| Review Article | ||
Open Vet. J.. 2025; 15(2): 565-593 Open Veterinary Journal, (2025), Vol. 15(2): 565-593 Review Article Polymeric rumen-stable delivery systems for delivering nutricinesYedi Herdiana1,2*1Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, Universitas Padjadjaran, Sumedang, Indonesia 2Veterinary Pharmaceutical Development Group, Faculty of Pharmacy, Universitas Padjadjaran, Sumedang, Indonesia *Corresponding Author: Yedi Herdiana, Department of Pharmaceutics and Pharmaceutical Technology, Faculty of Pharmacy, Universitas Padjadjaran, Sumedang, Indonesia. Email: y.herdiana [at] unpad.ac.id Submitted: 13/11/2024 Accepted: 21/01/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
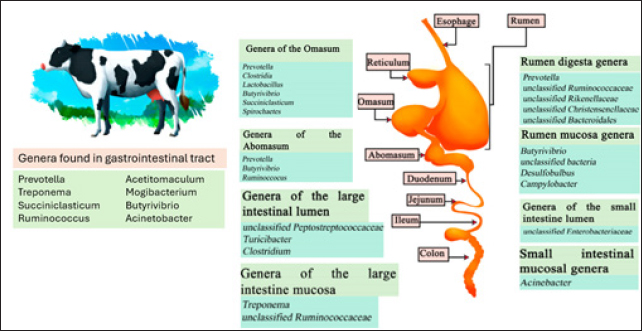
AbstractRuminants face unique drug and nutrient delivery challenges because of their symbiotic rumen microorganisms. Polymeric rumen-stable delivery systems (RDSs) have emerged as a promising solution for efficiently delivering nutrition and enhancing animal health and productivity. Traditional methods such as heat and chemical treatment have been improved with polymeric coatings that facilitate the slow postruminal release of bioactive substances. Polymeric coatings of nutrients offer significant potential for improving ruminant health, reducing farmer costs, and promoting sustainability in livestock. This paper explores the mechanisms of rumen protection and abomasal release provided by polymeric coatings, discusses other RSDs, and reviews methods for evaluating their performance in vitro and in vivo. Further research in this area could advance novel nutricine delivery solutions for ruminants. Keywords: Ruminants; Polymeric coatings; Nutricines; Livestock industry. IntroductionAs the global demand for animal food increases, especially in Indonesia, the need for efficient and sustainable methods to improve livestock health and productivity is becoming increasingly important. These methods help reduce malnutrition and increase household and food security (Kappes et al., 2023). Currently, animal husbandry faces several challenges due to a lack of available arable land, clean water, ongoing climate issues, competition for feed and fuel, and a shortage of animal feed ingredients (Malenica et al., 2023). Despite this, the demand for red meat is projected to double in 2050, while national beef production in Indonesia is currently only able to meet around 45% of the demand (Agus and Widi, 2018). A significant proportion of beef production is carried out by smallholders, with commercial entities contributing a small proportion. This presents an excellent opportunity to strengthen the Indonesian beef sector through technological innovation, thereby increasing the productivity and profitability of these smallholder farmers (Burrow, 2019). Historically, the livestock industry has faced several hurdles, such as limited access to innovative technologies, investment, veterinary drugs, and superior seeds (Jaime et al., 2022). With rapid growth, innovative technologies are playing an increasingly important role. Traditional procedures are labor-intensive, time-consuming, and technical and require skilled specialists and specialized equipment (Akhigbe et al., 2021; Džermeikaitė et al., 2023). Increased investments in technology, veterinary drugs, and superior seeds can reduce the country’s dependence on food imports, strengthen the national food security, and improve the welfare of small-scale beef farmers. Because local production cannot meet national food needs, import arrangements are made. However, this policy is unsuitable for the long term because of the risk of dependence on imported food (Zuhud, 2020). The emerging field of pharmaceutical sciences provides potential solutions to the challenge of effective nutritional delivery. Pharmaceutical sciences deal extensively with biocompatible drug carriers for the transport of molecules in pharmaceutical, cosmetic, and nutraceutical applications. The main advantages of this strategy are increasing efficacy, reducing the dose, and controlling the delivery of bioactive compounds (Halmemies-Beauchet-Filleau et al., 2018). Combining preparation modification and food delivery science will create a new “smart food” system that can improve health and well-being (Martínez-Ballesta et al., 2018). Controlled delivery systems for encapsulating bioactive compounds or nutrients to achieve the desired efficacy in animal feeds. Nanoencapsulation offers better protection, absorption, and delivery of bioactives (Siddiqui et al., 2022). Developing systems that can withstand the ruminant digestive system is essential for effective and cost-effective treatment regimens that support animal health and productivity. These delivery systems, made from biodegradable or nonbiodegradable polymers, provide controlled bioactive release, benefiting ruminant health and livestock productivity. Biodegradable materials are gaining attention due to their adaptable characteristics, such as electrical conductivity and biodegradability, which make them attractive in many applications (Bilhalva et al., 2018; Tran and Tran, 2019). Micronutrients are essential for optimizing animal feed use and represent the most significant single cost in livestock production, often accounting for 60% of the expenses. Micro ingredients such as soy, corn, and wheat can vary significantly in density and nutritional value, and antinutritional factors usually hinder their digestibility. Despite these challenges, advancements in feed technologies have enhanced feed quality and livestock productivity. Many of these technologies have been successfully adopted and scaled up to increase income (Balehegn et al., 2020). A key aspect of these polymeric-controlled delivery systems is the inclusion of nutricines—bioactive substances such as carotenoids, enzymes, fatty acids, flavors, oligosaccharides, organic acids, phospholipids, and polyphenols (Martínez-Ballesta et al., 2018; Borandeh et al., 2021). These components have been extensively researched for their roles in maintaining animal health and preventing disease, making them well-suited for long-term, controlled-release delivery systems (da Silva et al., 2020; Broda et al., 2024). Current methods such as heat and chemical treatments often fail to protect bioactive substances from rumen degradation and ensure their effective release in the abomasum (Iommelli et al., 2022; Pena et al., 2023; Davidson et al., 2024). This gap in effective delivery systems hinders optimal nutrient utilization and increases farmers’ costs. Polymeric rumen-stable delivery systems (RDSs) offer a promising solution for the slow, postruminal release of bioactive substances. While these systems show significant potential in improving ruminant health, reducing costs, and promoting sustainability, there is a need for comprehensive research to optimize their performance and explore new materials (Bešlo et al., 2022; Albuquerque et al., 2023). This study focused on the potential of polymeric RDS for delivering nutrients. We aim to explain how these delivery systems can enhance ruminant health and productivity by addressing the unique challenges of ruminant biology and finding ways to overcome them. As we examine these delivery systems in detail, we highlight their potential for sustainable livestock management, improved animal health, and overall food security. Rumen-stable Delivery SystemsThe digestive system of ruminantsUnderstanding the unique digestive tract and digestion process of ruminants, along with the harsh conditions of degradation in the rumen environment, is crucial for developing carriers for various active compounds and nutrients (Galyon et al., 2022). The advancement of these preparations offers promising prospects for ruminant drug delivery. The ruminant rumen hosts a complex ecosystem of bacteria, protozoa, and fungi that break down plant cell walls (Zhang et al., 2022). With their large numbers and diverse metabolic pathways, rumen bacteria dominate this ecosystem. They play a key role in the digestion of cellulose, whereas protists and fungi contribute through various mechanisms (Weimer, 2022). These bacteria break down approximately half of the crude fiber consumed by ruminants. The three most common fiber-degrading rumen bacteria are Ruminococcus flavefaciens, Ruminococcus albus, and Fibrobacter succinogenes. The digestion of cellulose and hemicellulose in food (Hua et al., 2022; Weimer, 2022; Gharechahi et al., 2023). Rumen bacteria help to break down nutrients such as starch, xylan, and pectin. This process is mainly performed by amylolytic bacteria such as Prevotella ruminicola and Streptococcus bovis (Palevich et al., 2019; Wei et al., 2022). Some bacteria, such as Fibrobacter succinogenes and Butyrivibrio fibrisolvens, also break down cellulose and starch, contributing further to the complex digestive processes in the rumen (Hua et al., 2022). Protein degradation is another vital function of rumen bacteria, particularly Ruminobacter amylophilus and Butyrivibrio fibrisolvens (Liu et al., 2019; Mohamaden et al., 2020). These bacteria convert plant and nonprotein nitrogen, which the host cannot use directly, into microbial protein (Wei et al., 2022; Zhu et al., 2022). Other bacterial species, including Clostridium spp., Eubacterium ruminantium, Prevotella spp., and Selenomonas ruminantium, also play roles in protein degradation (Zhu et al., 2022; Arjun et al., 2023). The distribution of bacteria in the ruminant digestive system is shown in Figure 1. Several bacteria produce lactic acid, an essential intermediate product, in the rumen. Lactobacillus, Streptococcus, Enterococcus, and Pediococcus are the primary lactic acid producers. Excessive lactic acid production due to an imbalance between lactic acid-producing and lactic acid-utilizing bacteria can lead to rumen acidosis (Lee et al., 2019; He et al., 2022; Hu et al., 2022). The rumen hosts methane-producing archaea known as methanogens. Recent studies suggest that feed additives can reduce methane emissions from ruminants, thereby mitigating the environmental impact of ruminant farming (Li et al., 2019b; Getabalew et al., 2020). Although less numerous than bacteria, protozoa comprise a significant portion of the rumen microbial biomass due to their larger size. Protozoa comprise 50% of the rumen biomass and play a key role in rumen metabolism, contributing significantly to volatile fatty acid (VFA) production through the fermentation of feedstuffs and engulfing bacteria. Removing protozoa (defaunation) can reduce animal performance by approximately 10% (Perez et al., 2024). Protozoa and rumen viruses influence the rumen microbiome through interactions between top-down (predation) and bottom-up (metabolic impact). Protozoa regulate other microbes through predation and metabolic effects, whereas viruses act as intracellular predators, lysing cells, and reprogramming host metabolism to enhance ecological fitness (Yu et al., 2024). Entodinium and Epidinium, two dominant genera of the order Entodiniomorphida, were found in over 99% of 592 rumen samples, with mean abundances of ~38% and 16%, respectively (Andersen et al., 2023). The diverse rumen viruses can infect most lineages of the rumen microbiomes, including 1,051 genera of bacteria, 25 genera of archaea, and 13 genera of protozoa (Yu et al., 2024).
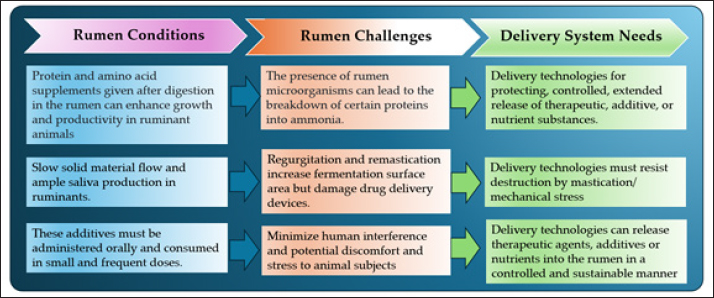
Figure 1. The digestive system of cows. Microorganisms are frequently found in different sections of the gastrointestinal tract compartments (Huaiquipán et al., 2023). Anaerobic fungi, such as Neocallimastix, Piromyces, and Orpinomyces, are significant in the initial colonization of plant material. Anaerobic fungi (phylum Neocallimastigomycota) inhabit the alimentary tract of herbivores and display multiple adaptive strategies that enable them to survive and thrive in this permanently anoxic, prokaryote-dominated ecosystem (Elshahed et al., 2022). Anaerobic fungi (Neocallimastigomycota) are common in the digestive tracts of mammalian herbivores and can comprise up to 20% of the rumen microbial biomass. They primarily degrade lignocellulosic plant material and have a syntrophic relationship with methanogenic archaea, enhancing fiber degradation (Edwards et al., 2017). Anaerobic gut fungi form rhizoidal structures to enhance plant attachment and colonization, which facilitate taxonomy. Hyphal coils wrap around plant fibers, maximizing contact, while appressoria develop as multilobed vesicles with penetration pegs to aid nutrient absorption (Hanafy et al., 2022). While producing highly active cellulases, ruminal fungi largely degrade plant material through the physical force exerted by growing hyphal tips (appressoria) that fracture plant tissues, facilitating bacterial invasion. This capability gives them a significant role in plant biomass degradation (Weimer, 2022). Methanogenic archaea are diverse microorganisms crucial to global carbon cycling, producing methane as a by-product of energy production (Volmer et al., 2023). Common species include Methanobrevibacter ruminantium and Methanobacterium formicicum. Methanogenic archaea in the gut act as a hydrogen sink, facilitating short-chain fatty acid production. Dysbiosis of these methanogens is linked to diseases such as inflammatory bowel disease (IBD). Although archaea diversity is higher in patients with IBD, methanogen prevalence and abundance decrease, particularly in ulcerative colitis (Cisek et al., 2024). Recent advancements in sequencing technology and omics have provided profound insights into the rumen world, wherein a consortium of archaea, bacteria, protozoa, fungi, and viruses exist and interact (Sanjorjo et al., 2023). Need for rumens-table delivery systemsProlonged drug release systems can reduce the need for human intervention in livestock and domestic animals, thereby enhancing therapeutic efficacy while minimizing potential discomfort and stress for the animals (Hayward et al., 2018). The ruminant digestive system presents a unique challenge, as developing delivery systems that can survive the rumen environment and ensure drugs reach the desired absorption site is crucial for effective disease prevention and treatment in ruminants (Fleming et al., 2019). The development of drug preparations is expected to have a long-term impact. Beyond merely addressing nutritional deficiencies, these systems will significantly contribute to maintaining optimal health and productivity in ruminants (Bionaz et al., 2020; Sprinkle et al., 2021). Long-acting drug delivery systems can also be used to prevent and treat infectious diseases caused by viruses, bacteria, protozoa, and fungi, which can lead to severe health issues if left untreated. Additionally, these systems are effective against parasites such as worms, ticks, and mites, which disrupt livestock productivity by interfering with feeding and resting patterns, thereby reducing feed conversion efficiency and weight gain. Rumen-stable formulations are particularly beneficial for treating conditions such as bloating, ketosis, and acidosis in livestock (Youssef et al., 2019). Digestive system of ruminants preparationPhysiological considerations Ruminants, such as cattle, have a unique digestive system with four distinct gastric compartments: the rumen, reticulum, omasum, and abomasum (Lei et al., 2018; Pokhrel and Jiang, 2024). Each compartment plays a different role in digestion. Rumen microbes are crucial for breaking down food into simpler, more digestible components (Xu et al., 2021). Plant nutrients are converted into energy that animals can use efficiently and produce B vitamins, vitamin K, and amino acids, which are vital for animal health and growth (Suarjana et al., 2021). Understanding these microbes will help in designing effective long-term drug delivery systems by considering their interactions to optimize drug delivery in ruminants. When designing an RDS, it is essential to consider the pH difference between the rumen and the abomasum (Diao et al., 2019; Hu et al., 2019). This pH difference can create a system that remains stable in the rumen and releases the drug into the abomasum, where it can be absorbed. New systems use ingredients sensitive to these pH changes to enable targeted drug or nutrient release. Rumen motility and rumination effects on particle degradation are also crucial. The size and density of particles in the delivery system affect their movement and behavior in the rumen. Recent studies have shown that nano- and microparticles can enhance the effectiveness and stability of drug delivery systems in the rumen. Thus, designing these systems requires consideration of the animal’s specific physiological conditions and the drug’s characteristics (Vítor et al., 2021). Ruminants differ from other mammals because most of their food is fermented in the rumen, reticulum, and omasum. Although postgastric fermentation occurs in the cecum and colon, it is less significant than that in other herbivores (Wang et al., 2020; Zou et al., 2020; Soltis et al., 2023). This unique digestive system allows for memorable interactions with dietary supplements. For instance, protein supplements and postruminal amino acids can improve growth and productivity. However, rumen microbes decompose some proteins into ammonia, which is then absorbed and excreted as urea, indicating nitrogen loss from the diet. This highlights the need for stable rumen formulations to ensure efficient nutrient delivery. Changes in the ruminant diet significantly affect the rumen bacterial communities (Ramos et al., 2021). The brooding process, which involves regurgitation and demystification, increases the substrate surface area for microbial fermentation (Wang et al., 2020; Zou et al., 2020). However, this can damage drug delivery devices, so the technology must be designed to withstand mechanical stress. The gases produced during rumen fermentation can also affect the performance of some delivery systems, presenting another challenge in designing RDS (Hamid et al., 2020; Ungerfeld, 2020). These gases could potentially be used to develop new drug delivery technologies. Nutrients must be administered orally and consumed in small, frequent doses, emphasizing the need for innovative delivery systems that efficiently and economically provide these additives to grazing ruminants. There is a growing demand for devices that can release therapeutic agents, additives, or nutrients into the rumen in a controlled and sustainable manner. Drug delivery systems are designed to improve user compliance by extending the release of therapeutic agents over time (McGrath et al., 2018; Hu et al., 2019; Fonseca et al., 2023). To achieve optimal drug effectiveness, it is crucial to consider physiological factors that influence drug absorption and distribution. In ruminants such as cattle and sheep, these factors play a key role in the success of drug delivery systems. Interactions between gastric acid, bile from the gallbladder, digestive enzymes from the pancreas, and relatively short retention times in the small intestine contribute to low microbial diversity in the midgut. By precisely analyzing these factors, as illustrated in Figure 2, the rumen conditions and associated challenges, advanced drug delivery systems can be optimized to achieve therapeutic objectives in ruminant animals more effectively (Hua and Lye, 2023). Formulation considerations Encapsulation protects vitamins and nutrients from degradation in the ruminant digestive system. Without encapsulation, rumen microorganisms can break down these nutrients before animals fully utilize them. Encapsulation involves coating nutrients with a protective layer that prevents rumen degradation. This ensures that nutrients reach the lower digestive tract where they can be absorbed and utilized by the animal (Mazinani et al., 2020; Besharati et al., 2022; Zabot et al., 2022). Encapsulation allows for the controlled release of nutrients, thereby enhancing efficiency (Ozturk and Temiz, 2018; Melo et al., 2021; Zabot et al., 2022). It also protects nutrients from environmental factors such as heat, light, and oxygen, during feed storage and processing, ensuring their stability until consumption by ruminants (Piñón-Balderrama et al., 2020; Amin et al., 2021; Zabot et al., 2022).
