 994-1008. doi:10.5455/OVJ.2025.v15.i2.48 Chicago Style El-sayed, Ahmed Al-montaser, Ahmed Adel El-sayed, Mohamed E. Ali, Attia Eissa, Ahmed Ragab Askar, and Sabry Mousa. "Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camels." Open Veterinary Journal 15 (2025), 994-1008. doi:10.5455/OVJ.2025.v15.i2.48 MLA (The Modern Language Association) Style El-sayed, Ahmed Al-montaser, Ahmed Adel El-sayed, Mohamed E. Ali, Attia Eissa, Ahmed Ragab Askar, and Sabry Mousa. "Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camels." Open Veterinary Journal 15.2 (2025), 994-1008. Print. doi:10.5455/OVJ.2025.v15.i2.48 APA (American Psychological Association) Style El-sayed, A. A., El-sayed, . A. A., Ali, . M. E., Eissa, . A., Askar, . A. R. & Mousa, . S. (2025) Pulsed-wave Doppler echocardiographic and hematobiochemical profiles of clinically healthy racing dromedary camels. Open Veterinary Journal, 15 (2), 994-1008. doi:10.5455/OVJ.2025.v15.i2.48 |