Figure 2. Physiological considerations for designing RDSs. The appropriate materials, especially polymers, are crucial for developing an RDS. The polymer must be safe and effective and must comply with regulatory requirements. Biocompatible and biodegradable polymers have advanced, offering safe materials with no adverse health effects. The polymer must be physiologically inert, non-absorbable, and unchanged during animal excretion. It should be nonmutagenic and should not cause harmful genetic changes. The polymer should not negatively impact long-term feeding, allowing for the safe and extended use of supplements (Galyon et al., 2023). Other essential factors include strength, solubility, permeability, stability, and cost. Organic materials such as polylactic acid, glycolic acid copolymers, and polypeptides are preferred for their ease of use, high encapsulation efficiency, and low toxicity (Wei et al., 2022). Inorganic materials such as double metal hydroxides, calcium carbonate, and silicates offer good chemical and thermal stability (Teixeira et al., 2021; Hamimed et al., 2022). The encapsulation process has progressed from single physical methods to chemical or combinations of both, enhancing efficiency and stability (Reis et al., 2022; Sousa et al., 2022). It is essential to maintain the activity of active ingredients and consider external factors such as touch, light, and pH, which can affect ingredient release. Research has focused on developing more efficient and cost-effective encapsulation techniques for drug delivery and active ingredients for ruminant digestion (Wei et al., 2022). The thermal stability of a polymer is essential for preventing damage during processing or storage at varying temperatures (Ur Rehman et al., 2020; Wang et al., 2022; Huang et al., 2023). The polymer must dissolve properly—not in rumen fluid but in abomasal fluid—to release active ingredients at the right time (MacHtakova et al., 2022). The coating efficiency depends on the solubility of the active ingredients, pellet size, and pellet surface smoothness (Hiew et al., 2019; Agrawal et al., 2022; Salawi, 2022). The development of pH-dependent coatings has improved the stability and effectiveness of RDSs (Albuquerque et al., 2020; Dijkstra et al., 2020). Consider these factors, as illustrated in Figure 3, and select the appropriate polymer to create a stable and effective RDS. Routes of administration considerations When designing a stable RDS, the route of drug administration is crucial. Different methods have benefits and limitations (McGrath et al., 2018; Jeong et al., 2020). Oral administration is commonly used, but it can vary in absorption and interaction with feed, which may affect bioavailability and efficacy (Mileva et al., 2023). In contrast, parenteral administration allows precise dosing with a faster onset but requires expertise in injection techniques, making it impractical for farm animals (Mileva et al., 2023). Topical and inhalation methods have specific purposes, particularly for the local treatment of respiratory problems (Amiri-Farahani et al., 2020; Windsor, 2022). Thus, designing an optimal RDS should ensure consistent and controlled release for sustained therapeutic effects. The design of the delivery system must also consider ease of use. The system should be user-friendly for farmers or ranchers and require minimal training and equipment (Kopper et al., 2023; Song et al., 2023). Methods such as boluses, implants, and rumen magnets have proven effective, but they require specialized training and consistent monitoring, increasing their complexity (Blakebrough-Hall et al., 2020; Neves et al., 2022). Recent innovations aim to simplify these systems, enhance ease of use, and encourage higher adoption rates. Furthermore, controlled shipping methods must be adapted to account for variations in animal size, weight, and health concerns. This includes ensuring animal comfort and welfare while maintaining the effectiveness of the drug delivery system. Modern biotechnological tools are increasingly used to create personalized delivery systems that consider each animal’s unique physiological and anatomical characteristics (Pech-Cervantes et al., 2020; Lobo and Faciola, 2021). This approach not only enhances treatment effectiveness but also improves animal welfare standards. Criteria for rumen-stable delivery systems RDSs are expected to be an alternative to traditional feed, providing medicine or nutrition to ruminants. However, creating a system that ensures sustainable release over long periods is a significant challenge. Differences in drug physiology and pharmacokinetics among species complicate the direct application of controlled-release technologies in livestock. The system must also protect the active ingredients from rumen fermentation and ensure their availability for absorption after passing through the rumen (Diez et al., 2022; Tajima et al., 2023).
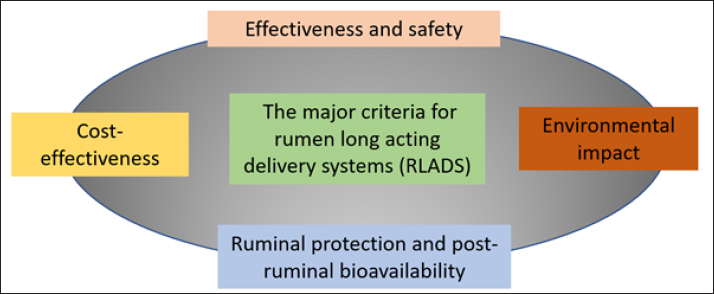
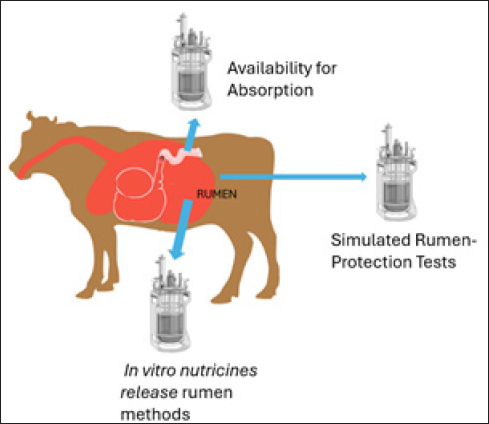
Figure 3. Formulation considerations for designing RDSs. Cost-effectiveness is another crucial factor. The costs of manufacturing, distributing, and administering RDS must be justified by the potential economic benefits, such as increased livestock productivity or reduced health expenses. It is essential to compare RDS with conventional methods, which might be cheaper but still provide similar or better benefits at a comparable or lower cost (García-Dios et al., 2020; Ungerfeld and Pitta, 2024). The environmental impact of RDSs is a critical consideration. Evaluations should include the effects of soil, water, air, and wildlife. Recent advancements aim to develop degradable systems that minimize environmental impact and support more sustainable farming practices. The major criteria considered in RDS planning, including environmental impact, are illustrated in Figure 4 (Smith et al., 2018; Neethirajan, 2024). Evaluation of rumens-table delivery systemsPhysical characterization Comprehensive characterization of polymeric delivery systems for rumen applications involves multiple analytical approaches. Modern nanoparticle characterization employs three primary technique categories. Light scattering methods (including dynamic light scattering, nanoparticle tracking analysis, and static light scattering) measure particle size, shape, and motion in systems. Electron microscopy techniques, namely scanning electron microscopy for surface imaging and transmission electron microscopy for internal structure analysis, provide high-resolution visualization at the nanoscale. Surface probing using atomic force microscopy studies particle–environment interactions. Additionally, combined microscopy–spectroscopy approaches enable simultaneous measurements of both physical and chemical properties (Caputo et al., 2019; Qiu et al., 2025). Chemical analysis through FTIR confirms polymer interactions and cross-linking, supported by XRD and DSC/TGA for structural properties (Mahmood et al., 2017). Nanoparticle formulations with zeta potentials beyond ±25 mV exhibit more excellent stability, as these charge levels prevent particle agglomeration and maintain dispersion by overcoming van der Waals attraction forces (Mahmood et al., 2017; Cottet et al., 2023). The physical characteristics of RDS include hardness, adhesiveness, drying time, flexibility, and elasticity. The hardness and adhesiveness of the coatings were measured using a texture analyzer. Drying time was visually evaluated by assessing uniformity, appearance, and peelability. Stickiness was assessed by applying low-pressure cotton wool. Mechanical properties such as flexibility and elasticity are determined by measuring the tensile strength and elongation at break. Microscopic techniques provide nanoscale information. Instrumental analyses, such as X-ray diffraction, calorimetry, spectroscopy, and nuclear magnetic resonance, may be performed for specific purposes (Tran and Tran, 2019). Protection against rumen environment In vitro and in situ rumen methods can be used to evaluate RDS stability in the rumen environment. These methods provide information on how rumen conditions, such as pH changes or specific microbial populations, affect the stability and release of the active ingredient (Chen et al., 2024, 2022).
Figure 4. The major criteria for designing a RDSs.
Figure 5. In vitro test for protection from rumen, release test, and absorption. The rumen of ruminants acts as a natural feed fermenter that contains diverse microorganisms such as bacteria, anaerobic fungi, archaea, protozoa, and viruses (Chen et al., 2021; Liu et al., 2023b; Chen et al., 2024). These microorganisms work together to ferment and break down nutrients, providing energy and VFAs for the host (Wei et al., 2022). Simulated rumen-protection tests are the tests that mimic the rumen environment and allow researchers to analyze key factors such as the release rate, duration of release, and stability of the active ingredient within RDS, as illustrated in Figure 5 (Wei et al., 2022; Silva et al., 2024). Rumen motility, which involves cyclic movements, including primary and secondary contractions, is crucial for ruminant nutrition. This significantly impacts nutrient degradation and can affect the functioning of RDS. For example, the frequency and duration of ruminal contractions typically increase around feeding, potentially influencing the effectiveness of RDS (Wang et al., 2023b). Microbial protein (MCP) synthesis in rumen depends on using ammonia-nitrogen (NH3-N). Efficient MCP production requires a balanced supply of nitrogen and energy. Rumen motility improves microbial colonization of substrates, which helps synchronize nitrogen and energy release and enhances the effectiveness of RDS in the rumen ( Li et al., 2019a). In vitro releaseIn vitro and in situ rumen methods can be used to evaluate RDS efficacy under actual rumen conditions. In vitro rumen methods use rumen fluid from a live animal to replicate rumen conditions in the laboratory. The fluid was incubated with RDS, and the degradation or release of the active ingredient was analyzed. In situ rumen methods involve placing a small bag with test material into the rumen of a live animal. After a specific time, the bag is removed, and its contents are analyzed to determine the degradation or release of the active ingredient (Gümüş et al., 2022; Guo et al., 2022). These methods can be used to assess several RDS-related factors, such as the release rate, duration of release, and active ingredient stability. By analyzing samples taken at various time points, researchers can measure the amount of the active ingredient released over time and its release rate (Gümüş et al., 2022; Guo et al., 2022). Absorption availability Simulated abomasal-release test to evaluate the effectiveness of RDS in delivering active ingredients to the small intestine. This test uses a buffer solution with a pH similar to the abomasum’s to mimic its environment. RDS was added to this solution and incubated to assess the active ingredient’s release rate, duration, and stability. By analyzing samples taken at various time points, researchers can measure the release rate and amount of active ingredients. This test helps determine whether the RDS can protect the active ingredient from rumen degradation and ensure its delivery to the small intestine, thereby improving animal health and productivity (Gümüş et al., 2022; Guo et al., 2022). Blood responses of ruminant animals The blood response of ruminant animals evaluates the delivery of active ingredients to the bloodstream. RDS is designed for slow, sustained release, leading to a prolonged blood response. Blood markers, such as glucose, insulin, and amino acids, are measured before and after RDS administration to assess the impacts of metabolism and nutrient utilization. For instance, RDS containing amino acids can improve protein utilization and milk production, as measured by markers such as branched-chain amino acids and milk protein yield. Similarly, related blood markers indicate that RDS supplementation with vitamins or minerals can boost health and productivity (Astuti et al., 2022; Kim et al., 2022). Supplemental enzymes improve metabolic processes by increasing apparent digestibility, optimizing the use of dietary proteins, and enhancing overall nutrient availability (Anil et al., 2022). The blood response to vaccination is shown in Figure 6, which illustrates the changes in blood parameters. Efficacy of rumen delivery systems The efficacy of RDS depends on the active ingredient or nutrient being delivered and its intended effect on the animal. Generally, RDS offers several benefits, including improved nutrient utilization, sustained release of active ingredients, protection from rumen degradation, reduced feed waste, and minimized environmental impacts. RDS enhances the utilization of proteins, amino acids, vitamins, and minerals, leading to better animal growth, productivity, and overall health (Hendawy et al., 2022; Ahmed et al., 2024). They can also deliver active ingredients over a longer period, decreasing the need for frequent administration and ensuring that these ingredients reach the small intestine for absorption (Loregian et al., 2023). By improving feed efficiency, RDS reduce feed waste and enhance the economic efficiency of animal production (Nakaishi and Takayabu, 2022; Nath et al., 2023). Overall, RDS contribute to animal health and welfare by providing essential nutrients and active ingredients in a controlled and sustained manner, making them valuable for improving animal production and reducing environmental impact (Cerbu et al., 2021; Jung et al., 2021).
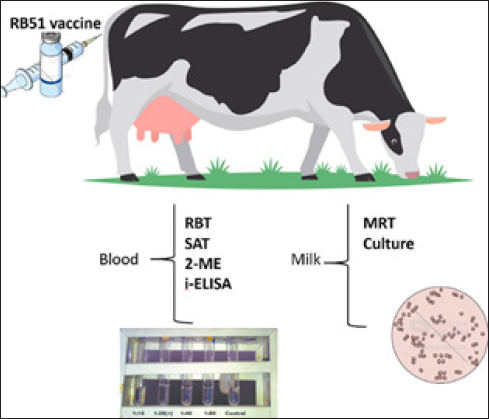
Figure 6. Sample of blood response in ruminant animals evaluation (Abnaroodheleh et al., 2023). Overall evaluation of the rumen delivery system Understanding rumen function requires knowledge of different feeds and nutritional assessment systems. In vitro gas production (IVGP) is a notable feed evaluation technique. It offers a less tedious and time-consuming alternative to in vivo digestibility measurements while showing high correlation with in vivo results. Artificial rumen models were compared with the rumen of dairy cows to assess their ability to support natural rumen microbiota and functions, including the production of VFAs and greenhouse gases (Shaw et al., 2023). Rumen fermentation parameters such as VFAs, pH, and total gas production are crucial for managing rumen ecology and microorganism growth (Budiman et al., 2024). These parameters help prevent rumen acidosis 12 hours after fermentation. During in vitro fermentation, rumen microbes break down complex nutrients such as carbohydrates, proteins, and organic polymers into monomers. These monomers ferment into VFAs, free ammonia (NH3), carbon dioxide (CO2), and hydrogen (H2). Methanogens, including Methanopyrales, Methanocellales, and Methanomicrobiales, then converted CO2 and H2 into methane (CH4) (Phupaboon et al., 2024). Other studies have shown that rumen fermentation can indicate reduced degradability rates and a lower microbial population (Yanza et al., 2021). In vitro/in vivo correlation (IVIVC) models are used to demonstrate the relationship between the in vitro release profile and in vivo performance of dosage forms, especially modified release drug products (Higgins-Gruber et al., 2013). IVIVC can be applied to all dosage forms and routes of administration. Developing effective IVIVC requires a well-designed, scientifically based approach (Tomic and Cardot, 2022). NutricinesNutricines, derived from “nutrition‟ and “medicine,” are substances incorporated into animal diets to enhance health, performance, and the production of agricultural products, such as milk, meat, fiber, and eggs (Górniak et al., 2018; Ferlisi et al., 2023). These include enzymes, which speed up chemical reactions in digestion; prebiotics, which are nondigestible ingredients that promote beneficial gut bacteria; probiotics, which are live microorganisms that confer health benefits; organic acids, which lower gut pH levels and improve nutrient absorption; plant extracts, which offer various health benefits; and trace elements, which are essential minerals for physiological functions. These substances, as shown in Figure. 7, collectively improve digestion, nutrient absorption, and overall animal health, ultimately resulting in better agricultural production productivity and efficiency (Tran and Tran, 2019; Garba and Firincioğlu, 2023). Nutricines are not essential for basic metabolism but can greatly enhance the well-being and performance of livestock by improving digestion, promoting better nutrient absorption, boosting immune function, and reducing disease susceptibility (Dell’anno et al., 2021; Dong et al., 2023; Pandey et al., 2023). EnzymesEnzymes secreted by microorganisms play crucial roles in plant degradation within the rumen ecosystem. Glycoside hydrolases break down plant biomass, with enzymes such as xylanase degrading β-1,4-xylan in hemicellulose and carboxymethyl cellulase targeting β-1,4-glucan in cellulose fibers. Lipases regulate fatty acid metabolism and control lipolysis, which limits the biohydrogenation of polyunsaturated fatty acids. Additionally, dehydrogenase, urease, and protease interact with protein and urea to supply essential nutrients to the host (Nunes and Kunamneni, 2018; Refat et al., 2021; Vittorazzi et al., 2021; Abid et al., 2023). Enzymes help break down complex dietary components such as proteins, fats, and carbohydrates, making nutrients more available for absorption (Saha and Pathak, 2021).
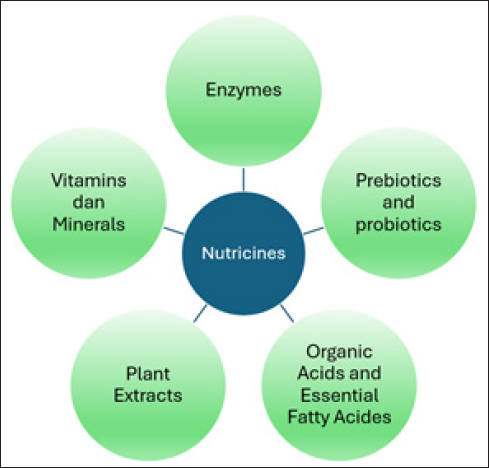
Figure 7. Nutricine components. Nanotechnology can deliver enzymes or digestive aids that improve feed digestion and nutrient utilization in the gastrointestinal tract of ruminants. These are live microorganisms that, when administered in adequate amounts, confer health benefits on the host by maintaining a healthy balance of gut microbiota (Agriopoulou et al., 2023; Gonzalez-bulnes and Hashem, 2023). Incorporating digestive enzymes such as amylase, protease, cellulase, xylanase, and beta-glucanase into bovine diets enhances growth performance. Exogenous enzymes increase the concentration of short-chain fatty acids in ruminal fluid, improve the proportion of unsaturated fatty acids, and decrease saturated fatty acids in meat. They also positively influence the oxidative stability of meat (Simon et al., 2024). Adding exogenous enzymes to animal feeds has significant potential to boost livestock productivity (Sridar, 2017). Prebiotics and probioticsProbiotic additives are beneficial, nondigestible food ingredients that selectively stimulate specific bacteria in the colon to improve host health. Encapsulation technology enhances the effectiveness, stability, and survival of probiotics during processing, storage, and gastrointestinal transit. Encapsulating probiotics in microspheres or microcapsules with materials such as alginate, chitosan, gelatin, plant mucilage, whey proteins, and polysaccharides protects probiotics from harsh conditions during fermentation. Chitosan nanoparticles can further protect probiotics, enhance their stability, and offer controlled release during fermentation (Agriopoulou et al., 2023). Phytobiotic additives, which are rich in secondary plant metabolites or phytochemicals, also support host health. Found in legume trees, medicinal plants, spices, and agricultural by-products, these phytobiotics can be combined with probiotics. The encapsulation of these combined additives ensures better stability, controlled release, and improved effectiveness in promoting a healthy gut microbiome (Ahmed et al., 2024). Organic acids and essential fatty acidsAlternative feed additives could offer nutritional strategies that help prevent metabolic disorders in ruminants by improving their metabolic and immune status. These additives lower the pH of the gut, which inhibits the growth of pathogenic bacteria and promotes digestion. Organic acids, such as citric acid and sorbic acid, and pure botanicals, such as thymol and vanillin, are widely used in animal nutrition because of their positive effects on production performance and their known impact on metabolic and immune status (Nkosi et al., 2021; Giorgino et al., 2023). Organic acids and essential oils are effective alternatives to antibiotic growth promoters in pig production due to their antibacterial, antiviral, and antioxidant properties (Nhara et al., 2024). Essential fatty acids, such as omega-3 and omega-6, play crucial roles in metabolic processes, including immune function and inflammation regulation. Nanoencapsulation protects these fatty acids from oxidation and enhances their delivery to animals (Tolve et al., 2021; Dumlu, 2024). Plant extractsPhytogenic extracts provide unique benefits as natural, abundant, renewable, and pollution-free sources with low-residue anti-inflammatory, antioxidant, and antimicrobial properties. They also stimulate appetite and enhance digestion (Piao et al., 2023; Orzuna-orzuna et al., 2024; Yang and Park, 2024). In addition to traditional vitamins and minerals, nanotechnology can deliver other bioactive compounds with potential health benefits, such as antioxidants, polyphenols, and probiotics. Nanoencapsulation protects these compounds from degradation and enhances their stability during storage and digestion (Pateiro et al., 2021; Andrade et al., 2024). Vitamins and mineralsAdequate mineral supplementation in small ruminants is crucial for proper physiological processes. Too little or too much supplementation can impair immune function, reproduction, and growth (Radke, 2021). The bioavailability of fat-soluble nutrients depends on their ability to form micelles (Borel and Desmarchelier, 2018; Šimoliūnas et al., 2019). Mineral bioavailability can be influenced by competition for binding sites and nutritional status, such as the effect of vitamin A on iron absorption. Chelation with dietary polyphenols can reduce iron uptake. Physiological factors such as gastric and intestinal secretions, mucosal cell regulation, and microflora also play a role. For instance, vitamin B12 absorption relies on gastric acid and intrinsic factor production (Brugger et al., 2022; Byrne and Murphy, 2022). Current delivery systems for rumen nutricine are limited (Garba and Firincioğlu, 2023). Enzymes are challenged by rapid proteolytic degradation, pH instability, and thermal sensitivity during feed processing while maintaining optimal concentrations remains difficult (Morgavi et al., 2000; Dijkstra et al., 2014; López-Trujillo et al., 2023). Prebiotics suffer from uncontrolled fermentation and nonspecific microbial stimulation, with the effectiveness of these interventions varying based on the existing microbiota. Probiotics are limited by poor survival during processing and storage, weak colonization in mature rumen, and frequent dosing requirements. The challenge is to establish ideal fermentative processes in which the maximal cell growth and biomass yield are in equilibrium with cell metabolism and stress tolerance (Mendonça et al., 2023; Rana et al., 2024). Organic acids are rapidly absorbed and neutralized (a rapidly fermentable substrate), requiring careful dosing to avoid pH disruption. However, their corrosive nature and short duration of action present practical challenges (Carro and Ungerfeld, 2015). Herbal extracts, despite their potential benefits, face issues with variable composition, inconsistent bioavailability, rapid degradation in the rumen, and possible interactions with other feed components (Alem, 2024; Subbiah et al., 2024; Wang et al., 2024). The former usually leads to inadequate dietary formulations that, in turn, cause unbalanced AA levels in the plasma (Albuquerque et al., 2023). These limitations highlight the need for improved delivery systems to enhance the efficacy of rumen nutricine. Different nutrients can work better together in the rumen. When enzymes and probiotics are combined, they collaborate to break down feed more effectively. The enzymes first break down complex plant materials, making it easier for beneficial bacteria to use them (Khademi et al., 2022; Mousa et al., 2022). Adding herbal extracts to enzymes can help protect the enzymes from breaking down too quickly in the rumen, while also providing extra benefits such as fighting harmful bacteria or acting as antioxidants (Yang and Park, 2024). When probiotics are paired with prebiotics (called synbiotics), the prebiotics act like a targeted food source, helping the good bacteria survive and thrive better in the rumen. Synbiotic formulation of 6 g FOS + L. plantarum CRD-7 in dairy calves improved digestibility, antioxidant enzymes, and immune status, as well as modulated the fecal microbiota and decreased diarrhea incidence (Sharma et al., 2023). Mixing herbal extracts with organic acids can help control harmful bacteria for longer periods while keeping the rumen pH stable (Ahmed et al., 2022; Okoye et al., 2023). Cinnamon extract can be used as an alternative antibiotic to monensin extract to control ruminal acidosis when corn is used as a basal diet (Ahmed et al., 2022). However, we need to be careful when mixing these ingredients: they need to be properly formulated to work well together and avoid any negative interactions that could make them less effective. In livestock, low levels of pasture micronutrients and gastrointestinal antagonisms can affect absorption. Direct supplementation helps prevent deficiencies, and animal excreta contributes micronutrients to the pasture. More research is needed to understand how feed and supplements affect micronutrient content in excreta and soil. Designing multispecies swards for optimal ruminant health requires understanding soil properties, forage types, and environmental conditions (Pinotti et al., 2020). Nuticine RumenS-table Delivery SystemsNuticine RDSsNanoengineering involves creating materials with unique properties using both organic and inorganic substances (Khan et al., 2019; Khalid et al., 2020). These materials enhance bioavailability, protect against gastrointestinal tract conditions, and enable controlled release. Key factors affecting nutritional value include particle size, physical state, and surface properties (Wang et al., 2023a; Altemimi et al., 2024). Coating materials used to protect core nutrients or feed from ruminal degradation should have specific properties:
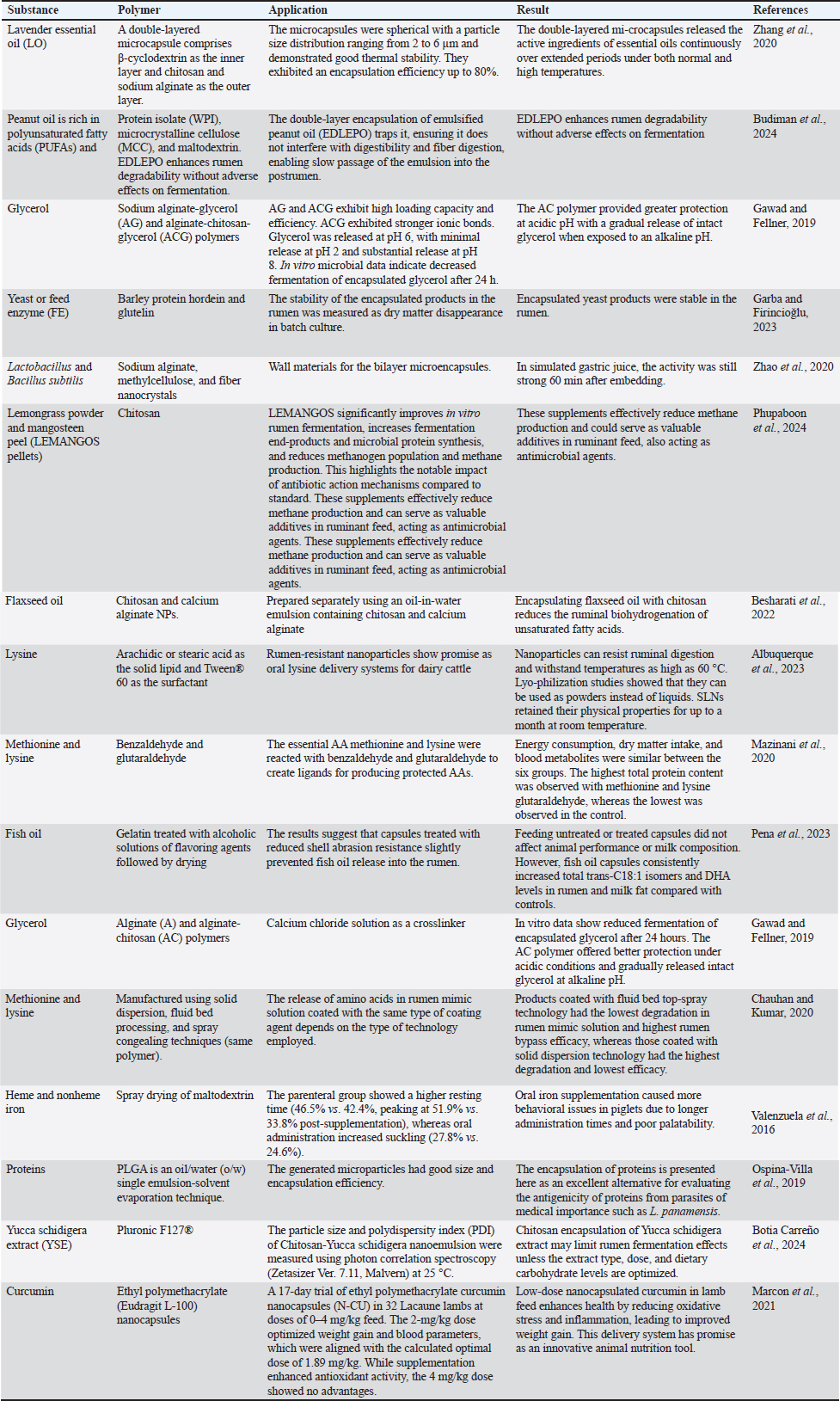
Encapsulated nanoparticles improve the delivery of vitamins D and E in supplements. Compared with high-dose salts, nanosized ZnO/Cu particles in piglet diets enhance growth and reduce environmental impact. Amino acid chelates improve mineral absorption, promoting animal performance and bone health while reducing trace mineral excretion (Upadhaya and Kim, 2020). Nutricines in RDS enhance rumen health, nutrient utilization, and animal performance. Careful formulation and testing are essential for ensuring stability, bioavailability, and efficacy. Antioxidant supplementation and methyl group status enhancement with vitamin E, selenium, and choline are recommended for stable metabolic health and optimized milk production (Pinotti et al., 2020). Table 1 illustrates the effectiveness of polymeric nutricine delivery systems. Specifically, the use of polymers in nutricine enhances the protection of active substances by ensuring stability within the rumen, regulates the controlled release of these substances, and mitigates environmental impacts such as methane emissions. The table further demonstrates the prevalent use of alginate and chitosan polymers in these systems. Alginate’s interaction with cationic compounds enhances its protective properties by improving its resistance to acidic pH and reducing porosity. Chitosan, known for its biocompatibility and gel-forming ability, is particularly suitable for targeted release applications. Modified chitosan particles are frequently employed as coatings in bioactive material delivery systems because of their controlled release. Chitosan dissolves at pH < 6 and can undergo polymerization through anionic cross-linking, which improves the survival of active substances, colon-targeted delivery, and thermal stability in applications across food, medicine, and agriculture (Sadeghi et al., 2024). Chitosan is an effective material for RDSs due to its distinctive properties. In the alkaline environment of the rumen, chitosan remains stable, preventing the premature release of encapsulated substances (Almassri et al., 2024). As the material progresses into the acidic abomasum, the chitosan dissolves, allowing for a controlled release of the nutrients. Additionally, chitosan is hydrolyzed by specific enzymes present in the abomasum, which enhances the targeted release of the encapsulated bioactives (Anil, 2022). Chitosan’s ability to form gels creates protective coatings around the nutrients, while its bioadhesive characteristics improve adherence to the intestinal walls, thereby increasing nutrient absorption. Moreover, chitosan’s biodegradable nature ensures a gradual breakdown over time without environmental accumulation, thereby optimizing the controlled release of nutrients throughout the digestive tract (Souza et al., 2020). Chauhan et al. revealed that the release of amino acids in rumen mimic solution, despite using the same coating agents, is significantly influenced by the type of technology used (Chauhan and Kumar, 2020). A review of polymeric materials for rumen-protected delivery systems reveals two primary categories: natural and synthetic polymers, each with distinct physicochemical properties and applications (Ghasemiyeh and Mohammadi-Samani, 2021; Zhou et al., 2024). Natural polymers exhibit specialized physicochemical properties: chitosan exhibits pH-dependent behavior with documented antimicrobial activity (pKa ≈ 6.5), whereas alginate facilitates gelation via multivalent cationic cross-linking mechanisms (Nasaj et al., 2024; Yilmaz Atay, 2019). Cellulose derivatives manifest thermally induced conformational changes, and zein proteins display pronounced hydrophobicity because of their nonpolar amino acid composition (Carvalho et al., 2021; Giteru et al., 2021; Liu et al., 2023b). Synthetic polymer systems demonstrate complementary characteristics: polymethacrylate (Eudragit) shows pH-dependent dissolution profiles correlating to specific functional group modifications (Patra et al., 2017; Nikam et al., 2023), poly(lactic-co-glycolic acid) undergoes hydrolytic degradation with tunable kinetics, and polyethylene glycol demonstrates enhanced colloidal stabilization through steric hindrance mechanisms (Masoudi et al., 2012; Zaaba and Jaafar, 2020; Lu et al., 2023). Multilayer coating systems, incorporating strategic combinations of these polymers, demonstrate enhanced rumen stability and controlled-release profiles. Examples include chitosan-alginate polyelectrolyte complexes, zein–pectin multilayers (Gawad and Fellner, 2019), and Eudragit-cellulose composite systems (Iffat et al., 2022). The selection of appropriate polymeric materials and coating architectures is governed by factors such as the physicochemical properties of the target nutrient, desired release kinetics, and environmental conditions. Economic viability and manufacturing scalability remain critical considerations for the commercial implementation of these delivery systems. Table 1. Encapsulation of nutrition with several polymers.
Scientists have developed novel synthetic polymers by enhancing existing materials: they have modified polyacrylate to be more sensitive to pH changes, created cross-linked methacrylic derivatives that release their contents in a controlled manner, and engineered biodegradable polyesters that break down at precise rates (Patra et al., 2017; Nikam et al., 2023). Researchers have successfully modified chitosan to improve its solubility (through carboxymethylation), enhanced alginate’s ability to stick to mucous membranes (via thiolation), created acetylated cellulose that breaks down in a controlled manner, and developed phosphorylated starches for targeted delivery (Herdiana et al., 2023). Exogenous fibrolytic enzymes enhance ruminant production and nutrient digestibility, although inconsistent results arise from a limited understanding of enzyme activity factors. These enzymes target the fibrous fraction of forage, which contain 30%–70% NDF, with digestibility typically below 65%. Despite high feed costs, specific enzyme supplementation improves nutritional value, digestion efficiency, and animal performance (Almassri et al., 2024). In many studies, L-tryptophan, L-ascorbic acid, niacin, and omega-3 fatty acids are provided in rumen-protected or coated forms to prevent degradation by rumen microbes (Ballard and Byrd, 2018; Chen et al., 2019). They are then incorporated into the feed (Wu et al., 2022). Phytochemicals are tested as feed additives for their potential as antioxidants, antimicrobials, immune stimulators, and modulators of rumen fermentation. They can improve metabolism, reduce antibiotic use, modulate appetite and digestion, and enhance immune, endocrine, and metabolic systems, leading to better efficiency, milk yield, and composition (Wu et al., 2022). Supplementation with bypass fat had no adverse effects on rumen fermentation, feed intake, digestibility of nutrients, or blood parameters of dairy animals. The milk yield is increased along with the improvement in postpartum recovery of the body weight, body condition score, and reproductive performance of the dairy animals (Wu et al., 2022). Commercial challenges in RDSsThe commercialization of RDS and other controlled-release drug delivery technologies in the veterinary field faces several challenges. The limited availability of new active pharmaceutical ingredients has restricted the development of novel RDS formulations. Financial constraints, such as limited research budgets and high development costs, hinder investment in new product development (Kipperman et al., 2022). The complexity of the animal environment necessitates a deep understanding of formulation science to create effective and safe RDS tailored to various animal populations. The aim is to minimize animal handling, reduce stress, and lower treatment costs through sustained and controlled drug release. Flexibility, ease of administration, and safety are key considerations when designing RDS for veterinary use (Lloyd, 2017). Safety, efficacy, and stability are crucial for the development of veterinary products. Veterinary settings have unique environmental conditions, such as temperature fluctuations and pathogen exposure, which require robust formulations that maintain stability and efficacy over time (Francis, 2020; Vidhamaly et al., 2022). Regulatory requirements for veterinary products, similar to those for human drugs, include adherence to good manufacturing practices (GMP), adding complexity to the development and commercialization process. Collaboration, research, and investment from industry, academia, and regulatory bodies are necessary to fully realize the potential of RDS in veterinary medicine (Michael et al., 2022; Garba and Firincioğlu, 2023). PerspectiveSmart farming technologies are being adopted to increase food production while minimizing environmental impacts. With the global population projected to reach 10 billion by 2050, the demand for animal products is also rising. However, livestock farming is complex, and production optimization, waste reduction, and cost reduction are essential (Monteiro et al., 2021). These challenges are further complicated by a shrinking workforce and rising production costs. Although meat provides essential nutrients, increasing production is challenging because of limited natural resources (Kumar et al., 2021). Advances in animal health biotechnology, including vaccines, antimicrobials, and diagnostic tools, have supported the growth of livestock systems (Siddiqui et al., 2022). Veterinary drug delivery systems offer benefits such as reduced dosage, minimized side effects, and reduced animal stress, thus increasing profitability (Li et al., 2022). Encapsulation is essential in ruminant nutrition, optimizing nutrient delivery through appropriate material selection and understanding factors affecting efficiency (Garba and Firincioğlu, 2023). Effective delivery systems for bioactive compounds improve stability, solubility, and targeted delivery, advancing ethnoveterinary medicine, using herbs, offering synthetic drug alternatives, and addressing antimicrobial overuse. Probiotics positively impact the gut microbiota, immune response, nutrient digestibility, absorption, animal growth, and meat quality, thereby improving overall health and productivity (Nwafor and Nwafor, 2022). The encapsulation of feed additives enhances voluntary feed intake and the overall welfare of ruminant livestock (Garba and Firincioğlu, 2023). Nanoparticles play a significant role by protecting encapsulated bioactive compounds from degradation by gastrointestinal digestion and cellular metabolism. They enable controlled release, enhance biodistribution, and target tissues affected by biological disturbances. Nanoparticles also protect the lysine content from the ruminal microbiota (Albuquerque et al., 2020). Controlled delivery systems offer safer and more effective treatment for chronic diseases than immediate-release drugs. They have prolonged effects, increase efficacy and safety, and improve ruminant nutrition and health. These systems optimize nutrient delivery, control important parameters, and protect high-value nutrients and drugs. Rumen-controlled delivery systems improve animal welfare and production efficiency. Future efforts will focus on optimizing rumen and postrumen function, developing low-cost formulations, and targeting high-value micronutrients and drugs. Cost constraints will shape future developments in this area (Al-Shawi et al., 2020). Ruminants need coarse ‟physically effective fiber” (peNDF) to stimulate chewing and ruminal activity. peNDF, defined by particle size, increases saliva flow, the acetate-to-propionate ratio, and milk fat levels, and maintains rumen pH (Belverdy et al., 2019). Fiber enhances chewing, salivation, rumination, and ruminal motility; alleviates rumen acidosis; regulates dietary intake; aids in milk fat synthesis; and promotes solid particle digestion. Evaluating and improving dietary fiber utilization are crucial for formulating ruminant diets (Zhou et al., 2022). Grass–legume mixtures can increase daily intake due to the preference of animals for mixed forage. Legumes with condensed tannins alter ruminal protein degradation by shifting nitrogen excretion from urine to feces, which is environmentally beneficial. Tannin-rich diets also reduce enteric methane emissions per unit of intake. These mixtures benefit animals, increase biomass yield, and reduce fertilizer use (Seoni et al., 2021; O.S. van Cleef et al., 2022). High-concentration diets fed to finishing beef cattle significantly affect the rumen microbial community. The metabolic activities of these microbial communities, associated with specific basal diets, explain variations in methane and short-chain fatty acid production in cattle. Longitudinal sampling showed that once the rumen microbial community adapts to a dietary change, it maintains a relatively stable state (Snelling et al., 2019). Mathematical modeling and fermentation prediction techniques can help design drug delivery systems and manipulate rumen fermentation (Teixé-Roig et al., 2023). To meet the increasing demand for large-scale in vitro meat production is being explored, leading to self-sufficiency (Kumar et al., 2021). Research has focused on obtaining bioactive compounds from microalgae, agrifood residues, and edible insects as alternative protein sources for functional foods (Teixé-Roig et al., 2023). Access to veterinary drugs for livestock has become a significant issue, often analyzed in terms of demand and farmer behavior. However, drug use also depends on structural factors that influence the drug supply chain and farmers’ access. Veterinary medicine is crucial to the animal-based food chain (van Herten and Meijboom, 2018; Jaime et al., 2022). Many challenges in veterinary drug administration stem from limitations in traditional methods and dosage forms, which lead to compliance issues among animals. These challenges are exacerbated by the lack of suitable dosage forms and control measures for drug release and timing (Unde et al., 2024). Biosafety concerns about the coating materials, their potential adverse effects or unwanted interactions with gut microbiota, and their scalability and cost-effectiveness should be addressed (Sadeghi et al., 2024). Plant extract is introduced as a green additive and is likely to share similar functions with synthetic additives to enhance the protection ability of the coating. Moreover, they are non-toxic, safe to use, abundant, and environmentally friendly (Wu et al., 2022). N. oceanica shows a strong potential to be used as a natural dietary source of eicosapentaenoic acid EPA to ruminants; nevertheless, further studies are needed to verify its protection in vivo whole microalga biomass is a natural rumen-protected source of EPA) for ruminants (Alves et al., 2018). Concerns have arisen regarding drug residues in food products due to their potential adverse health effects and regulatory implications. Residues refer to pharmaceutical compounds or their metabolites found in meat, fish, eggs, poultry, and ready-to-eat foods intended for human consumption (Pratiwi et al., 2023). Reducing methane emissions from ruminants is critical for mitigating the environmental impact of livestock farming. Encapsulation technology has been explored in dairy cows using encapsulated lipids in their diets to inhibit methanogenesis in the rumen. Research has shown a significant reduction in methane production without adverse effects on milk production or cow health (Garba and Firincioğlu, 2023). Encapsulation technology enhances ruminant nutrition and product quality through controlled nutrient delivery, improving stability and targeted release during digestion. This treatment boosts nutrient utilization, reduces wastage, and enhances animal performance. Protecting sensitive compounds such as vitamins and probiotics from degradation ensures optimal health and growth. In ruminant products, they improve taste, texture, and shelf life by masking unpalatable compounds and controlling flavor release, though challenges such as cost and regulatory hurdles remain. Continued research and development are crucial for maximizing the benefits of sustainable ruminant farming. ConclusionThis study shows that polymeric RDS is a promising method for efficiently delivering nutrients to ruminants. Polymeric coatings for the slow, postruminal release of bioactive substances can greatly improve ruminant health and productivity. The results indicate that polymeric coatings effectively protect nutricine in the rumen and ensure their release in the abomasum. This research highlights the prominent use of alginate and chitosan polymers in RDS. The evaluation of these systems using both in vitro and in vivo methods provides a thorough understanding of their performance and effectiveness. Future research should focus on optimizing these delivery systems and exploring new materials to further enhance their efficacy and application. The continued development of novel nutricine delivery solutions will advance animal health and benefit the agricultural industry. AcknowledgmentsThe authors would sincerely and gratefully acknowledge the Rector of Padjadjaran University for funding the Article Processing Charges (APC) for this manuscript. Conflicts of interestThe author declares no conflict of interest. FundingThe APC was funded by Padjadjaran University via the Directorate of Research and Community Engagement. Authors’ contributionsThe author is solely responsible for all aspects of this work, including conceptualization, methodology, analysis, and manuscript preparation. Data availabilityAll data are presented in the article. ReferencesAbid, K., Jabri, J., Yaich, H., Malek, A., Rekhis, J. and Kamoun, M. 2023. Improving the nutritional value and rumen fermentation characteristics of sesame seed coats through bioconversion approach using exogenous fibrolytic enzymes produced by Trichoderma longibrachiatum. Biomass Convers. Biorefin. 13, 14917–14925; doi:10.1007/s13399-022-03402-3 Abnaroodheleh, F., Emadi, A., Dashtipour, S., Jamil, T., Mousavi Khaneghah, A. and Dadar, M. 2023. Shedding rate of Brucella spp. in the milk of seropositive and seronegative dairy cattle. Heliyon 9, e15085; doi:10.1016/j.heliyon.2023.e15085 Agrawal, S., Fernandes, J., Shaikh, F. and Patel, V. 2022. Quality aspects in the development of pelletized dosage forms. Heliyon 8, e08956; doi:10.1016/j.heliyon.2022.e08956 Agriopoulou, S., Tarapoulouzi, M., Varzakas, T. and Jafari, S.M. 2023. Application of encapsulation strategies for probiotics: from individual loading to co-encapsulation. Microorganisms 11, 1–25; doi:10.3390/microorganisms11122896 Agus, A. and Widi, T.S.M. 2018. Current situation and future prospects for beef production in Thailand—a review. Asian-Australasian J. Anim. Sci. 31, 968–975; doi:10.5713/ajas.18.0201 Ahmed, M.G., Al-Sagheer, A.A., El-Zarkouny, S.Z. and Elwakeel, E.A. 2022. Potential of selected plant extracts to control severe subacute ruminal acidosis in vitro as compared with monensin. BMC Vet. Res. 18, 1–11; doi:10.1186/s12917-022-03457-4 Ahmed, M.G., Elwakeel, E.A., El-Zarkouny, S.Z. and Al-Sagheer, A.A. 2024. Environmental impact of phytobiotic additives on greenhouse gas emission reduction, rumen fermentation manipulation, and performance in ruminants: an updated review. Environ. Sci. Pollut. Res. 31, 37943–37962; doi:10.1007/s11356-024-33664-5 Akhigbe, I., Munir, K., Akinade, O., Akanbi, L. and Oyedele, L.O. 2021. Iot technologies for livestock management: a review of present status, opportunities, and future trends bernard. Big Data Cogn. Comput. 5, 10; doi:10.3390/bdcc5010010 Albuquerque, J., Casal, S., Páscoa, R.N.M. de J., Van Dorpe, I., Fonseca, A.J.M., Cabrita, A.R.J., Neves, A.R. and Reis, S. 2020. Applying nanotechnology to increase the rumen protection of amino acids in dairy cows. Sci. Rep. 10, 1–12; doi:10.1038/s41598-020-63793-z Albuquerque, J., Neves, A.R., Van Dorpe, I., Fonseca, A.J.M., Cabrita, A.R.J. and Reis, S. 2023. Production of rumen- and gastrointestinal-resistant nanoparticles to deliver lysine to dairy cows. Sci. Rep. 13, 1–14; doi:10.1038/s41598-023-43865-6 Alem, W.T. 2024. Effect of herbal extracts in animal nutrition as feed additives. Heliyon 10, e24973; doi:10.1016/j.heliyon.2024.e24973 Almassri, N., Trujillo, F.J. and Terefe, N.S. 2024. Microencapsulation technology for delivery of enzymes in ruminant feed. Front. Vet. Sci. 11, 1352375; doi:10.3389/fvets.2024.1352375 Al-Shawi, S.G., Dang, D.S., Yousif, A.Y., Al-Younis, Z.K., Najm, T.A. and Matarneh, S.K. 2020. The potential use of probiotics to improve animal health, efficiency, and meat quality: a review. Agriculture 10, 1–14; doi:10.3390/agriculture10100452 Altemimi, A.B., Farag, H.A.M., Salih, T.H., Awlqadr, F.H., Al-Manhel, A.J.A., Vieira, I.R.S. and Conte-Junior, C.A. 2024. Application of nanoparticles in human nutrition: a review. Nutrients 16, 1–20; doi:10.3390/nu16050636 Alves, S.P., Mendonça, S.H., Silva, J.L. and Bessa, R.J.B. 2018. Nannochloropsis oceanica, a novel natural source of rumen-protected eicosapentaenoic acid (EPA) for ruminants. Sci. Rep. 8, 2–11; doi:10.1038/s41598-018-28576-7 Amin, N., Tagliapietra, F., Arango, S., Guzzo, N. and Bailoni, L. 2021. Free and microencapsulated essential oils incubated in vitro: ruminal stability and fermentation parameters. Anim. Open Access J. MDPI 11, 10180; doi:10.3390/ani11010180 Amiri-Farahani, L., Sharifi-Heris, Z. and Mojab, F. 2020. The anti-inflammatory properties of the topical application of human milk in dermal and optical diseases. Evid.-Based Complement. Altern. Med. 2020, 4578153; doi:10.1155/2020/4578153 Andersen, T.O., Altshuler, I., Vera-Ponce de León, A., Walter, J.M., McGovern, E., Keogh, K., Martin, C., Bernard, L., Morgavi, D.P., Park, T., Li, Z., Jiang, Y., Firkins, J.L., Yu, Z., Hvidsten, T.R., Waters, S.M., Popova, M., Arntzen, M., Hagen, L.H. and Pope, P.B. 2023. Metabolic influence of core ciliates within the rumen microbiome. ISME J. 17, 1128–1140; doi:10.1038/s41396-023-01407-y Andrade, D., Maldonado-Bravo, F., Alburquerque, A., Pérez, C., Gamboa, A., Caro, N., Díaz-Dosque, M., Gotelland, M., Abugoch, L. and Tapia, C. 2024. Nanoencapsulation of Maqui (Aristotelia chilensis) extract in chitosan–tripolyphosphate and chenopodin-based systems. Antioxidants 13, 273; doi:10.3390/antiox13030273 Anil, S. 2022. Potential medical applications of chitooligosaccharides. Polymers 14, 3558; doi:10.3390/polym14173558 Anil., Yadav, S., Anand, V.M., Chouraddi, R., Yadav, S.K., Singh, A.K., Nair, P.M., Prabhakar., Tiwari, J. and Durge, A. 2022. A review on the role of exogenous fibrolytic enzymes in ruminant nutrition. Curr. J. Appl. Sci. Technol. 41, 45–58; doi:10.9734/cjast/2022/v41i363966 Arjun, S., Neha, P., Mohith Sai, S.R. and Ravi, L. 2023. Chapter 27—Microbial symbionts in ruminants. In Developments in applied microbiology and biotechnology. Ed., Dharumadurai, D.B.T.-M.S, Academic Press, pp. 493–509; doi:10.1016/B978-0-323-99334-0.00011-6 Astuti, A., Rochijan, Prasetyo Widyobroto, B. and Tri Noviandi, C. 2022. Nutrient status, hematological and blood metabolite profile of mid-lactating dairy cows during wet and dry seasons raised under Indo tropical environmental conditions. J. Anim. Behav. Biometeorol. 10, 1–6; doi:10.31893/jabb.22007 Balehegn, M., Duncan, A., Tolera, A., Ayantunde, A.A., Issa, S., Karimou, M., Zampaligré, N., André, K., Gnanda, I., Varijakshapanicker, P., Kebreab, E., Dubeux, J., Boote, K., Minta, M., Feyissa, F. and Adesogan, A.T. 2020. Improving adoption of technologies and interventions for increasing supply of quality livestock feed in low- and middle-income countries. Glob. Food Sec. 26, 100372; doi:10.1016/j.gfs.2020.100372 Ballard, M. and Byrd, A.T. 2018. Evaluation of a rumen protected omega 3 supplement for reproduction in dairy cows as determined in three large herd field trials. Open J. Anim. Sci. 8, 346–355; doi:10.4236/ojas.2018.83026 Belverdy, M.S., Alamouti, A.A. and Azizi, M.H. 2019. Microencapsulation in the ruminant feed industry. Dellait 1, 1. Available via https://dellait.com/dairyknowledgecenter/microencapsulation-in-the-ruminant-feed-industry/ (Accessed 04 July 2024) Besharati, M., Giannenas, I., Palangi, V., Ayasan, T., Noorian, F., Maggiolino, A. and Lorenzo, J.M. 2022. Chitosan/calcium–alginate encapsulated flaxseed oil on dairy cattle diet: in vitro fermentation and fatty acid biohydrogenation. Animals 12, 1400; doi:10.3390/ani12111400 Bešlo, D., Došlić, G., Agić, D., Rastija, V., Šperanda, M., Gantner, V. and Lučić, B. 2022. Polyphenols in ruminant nutrition and their effects on reproduction. Antioxidants 11, 1–22; doi:10.3390/antiox11050970 Bilhalva, A.F., Finger, I.S., Pereira, R.A., Corrêa, M.N. and Del Pino, F.A.B. 2018. Utilization of biodegradable polymers in veterinary science and routes of administration: a literature review. J. Appl. Anim. Res. 46, 643–649; doi:10.1080/09712119.2017.1378104 Bionaz, M., Vargas-Bello-Pérez, E. and Busato, S. 2020. Advances in fatty acids nutrition in dairy cows: from gut to cells and effects on performance. J. Anim. Sci. Biotechnol. 11, 110; doi:10.1186/s40104-020-00512-8 Blakebrough-Hall, C., Dona, A., D’occhio, M.J., McMeniman, J. and González, L.A. 2020. Diagnosis of bovine respiratory disease in feedlot cattle using blood (1)H NMR metabolomics. Sci. Rep. 10, 115; doi:10.1038/s41598-019-56809-w Borandeh, S., van Bochove, B., Teotia, A. and Seppälä, J. 2021. Polymeric drug delivery systems by additive manufacturing. Adv. Drug Deliv. Rev. 173, 349–373; doi:10.1016/j.addr.2021.03.022 Borel, P. and Desmarchelier, C. 2018. Bioavailability of fat-soluble vitamins and phytochemicals in humans: effects of genetic variation. Annu. Rev. Nutr. 38, 69–96; doi:10.1146/annurev-nutr-082117-051628 Botia Carreño, E.O., Alvarado, T.D., Diego Acosta, J.A., Ruiz, P.E.H., Elghandour, M.M.M.Y., Dada, O.A., Lackner, M. and Salem, A.Z.M. 2024. Influence of nano-encapsulated Yucca schidigera extract on ruminal anaerobic gases of methane, carbon monoxide, and hydrogen sulfide production of different carbohydrate-based diets. J. Agric. Food Res. 18, 101450; doi:10.1016/j.jafr.2024.101450 Broda, M., Yelle, D.J. and Serwańska-Leja, K. 2024. Biodegradable polymers in veterinary medicine—a review. Molecules 29, 1–30; doi:10.3390/molecules29040883 Brugger, D., Wagner, B., Windisch, W.M., Schenkel, H., Schulz, K., Südekum, K.-H., Berk, A., Pieper, R., Kowalczyk, J. and Spolders, M. 2022. Review: bioavailability of trace elements in farm animals: definition and practical considerations for improved assessment of efficacy and safety. Animal 16, 100598; doi:10.1016/j.animal.2022.100598 Budiman, A., Nurhadi, B., Supratman, H., Rahman, M.M., Yanza, Y.R., Hernaman, I. and 2024. The effects of encapsulation and double-layer emulsion of peanut oil on in vitro rumen degradability rates and fermentation profile in sheep. Indian J. Anim. Res. 1–7; doi:10.18805/ijar.bf-1761 Burrow, H. 2019. Strategies for increasing beef cattle production under dryland farming systems. Indones. Bull. Anim. Vet. Sci. 29, 161; doi:10.14334/wartazoa.v29i4.2452 Byrne, L. and Murphy, R.A. 2022. Relative bioavailability of trace minerals in production animal nutrition: a review. Anim. J. MDPI 12, 981; doi:10.3390/ani12151981 Michael, B., Canci, J. and Mekler, P. 2022. Value creation, valuation and business models in the pharmaceutical sector. In Quantitative models in life science business. Eds., Canci, J.K., Mekler, P. and Mu, G. Springer, pp: 3–16; doi:10.1007/978-3-031-11814-2_1 Caputo, F., Clogston, J., Calzolai, L., Rösslein, M. and Prina-Mello, A. 2019. Measuring particle size distribution of nanoparticle enabled medicinal products, the joint view of EUNCL and NCI-NCL. A step by step approach combining orthogonal measurements with increasing complexity. J. Control. Release 299, 31–43; doi:10.1016/j.jconrel.2019.02.030 Carro, M. and Ungerfeld, E. 2015. Utilization of organic acids to manipulate ruminal fermentation and improve ruminant productivity. In: Rumen microbiology: from evolution to revolution. Eds., Puniya, A., Singh, R. and Kamra, D. New Delhi: Springer.; doi:10.1007/978-81-322-2401-3_13 Carvalho, J.P.F., Silva, A.C.Q., Silvestre, A.J.D., Freire, C.S.R. and Vilela, C. 2021. Spherical cellulose micro and nanoparticles: a review of recent developments and applications. Nanomaterials 11, 744; doi:10.3390/nano11102744 Cerbu, C., Kah, M., White, J.C. and Astete, C.E. 2021. Fate of biodegradable engineered nanoparticles used in health perspective. Molecules 26, 523. Chauhan, S. and Kumar, S. 2020. In vitro thermostability and rumen dissolution evaluation of various rumen protected lysine and methionine products. Pharma. Innov. 9, 328–330. Chen, J., Yang, Z. and Dong, G. 2019. Niacin nutrition and rumen-protected niacin supplementation in dairy cows: an updated review. Br. J. Nutr. 122, 1103–1112; doi:10.1017/S0007114519002216 Chen, P., Li, Y., Shen, Y., Cao, Y., Li, Q., Wang, M., Liu, M., Wang, Z., Huo, Z., Ren, S., Gao, Y. and Li, J. 2022. Effect of dietary rumen-degradable starch to rumen-degradable protein ratio on in vitro rumen fermentation characteristics and microbial protein synthesis. Animals 12, 1–13; doi:10.3390/ani12192633 Chen, P., Li, Y., Wang, M., Shen, Y., Liu, M., Xu, H., Ma, N., Cao, Y., Li, Q., Abdelsattar, M.M., Wang, Z., Huo, Z., Ren, S., Hu, L., Liu, J., Gao, Y. and Li, J. 2024. Optimizing dietary rumen-degradable starch to rumen-degradable protein ratio improves lactation performance and nitrogen utilization efficiency in mid-lactating Holstein dairy cows. Front. Vet. Sci. 11, 1–13; doi:10.3389/fvets.2024.1330876 Chen, X., Su, X., Li, J., Yang, Y., Wang, P., Yan, F., Yao, J. and Wu, S. 2021. Real-time monitoring of ruminal microbiota reveals their roles in dairy goats during subacute ruminal acidosis. NPJ Biofilms Microb. 7, 1–14; doi:10.1038/s41522-021-00215-6 Cisek, A.A., Szymańska, E., Aleksandrzak-Piekarczyk, T. and Cukrowska, B. 2024. The role of methanogenic archaea in inflammatory bowel disease—a review. J. Pers. Med. 14, 196; doi:10.3390/jpm14020196 Cottet, J., Oshodi, J.O., Yebouet, J., Leang, A., Furst, A.L. and Buie, C.R. 2023. Zeta potential characterization using commercial microfluidic chips. Lab. Chip. 24, 234–243; doi:10.1039/d3lc00825h da Silva, C.F., Almeida, T., de Melo Barbosa, R., Cardoso, J.C., Morsink, M., Souto, E.B. and Severino, P. 2020. New trends in drug delivery systems for veterinary applications. Pharm. Nanotechnol. 9, 15–25; doi:10.2174/2211738508666200613214548 Davidson, B.D., Zambon, A.A., Guadagnin, A.R., Hoppmann, A., Larsen, G.A., Sherlock, D.N., Luchini, D., Apelo, S.I.A. and Laporta, J. 2024. Rumen-protected methionine supplementation during the transition period under artificially induced heat stress: impacts on cow-calf performance. J. Dairy Sci; doi:10.3168/jds.2024-24739 [Epub ahead of print]. Dell’anno, M., Reggi, S., Caprarulo, V., Hejna, M., Rossi, C.A.S., Callegari, M.L., Baldi, A. and Rossi, L. 2021. Evaluation of tannin extracts, leonardite and tributyrin supplementation on diarrhoea incidence and gut microbiota of weaned piglets. Animals 11, 693; doi:10.3390/ani11061693 Diao, Q., Zhang, R. and Fu, T. 2019. Review of strategies to promote rumen development in calves. Anim. J. MDPI 9, 490; doi:10.3390/ani9080490 Diez, R., Diez, M.J., Garcia, J.J., Rodríguez, J.M., Lopez, C., Fernandez, N., Sierra, M. and Sahagun, A.M. 2022. Improvement of albendazole bioavailability with menbutone administration in sheep. Animals 12, 1–10; doi:10.3390/ani12040463 Dijkstra, J., Reynolds, C.K., Kebreab, E. and Bannink, A. 2014. Energy and protein metabolism and nutrition in sustainable animal production. Energy Protein Metab. Nutr. Sustain. Anim. Prod; doi:10.3920/978-90-8686-781-3 Dijkstra, J., van Gastelen, S., Dieho, K., Nichols, K. and Bannink, A. 2020. Review: rumen sensors: data and interpretation for key rumen metabolic processes. Animal 14, s176–s186; doi:10.1017/S1751731119003112 Dong, L., Li, Y., Zhang, Yonghong, Zhang, Yan, Ren, J., Zheng, J., Diao, J., Ni, H., Yin, Y., Sun, R., Liang, F., Li, P., Zhou, C. and Yang, Y. 2023. Effects of organic zinc on production performance, meat quality, apparent nutrient digestibility and gut microbiota of broilers fed low-protein diets. Sci. Rep. 13, 1–14; doi:10.1038/s41598-023-37867-7 Dumlu, B. 2024. Importance of nano-sized feed additives in animal nutrition. J. Agric. Prod. 5, 55–72; doi:10.56430/japro.1433614 Džermeikaitė, K., Bačėninaitė, D. and Antanaitis, R. 2023. Innovations in cattle farming: application of innovative technologies and sensors in the diagnosis of diseases. Animals 13, 1–23; doi:10.3390/ani13050780 Edwards, J.E., Forster, R.J., Callaghan, T.M., Dollhofer, V., Dagar, S.S., Cheng, Y., Chang, J., Kittelmann, S., Fliegerova, K., Puniya, A.K., Henske, J.K., Gilmore, S.P., O’Malley, M.A., Griffith, G.W. and Smidt, H. 2017. PCR and omics based techniques to study the diversity, ecology and biology of anaerobic fungi: Insights, challenges and opportunities. Front. Microbiol. 8; doi:10.3389/fmicb.2017.01657 Elshahed, M.S., Hanafy, R.A., Cheng, Y., Dagar, S.S., Edwards, J.E., Flad, V., Fliegerová, K.O., Griffith, G.W., Kittelmann, S., Lebuhn, M., O’malley, M.A., Podmirseg, S.M., Solomon, K. V., Vinzelj, J., Young, D. and Youssef, N.H. 2022. Characterization and rank assignment criteria for the anaerobic fungi (Neocallimastigomycota). Int. J. Syst. Evol. Microbiol. 72, 1–7; doi:10.1099/ijsem.0.005449 Ferlisi, F., Tang, J., Cappelli, K. and Trabalza-Marinucci, M. 2023. Dietary supplementation with olive oil co-products rich in polyphenols: a novel nutraceutical approach in monogastric animal nutrition. Front. Vet. Sci. 10, 1–12; doi:10.3389/fvets.2023.1272274 Fleming, A.J., Estes, K.A., Choi, H., Barton, B.A., Zimmerman, C.A. and Hanigan, M.D. 2019. Assessing bioavailability of ruminally protected methionine and lysine prototypes. J. Dairy Sci. 102, 4014–4024; doi:10.3168/jds.2018-14667 Fonseca, N.V.B., Cardoso, A. da S., Bahia, A.S.R. de S., Messana, J.D., Vicente, E.F. and Reis, R.A. 2023. Additive tannins in ruminant nutrition: an alternative to achieve sustainability in animal production. Sustainability. 15, 4162; doi:10.3390/su15054162 Francis, M.J. 2020. A veterinary vaccine development process map to assist in the development of new vaccines. Vaccine 38, 4512–4515; doi:10.1016/j.vaccine.2020.05.007 Galyon, H., Vibostok, S., Duncan, J., Ferreira, G., Whittington, A. and Cockrum, R. 2023. Long-term in situ ruminal degradation of biodegradable polymers in Holstein dairy cattle. JDS Commun. 4, 70–74; doi:10.3168/jdsc.2022-0319 Galyon, H., Vibostok, S., Duncan, J., Ferreira, G., Whittington, A., Havens, K., McDevitt, J. and Cockrum, R. 2022. Digestibility kinetics of polyhydroxyalkanoate and Poly(butylene succinate-co-adipate) after in vitro fermentation in rumen fluid. Polymers 14, 2103; doi:10.3390/polym14102103 Garba, A.M. and Firincioğlu, S.Y. 2023. Role of encapsulation nutrients for improvement of ruminant performance and ruminant derived—products. Eurasian J. Agric. Res. 7, 109–126. García-Dios, D., Díaz, P., Viña, M., Remesar, S., Prieto, A., López-Lorenzo, G., Cao, J.M.D., Panadero, R., Díez-Baños, P. and López, C.M. 2020. Efficacy of oxyclozanide and closantel against rumen flukes (paramphistomidae) in naturally infected sheep. Animals 10, 1–9; doi:10.3390/ani10111943 Gawad, R. and Fellner, V. 2019. Evaluation of glycerol encapsulated with alginate and alginate-chitosan polymers in gut environment and its resistance to rumen microbial degradation. Asian-Austral. J. Anim. Sci. 32, 72–81; doi:10.5713/ajas.18.0110 Getabalew, M., Alemneh, T. and Bzuneh, E. 2020. Review on methanogenesis and its role. World J. Agric. Soil Sci. 6, 1–7; doi:10.33552/wjass.2020.06.000632 Gharechahi, J., Vahidi, M.F., Sharifi, G., Ariaeenejad, S., Ding, X.-Z., Han, J.-L. and Salekdeh, G.H. 2023. Lignocellulose degradation by rumen bacterial communities: new insights from metagenome analyses. Environ. Res. 229, 115925; doi:10.1016/j.envres.2023.115925 Ghasemiyeh, P. and Mohammadi-Samani, S. 2021. Polymers blending as release modulating tool in drug delivery. Front. Mater. 8, 1–12; doi:10.3389/fmats.2021.752813 Giorgino, A., Raspa, F., Valle, E., Bergero, D., Cavallini, D., Gariglio, M., Bongiorno, V., Bussone, G., Bergagna, S., Cimino, F., Dellepiane, L., Mancin, G., Paratte, R., Maza-Escolà, V.S. de la. and Forte, C. 2023. Effect of dietary organic acids and botanicals on metabolic status and milk parameters in mid-late lactating goats. Anim. J. MDPI 13, 797; doi:10.3390/ani13050797 Giteru, S.G., Ali, M.A. and Oey, I. 2021. Recent progress in understanding fundamental interactions and applications of zein. Food Hydrocoll. 120, 106948; doi:10.1016/j.foodhyd.2021.106948 Gonzalez-bulnes, A. and Hashem, N.M. 2023. Nanotechnology in animal science. Basel, Switzerland: MDPI; doi:10.3390/books978-3-0365-5946-9 Górniak, W., Cholewińska, P. and Konkol, D. 2018. Feed additives produced on the basis of organic forms of micronutrients as a means of biofortification of food of animal origin. J. Chem. 2018, 1–8; doi:10.1155/2018/8084127 Gümüş, H., Karakaş Oğuz, F., Oğuz, M.N., Buğdayci, K.E. and Dağli, H. 2022. Effects of replacing grain feed with rumen-protected fat on feedlot performance, ruminal parameters and blood metabolites in growing Merino lambs’ diets during the hot season. Ankara Univ. Vet. Fak. Derg. 69, 131–138; doi:10.33988/auvfd.856477 Guo, Y., Xiao, L., Jin, L., Yan, S., Niu, D. and Yang, W. 2022. Effect of commercial slow-release urea product on in vitro rumen fermentation and ruminal microbial community using RUSITEC technique. J. Anim. Sci. Biotechnol. 13, 1–14; doi:10.1186/s40104-022-00700-8 Halmemies-Beauchet-Filleau, A., Rinne, M., Lamminen, M., Mapato, C., Ampapon, T., Wanapat, M. and Vanhatalo, A. 2018. Review: alternative and novel feeds for ruminants: nutritive value, product quality and environmental aspects. Animal 12, S295–S309; doi:10.1017/S1751731118002252 Hamid, M.M.A., Moon, J., Yoo, D., Kim, H., Lee, Y.K., Song, J. and Seo, J. 2020. Rumen fermentation, methane production, and microbial composition following in vitro evaluation of red ginseng byproduct as a protein source. J. Anim. Sci. Technol. 62, 801–811; doi:10.5187/jast.2020.62.6.801 Hamimed, S., Jabberi, M. and Chatti, A. 2022. Nanotechnology in drug and gene delivery. Naunyn. Schmiedebergs. Arch. Pharmacol. 395, 769–787; doi:10.1007/s00210-022-02245-z Hanafy, R.A., Dagar, S.S., Griffith, G.W., Pratt, C.J., Youssef, N.H. and Elshahed, M.S. 2022. Taxonomy of the anaerobic gut fungi (Neocallimastigomycota): a review of classification criteria and description of current taxa. Int. J. Syst. Evol. Microbiol. 72, 1–38; doi:10.1099/ijsem.0.005322 Hayward, A., Bensel, T., Mazdiyasni, H., Rogner, J., Kirtane, A.R., Lee, Y.A.L., Hua, T., Bajpayee, A., Collins, J., McDonnell, S., Cleveland, C., Lopes, A., Wahane, A., Langer, R. and Traverso, G. 2018. Scalable gastric resident systems for veterinary application. Sci. Rep. 8, 1–10; doi:10.1038/s41598-018-30212-3 He, B., Fan, Y. and Wang, H. 2022. Lactate uptake in the rumen and its contributions to subacute rumen acidosis of goats induced by high-grain diets. Front. Vet. Sci. 9, 964027; doi:10.3389/fvets.2022.964027 Hendawy, A.O., Sugimura, S., Sato, K., Mansour, M.M., Abd El‐Aziz, A.H., Samir, H., Islam, M.A., Rubayet Bostami, A.B.M., Mandour, A.S., Elfadadny, A., Ragab, R.F., Abdelmageed, H.A. and Ali, A.M. 2022. Effects of selenium supplementation on rumen microbiota, rumen fermentation and apparent nutrient digestibility of ruminant animals: a review. Fermentation 8, 10004; doi:10.3390/fermentation8010004 Herdiana, Y., Wathoni, N., Gozali, D., Shamsuddin, S. and Muchtaridi, M. 2023. Chitosan-based nano-smart drug delivery system in breast cancer therapy. Pharmaceutics 15, 879; doi:10.3390/pharmaceutics15030879 Hiew, T.N., Tan, D.L.H., Tiang, Y.L. and Heng, P.W.S. 2019. Understanding the release performance of pellets with hydrophobic inclusions in sustained-release coating. Int. J. Pharm. 557, 229–237; doi:10.1016/j.ijpharm.2018.12.061 Higgins-Gruber, S., Rathbone, M.J. and Brumfield, J.C. 2013. In vitro drug release testing of veterinary pharmaceuticals BT—long acting animal health drug products: fundamentals and applications. In Long Acting Animal Health Drug Products. Eds., Rathbone, M.J. and McDowell, A. New York, NY: Springer, pp: 193–220; doi:10.1007/978-1-4614-4439-8_9 Hu, G., Jiang, H., Zong, Y., Datsomor, O., Kou, L., An, Y., Zhao, J. and Miao, L. 2022. Characterization of lactic acid-producing bacteria isolated from rumen: growth, acid and bile salt tolerance, and antimicrobial function. Fermentation 8, 385; doi:10.3390/fermentation8080385 Hu, R., Zou, H., Wang, Z., Cao, B., Peng, Q., Jing, X., Wang, Y., Shao, Y., Pei, Z., Zhang, X., Xue, B., Wang, L., Zhao, S., Zhou, Y. and Kong, X. 2019. Nutritional interventions improved rumen functions and promoted compensatory growth of growth-retarded yaks as revealed by integrated transcripts and microbiome analyses. Front. Microbiol. 10, 318; doi:10.3389/fmicb.2019.00318 Hua, D., Hendriks, W.H., Xiong, B. and Pellikaan, W.F. 2022. Starch and cellulose degradation in the rumen and applications of metagenomics on ruminal microorganisms. Anim. J. MDPI 12, 3020; doi:10.3390/ani12213020 Hua, S. and Lye, E.C. 2023. Impact of gastric and bowel surgery on gastrointestinal drug delivery. Drug Deliv. Transl. Res. 13, 37–53; doi:10.1007/s13346-022-01179-6 Huaiquipán, R., Quiñones, J., Díaz, R., Velásquez, C., Sepúlveda, G., Velázquez, L., Paz, E.A., Tapia, D., Cancino, D. and Sepúlveda, N. 2023. Review: effect of experimental diets on the microbiome of productive animals. Microorganisms 11, 2219; doi:10.3390/microorganisms11092219 Huang, Y., Stonehouse, A. and Abeykoon, C. 2023. Encapsulation methods for phase change materials—a critical review. Int. J. Heat Mass Transf. 200, 123458; doi:10.1016/j.ijheatmasstransfer.2022.123458 Iffat, W., Shoaib, M.H., Yousuf, R.I., Qazi, F., Mahmood, Z.A., Muhammad, I.N., Ahmed, K., Ahmed, F.R. and Imtiaz, M.S. 2022. Use of eudragit RS PO, HPMC K100M, ethyl cellulose, and their combination for controlling nicorandil release from the bilayer tablets with atorvastatin as an immediate-release layer. J. Pharm. Innov. 17, 429–448; doi:10.1007/s12247-020-09513-6 Iommelli, P., Zicarelli, F., Musco, N., Sarubbi, F., Grossi, M., Lotito, D., Lombardi, P., Infascelli, F. and Tudisco, R. 2022. Effect of cereals and legumes processing on in situ rumen protein degradability: a review. Fermentation 8, 1–16; doi:10.3390/fermentation8080363 Jaime, G., Hobeika, A., Figuié, M. 2022. Access to veterinary drugs in Sub-Saharan Africa: roadblocks and current solutions. Front. Vet. Sci. 8, 1–13; doi:10.3389/fvets.2021.558973 Jeong, S.H., Jang, J.H. and Lee, Y.B. 2020. Pharmacokinetic comparison of three different administration routes for topotecan hydrochloride in rats. Pharmaceuticals 13, 1–16; doi:10.3390/ph13090231 Jung, F., Thurn, M., Krollik, K., Gao, G.F., Hering, I., Eilebrecht, E., Emara, Y., Weiler, M., Günday-Türeli, N., Türeli, E., Parnham, M.J. and Wacker, M.G. 2021. Predicting the environmental emissions arising from conventional and nanotechnology-related pharmaceutical drug products. Environ. Res. 192, 110219; doi:10.1016/j.envres.2020.110219 Kappes, A., Tozooneyi, T., Shakil, G., Railey, A.F., McIntyre, K.M., Mayberry, D.E., Rushton, J., Pendell, D.L. and Marsh, T.L. 2023. Livestock health and disease economics: a scoping review of selected literature. Front. Vet. Sci. 10, 1168649; doi:10.3389/fvets.2023.1168649 Khademi, A.R., Hashemzadeh, F., Khorvash, M., Mahdavi, A.H., Pazoki, A. and Ghaffari, M.H. 2022. Use of exogenous fibrolytic enzymes and probiotic in finely ground starters to improve calf performance. Sci. Rep. 12, 1–14; doi:10.1038/s41598-022-16070-0 Khalid, K., Tan, X., Mohd Zaid, H.F., Tao, Y., Lye Chew, C., Chu, D.-T., Lam, M.K., Ho, Y.-C., Lim, J.W. and Chin Wei, L. 2020. Advanced in developmental organic and inorganic nanomaterial: a review. Bioengineered 11, 328–355; doi:10.1080/21655979.2020.1736240 Khan, I., Saeed, K. and Idrees, K. 2019. Nanoparticles : properties, applications and toxicities. Arab. J. Chem. 12, 908–931; doi:10.1016/j.arabjc.2017.05.011 Kim, S.H., Ramos, S.C., Valencia, R.A., Cho, Y. and Lee, S.S. 2022. Heat stress: effects on rumen microbes and host physiology, and strategies to alleviate the negative impacts on lactating dairy cows. Front. Microbiol. 13, 804562; doi:10.3389/fmicb.2022.804562 Kipperman, B., Block, G. and Forsgren, B. 2022. Economic issues. In Ethics in veterinary practice. Eds., Kipperman, B. and Rollin, B.E. Hoboken, NJ: Wiley, pp: 145–166; doi:10.1002/9781119791256.ch8 Kopper, G., Mirecki, S., Kljujev, I.S., Raicevic, V.B., Lalevic, B.T., Jovicic-Petrovic, J., Stojanovski, S. and Blazekovic-Dimovska, D. 2023. Chapter 27—Hygiene in primary production. In Food Safety Management. Eds., Andersen, V., Lelieveld, H. and Motarjemi, Y.B.T.-F.S.M. San Diego, CA: Academic Press, pp: 521–585; doi:10.1016/B978-0-12-820013-1.00013-9 Kumar, P., Sharma, N., Sharma, S., Mehta, N., Verma, A.K., Chemmalar, S. and Sazili, A.Q. 2021. In-vitro meat: a promising solution for sustainability of meat sector. J. Anim. Sci. Technol. 2021, e85; doi:10.5187/jast.2021.e85 Lee, M., Jeong, S., Seo, J. and Seo, S. 2019. Changes in the ruminal fermentation and bacterial community structure by a sudden change to a high-concentrate diet in Korean domestic ruminants. Asian-Austral. J. Anim. Sci. 32, 92–102; doi:10.5713/ajas.18.0262 Lei, Y., Zhang, K., Guo, M., Li, G., Li, C., Li, B., Yang, Y., Chen, Y. and Wang, X. 2018. Exploring the spatial-temporal microbiota of compound stomachs in a pre-weaned goat model. Front. Microbiol. 9, 1–11; doi:10.3389/fmicb.2018.01846 Li, L., Lee, C., Cruz, D.F., Krovi, S.A., Hudgens, M.G., Cottrell, M.L. and Johnson, L.M. 2022. Reservoir-style polymeric drug delivery systems: empirical and predictive models for implant design. Pharmaceuticals 15, 1226; doi:10.3390/ph15101226 Li, M.M., Titgemeyer, E.C. and Hanigan, M.D. 2019a. A revised representation of urea and ammonia nitrogen recycling and use in the Molly cow model. J. Dairy Sci. 102, 5109–5129; doi:10.3168/jds.2018-15947 Li, Z., Wang, X., Zhang, T., Si, H., Xu, C., Wright, A.-D.G. and Li, G. 2019b. Heterogeneous development of methanogens and the correlation with bacteria in the rumen and cecum of sika deer (Cervus nippon) during early life suggest different ecology relevance. BMC Microbiol. 19, 129; doi:10.1186/s12866-019-1504-9 Liu, C., Li, D., Chen, W., Li, Y., Wu, H., Meng, Q. and Zhou, Z. 2019. Estimating ruminal crude protein degradation from beef cattle feedstuff. Sci. Rep. 9, 11368; doi:10.1038/s41598-019-47768-3 Liu, G., An, D., Li, J. and Deng, S. 2023a. Zein-based nanoparticles: preparation, characterization, and pharmaceutical application. Front. Pharmacol. 14, 1–14; doi:10.3389/fphar.2023.1120251 Liu, X., Floate, K.D., Gorzelak, M.A., Holman, D.B., Hrycauk, S., Kubota, H., Lupwayi, N., Neilson, J.A.D., Ortega Polo, R., Petri, R.M., Tran, L., Wang, H., Wilches, D., Yang, X., Zorz, J. and Guarna, M.M. 2023b. Prairie agroecosystems: interconnected microbiomes of livestock, soil and insects. Agric. 13, 1–28; doi:10.3390/agriculture13020326 Lloyd, J.K.F. 2017. Minimising stress for patients in the veterinary hospital : why it is important and what can be done about it. Vet. Sci. 4, 1–19; doi:10.3390/vetsci4020022 Lobo, R.R. and Faciola, A.P. 2021. Ruminal phages—a review. Front. Microbiol. 12, 1–10; doi:10.3389/fmicb.2021.763416 López-Trujillo, J., Mellado-Bosque, M., Ascacio-Valdés, J.A., Prado-Barragán, L.A., Hernández-Herrera, J.A. and Aguilera-Carbó, A.F. 2023. Temperature and pH optimization for protease production fermented by Yarrowia lipolytica from agro-industrial waste. Fermentation 9, 1–15; doi:10.3390/fermentation9090819 Loregian, K.E., Pereira, D.A.B., Rigon, F., Magnani, E., Marcondes, M.I., Baumel, E.A., Branco, R.H., Del Bianco Benedeti, P. and Paula, E.M. 2023. Effect of tannin inclusion on the enhancement of rumen undegradable protein of different protein sources. Ruminants 3, 413–424; doi:10.3390/ruminants3040034 Lu, Y., Cheng, D., Niu, B., Wang, X., Wu, X. and Wang, A. 2023. Properties of poly (lactic-co-glycolic acid) and progress of poly (lactic-co-glycolic acid)-based biodegradable materials in biomedical research. Pharmaceuticals 16, 454; doi:10.3390/ph16030454 MacHtakova, M., Thérien-Aubin, H. and Landfester, K. 2022. Polymer nano-systems for the encapsulation and delivery of active biomacromolecular therapeutic agents. Chem. Soc. Rev. 51, 128–152; doi:10.1039/d1cs00686j Mahmood, S., Mandal, U.K., Chatterjee, B. and Taher, M. 2017. Advanced characterizations of nanoparticles for drug delivery: investigating their properties through the techniques used in their evaluations. Nanotechnol. Rev. 6, 355–372; doi:10.1515/ntrev-2016-0050 Malenica, D., Kass, M. and Bhat, R. 2023. Sustainable management and valorization of agri-food industrial wastes and by-products as animal feed: for ruminants, non-ruminants and as poultry feed. Sustainability 15, 10117; doi:10.3390/su15010117 Marcon, H., Griss, L.G., Molosse, V.L., Cecere, B.G.O., Alba, D.F., Leal, K.W., Galli, G.M., Souza, C.F., Baldissera, M.D., Gundel, S., de A Bassotto, V., Ourique, A.F., Vedovatto, M. and Da Silva, A.S. 2021. Dietary supplementation with curcumin-loaded nanocapsules in lambs: nanotechnology as a new tool for nutrition. Anim. Nutr. (Zhongguo xu mu shou yi xue hui) 7, 521–529; doi:10.1016/j.aninu.2020.06.014 Martínez-Ballesta, M., Gil-Izquierdo, Á., García-Viguera, C. and Domínguez-Perles, R. 2018. Nanoparticles and controlled delivery for bioactive compounds: outlining challenges for new “smart-foods” for health. Foods 7, 1–29; doi:10.3390/foods7050072 Masoudi, A., Madaah Hosseini, H.R., Shokrgozar, M.A., Ahmadi, R. and Oghabian, M.A. 2012. The effect of poly(ethylene glycol) coating on colloidal stability of superparamagnetic iron oxide nanoparticles as potential MRI contrast agent. Int. J. Pharm. 433, 129–141; doi:10.1016/j.ijpharm.2012.04.080 Mazinani, M., Naserian, A.A., Rude, B.J., Tahmasbi, A.M. and Valizadeh, R. 2020. Effects of feeding rumen-protected amino acids on the performance of feedlot calves. J. Adv. Vet. Anim. Res. 7, 229–233; doi:10.5455/javar.2020.g414 McGrath, J., Duval, S.M., Tamassia, L.F.M., Kindermann, M., Stemmler, R.T., de Gouvea, V.N., Acedo, T.S., Immig, I., Williams, S.N. and Celi, P. 2018. Nutritional strategies in ruminants: a lifetime approach. Res. Vet. Sci. 116, 28–39; doi:10.1016/j.rvsc.2017.09.011 Melo, M., da Silva, A., Filho, E.S., Oliveira, R., Junior, J.S., Oliveira, J.P., Vaz, A., Moura, J., Filho, J.P. and Bezerra, L. 2021. Polymeric microparticles of calcium pectinate containing urea for slow release in ruminant diet. Polymers (Basel). 13, 3776; doi:10.3390/polym13213776 Mendonça, A.A., Pinto-Neto, W. de P., da Paixão, G.A., Santos, D. da S., De Morais, M.A. and De Souza, R.B. 2023. Journey of the Probiotic Bacteria: Survival of the Fittest. Microorganisms 11, 10095; doi:10.3390/microorganisms11010095 Mileva, R., Petkova, T., Yaneva, Z. and Milanova, A. 2023. Investigation of the effect of pH on the adsorption-desorption of doxycycline in feed for small ruminants. Antibiot. 12, 268; doi:10.3390/antibiotics12020268 Mohamaden, W.I., Hegab, I.M., Hui, C. and Shang-li, S. 2020. In situ ruminal degradation kinetics and blood metabolites as affected by feeding different sources of tannin and flavonoids to small-tailed Han rams. Livest. Sci. 239, 104029; doi:10.1016/j.livsci.2020.104029 Monteiro, A., Santos, S. and Gonçalves, P. 2021. Precision agriculture for crop and livestock farming—brief review. Animals 11, 1–18; doi:10.3390/ani11082345 Morgavi, D., Newbold, C., Beever, D. and Wallace, J. 2000. Stability and stabilization of potential feed additive enzymes in rumen fluid*. Enzyme Microb. Technol. 26, 171–177; doi:10.1016/S0141-0229(99)00133-7 Mousa, G.A., Allak, M.A., Shehata, M.G., Hashem, N.M. and Hassan, O.G.A. 2022. Dietary supplementation with a combination of fibrolytic enzymes and probiotics improves digestibility, growth performance, blood metabolites, and economics of fattening lambs. Animals 12, 476; doi:10.3390/ani12040476 Nakaishi, T. and Takayabu, H. 2022. Production efficiency of animal feed obtained from food waste in Japan. Environ. Sci. Pollut. Res. 29, 61187–61203; doi:10.1007/s11356-022-20221-1 Nasaj, M., Chehelgerdi, M., Asghari, B., Ahmadieh-Yazdi, A., Asgari, M., Kabiri-Samani, S., Sharifi, E. and Arabestani, M. 2024. Factors influencing the antimicrobial mechanism of chitosan action and its derivatives: a review. Int. J. Biol. Macromol. 277, 134321; doi:10.1016/j.ijbiomac.2024.134321 Nath, P.C., Ojha, A., Debnath, S., Sharma, M., Nayak, P.K., Sridhar, K. and Inbaraj, B.S. 2023. Valorization of food waste as animal feed: a step toward sustainable food waste management and circular bioeconomy. Animals 13, 1366; doi:10.3390/ani13081366 Neethirajan, S. 2024. Innovative strategies for sustainable dairy farming in canada amidst climate change. Sustainability 16, 10265; doi:10.3390/su16010265 Neves, S.F., Silva, M.C.F., Miranda, J.M., Stilwell, G. and Cortez, P.P. 2022. Predictive models of dairy cow thermal state: a review from a technological perspective. Vet. Sci. 9, 416; doi:10.3390/vetsci9080416 Nhara, R.B., Marume, U. and Nantapo, C.W.T. 2024. Potential of organic acids, essential oils and their blends in pig diets as alternatives to antibiotic growth promoters. Animals 14, 762; doi:10.3390/ani14050762 Nikam, A., Sahoo, P.R., Musale, S., Pagar, R.R., Paiva-Santos, A.C. and Giram, P.S. 2023. A systematic overview of Eudragit® based copolymer for smart healthcare. Pharmaceutics 15, 587; doi:10.3390/pharmaceutics15020587 Nkosi, D.V., Bekker, J.L. and Hoffman, L.C. 2021. The use of organic acids (lactic and acetic) as a microbial decontaminant during the slaughter of meat animal species: a review. Foods 10, 2293; doi:10.3390/foods10102293 Nunes, C.S. and Kunamneni, A. 2018. Laccases—properties and applications. In Enzymes in Human and Animal Nutrition. Eds., Nunes, C.S. and Kumar, V.B.T.-E. San Diego, CA: Academic Press, pp. 133–161; doi:10.1016/B978-0-12-805419-2.00007-1 Nwafor, I.C. and Nwafor, C.U. 2022. African smallholder farmers and the treatment of livestock diseases using ethnoveterinary medicine: a commentary. Pastoralism 12, 10–13; doi:10.1186/s13570-022-00244-6 O.S. van Cleef, F., José, J.C., M. Ciriaco, F., Henry, D.D., Ruiz-Moreno, M., M. Jaramillo, D., Garcia, L., Erick, E.R., DiLorenzo, N., João, J.M., Naumann, H.D. and Sollenberger, L.E. 2022. Inclusion of a tannin-rich legume in the diet of beef steers reduces greenhouse gas emissions from their excreta. Sci. Rep. 12, 1–11; doi:10.1038/s41598-022-18523-y Okoye, C.O., Wang, Y., Gao, L., Wu, Y., Li, X., Sun, J. and Jiang, J. 2023. The performance of lactic acid bacteria in silage production: a review of modern biotechnology for silage improvement. Microbiol. Res. 266, 127212; doi:10.1016/j.micres.2022.127212 Orzuna-orzuna, J.F., Godina, E.J., Martínez, J.G. and Lara-Bueno, A. 2024. Capsaicin as a Dietary Additive for Dairy Cows. Animals 14(7), 1075. Ospina-Villa, J.D., Gómez-Hoyos, C., Zuluaga-Gallego, R. and Triana-Chávez, O. 2019. Encapsulation of proteins from Leishmania panamensis into PLGA particles by a single emulsion-solvent evaporation method. J. Microbiol. Methods 162, 1–7; doi:10.1016/j.mimet.2019.05.004 Ozturk, E. and Temiz, U. 2018. Encapsulation methods and use in animal nutrition. Selcuk J. Agric. Food Sci. 32, 624–631; doi:10.15316/SJAFS.2018.145 Palevich, N., Kelly, W.J., Ganesh, S., Rakonjac, J. and Attwood, G.T. 2019. Butyrivibrio hungatei MB2003 competes effectively for soluble sugars released by butyrivibrio proteoclasticus B316(T) during growth on xylan or pectin. Appl. Environ. Microbiol. 85, 18; doi:10.1128/AEM.02056-18 Pandey, S., Kim, E.S., Cho, J.H., Song, M., Doo, H., Kim, S., Keum, G.B., Kwak, J., Ryu, S., Choi, Y., Kang, J., Choe, J. and Kim, H.B. 2023. Cutting-edge knowledge on the roles of phytobiotics and their proposed modes of action in swine. Front. Vet. Sci. 10, 1–9; doi:10.3389/fvets.2023.1265689 Pateiro, M., Gómez, B., Munekata, P.E.S., Barba, F.J., Putnik, P., Kovačević, D.B. and Lorenzo, J.M. 2021. Nanoencapsulation of promising bioactive compounds to improve their absorption, stability, functionality and the appearance of the final food products. Molecules 26, 1547; doi:10.3390/molecules26061547 Patra, C.N., Priya, R., Swain, S., Kumar Jena, G., Panigrahi, K.C. and Ghose, D. 2017. Pharmaceutical significance of Eudragit: a review. Futur. J. Pharm. Sci. 3, 33–45; doi:10.1016/j.fjps.2017.02.001 Pech-Cervantes, A.A., Irfan, M., Estrada-Reyes, Z.M. and Ogunade, I.M. 2020. Recombinant technologies to improve ruminant production systems: the past, present and future. Processes 8, 1–28; doi:10.3390/pr8121633 Pena, O.M., Murphy, K., Long, N., Lascano, G.J., Jenkins, T.C. and Aguerre, M.J. 2023. Evaluating the rumen degradation of novel protected gelatin capsules containing fish oil fed to lactating dairy cows. Animal 13, 555; doi:10.3390/ani13162555 Perez, H.G., Stevenson, C.K., Lourenco, J.M. and Callaway, T.R. 2024. Understanding rumen microbiology: an overview. Encyclopedia 4, 148–157; doi:10.3390/encyclopedia4010013 Phupaboon, S., Matra, M., Sommai, S., Dagaew, G., Suriyapha, C., Prachumchai, R. and Wanapat, M. 2024. Microencapsulation efficiency of fruit peel phytonutrient-based antimicrobial to mitigate rumen emission using in vitro fermentation technique. Ital. J. Anim. Sci. 23, 664–677; doi:10.1080/1828051X.2024.2337688 Piao, M., Tu, Y., Zhang, N., Diao, Q. and Bi, Y. 2023. Advances in the application of phytogenic extracts as antioxidants and their potential mechanisms in ruminants. Antioxidants 12, 879; doi:10.3390/antiox12040879 Piñón-Balderrama, C.I., Leyva-Porras, C., Terán-Figueroa, Y., Espinosa-Solís, V., Álvarez-Salas, C. and Saavedra-Leos, M.Z. 2020. Encapsulation of active ingredients in food industry by spray-drying and nano spray-drying technologies. Processes 8, 889; doi:10.3390/PR8080889 Pinotti, L., Manoni, M., Fumagalli, F., Rovere, N., Tretola, M. and Baldi, A. 2020. The role of micronutrients in high-yielding dairy ruminants: choline and vitamin E. Ankara Univ. Vet. Fak. Derg. 67, 209–214; doi:10.33988/auvfd.695432 Pokhrel, B. and Jiang, H. 2024. Postnatal growth and development of the rumen: integrating physiological and molecular insights. Biology 13, 269; doi:10.3390/biology13040269 Pratiwi, R., Ramadhanti, S.P., Amatulloh, A., Megantara, S. and Subra, L. 2023. Recent advances in the determination of veterinary drug residues in food. Foods 12, 1–25; doi:10.3390/foods12183422 Qiu, Y., Dong, Z. and Su, L. 2025. Chapter 16 - Observation and physical characterization of nanoparticles. In Analysis of Microplastics and Nanoplastics. Eds., Shi, H. and Sun, C.B.T.-A. Amsterdam: Elsevier, pp: 295–314; doi:10.1016/B978-0-443-15779-0.00013-4 Radke, S. 2021. Small ruminant vitamins and minerals. Aabp Proc. 54, 32–34. Ramos, S.C., Jeong, C.D., Mamuad, L.L., Kim, S.H., Kang, S.H., Kim, E.T., Cho, Y., Lee, S.S. and Lee, S.S. 2021. Diet transition from high-forage to high-concentrate alters rumen bacterial community composition, epithelial transcriptomes and ruminal fermentation parameters in dairy cows. Animal 11, 838; doi:10.3390/ani11030838 Rana, A., Taneja, N.K., Raposo, A., Alarifi, S.N., Teixeira-Lemos, E., Lima, M.J., Gonçalves, J.C. and Dhewa, T. 2024. Exploring prebiotic properties and its probiotic potential of new formulations of soy milk-derived beverages. Front. Microbiol. 15, 1–13; doi:10.3389/fmicb.2024.1404907 Refat, B., Christensen, D.A., Ismael, A., Feng, X., Rodríguez-Espinosa, M.E., Guevara-Oquendo, V.H., Yang, J., Alzahal, O. and Yu, P. 2021. Evaluating the effects of fibrolytic enzymes on rumen fermentation, omasal nutrient flow, and production performance in dairy cows during early lactation. Can. J. Anim. Sci. 102, 39–49; doi:10.1139/cjas-2020-0062 Reis, D.R., Ambrosi, A. and Luccio, M. Di, 2022. Encapsulated essential oils: a perspective in food preservation. Futur. Foods 5, 100126; doi:10.1016/j.fufo.2022.100126 Sadeghi, A., Ebrahimi, M., Shahryari, S., Assadpour, E. and Jafari, S.M. 2024. Potential applications of encapsulated yeasts especially within alginate and chitosan as smart bioreactors and intelligent micro-machines. Carbohydr. Polym. Technol. Appl. 7, 100513; doi:10.1016/j.carpta.2024.100513 Saha, S.K. and Pathak, N.N. 2021. Digestion, absorption and metabolism of nutrients BT . In Fundamentals of Animal Nutrition. Eds., Saha, S.K. and Pathak, N.N. Singapore: Springer Singapore, pp: 219–246; doi:10.1007/978-981-15-9125-9_14 Salawi, A. 2022. Pharmaceutical coating and its different approaches, a review. Polymers 14, 318; doi:10.3390/polym14163318 Sanjorjo, R.A., Tseten, T., Kang, M.K., Kwon, M. and Kim, S.W. 2023. In pursuit of understanding the rumen microbiome. Fermentation 9, 1–19; doi:10.3390/fermentation9020114 Seoni, E., Rothacher, M., Arrigo, Y., Ampuero Kragten, S., Bee, G. and Dohme-Meier, F. 2021. The fate of tannins from birdsfoot trefoil and their effect on the nitrogen balance in growing lambs fed diets varying in protein level. Animal 11, 190; doi:10.3390/ani11010190 Sharma, A.N., Chaudhary, P., Kumar, S., Grover, C.R. and Mondal, G. 2023. Effect of synbiotics on growth performance, gut health, and immunity status in pre-ruminant buffalo calves. Sci. Rep. 13, 1–12; doi:10.1038/s41598-023-37002-6 Shaw, C.A., Park, Y., Gonzalez, M., Duong, R.A., Pandey, P.K., Brooke, C.G. and Hess, M. 2023. A comparison of three artificial rumen systems for rumen microbiome modeling. Fermentation 9, 1–21; doi:10.3390/fermentation9110953 Siddiqui, S.A., Bahmid, N.A., Taha, A., Abdel-Moneim, A.M.E., Shehata, A.M., Tan, C., Kharazmi, M.S., Li, Y., Assadpour, E., Castro-Muñoz, R. and Jafari, S.M. 2022. Bioactive-loaded nanodelivery systems for the feed and drugs of livestock; purposes, techniques and applications. Adv. Colloid Interface Sci. 308, 102772; doi:10.1016/j.cis.2022.102772 Silva, É.B.R. da, Silva, J.A.R. da, Silva, W.C. da, Belo, T.S., Sousa, C.E.L., Santos, M.R.P. dos, Neves, K.A.L., Rodrigues, T.C.G. de C., Camargo-Júnior, R.N.C. and Lourenço-Júnior, J. de B. 2024. A review of the rumen microbiota and the different molecular techniques used to identify microorganisms found in the rumen fluid of ruminants. Animals 14, 448; doi:10.3390/ani14101448 Šimoliūnas, E., Rinkūnaitė, I., Bukelskienė, Ž. and Bukelskienė, V. 2019. Bioavailability of different vitamin D oral supplements in laboratory animal model. Med. 55, 1–7; doi:10.3390/medicina55060265 Simon, A.L., Copetti, P.M., Lago, R.V.P., Vitt, M.G., Nascimento, A.L., Silva, L.E.L. e, Wagner, R., Klein, B., Martins, C.S., Kozloski, G. V., Da Silva, A.S. 2024. Inclusion of exogenous enzymes in feedlot cattle diets: Impacts on physiology, rumen fermentation, digestibility and fatty acid profile in rumen and meat. Biotechnol. Rep. 41, e00824; doi:10.1016/j.btre.2023.e00824 Smith, S.B., Gotoh, T. and Greenwood, P.L. 2018. Current situation and future prospects for global beef production: overview of special issue. Asian-Australasian J. Anim. Sci. 31, 927–932; doi:10.5713/ajas.18.0405 Snelling, T.J., Auffret, M.D., Duthie, C.-A., Stewart, R.D., Watson, M., Dewhurst, R.J., Roehe, R. and Walker, A.W. 2019. Temporal stability of the rumen microbiota in beef cattle, and response to diet and supplements. Anim. microbiome 1, 16; doi:10.1186/s42523-019-0018-y Soltis, M.P., Moorey, S.E., Egert-McLean, A.M., Voy, B.H., Shepherd, E.A. and Myer, P.R. 2023. Rumen biogeographical regions and microbiome variation. Microorganisms 11, 747; doi:10.3390/microorganisms11030747 Song, Y., Day, C.M., Afinjuomo, F., Tan, J.-Q.E., Page, S.W. and Garg, S. 2023. Advanced strategies of drug delivery via oral, topical, and parenteral administration routes: where do equine medications stand? Pharmaceutics 15, 186; doi:10.3390/pharmaceutics15010186 Sousa, V.I., Parente, J.F., Marques, J.F., Forte, M.A. and Tavares, C.J. 2022. Microencapsulation of essential oils: a review. Polymers 14, 1730; doi:10.3390/polym14091730 Souza, M.P.C. de, Sábio, R.M., Ribeiro, T. de C., Santos, A.M. Dos, Meneguin, A.B. and Chorilli, M. 2020. Highlighting the impact of chitosan on the development of gastroretentive drug delivery systems. Int. J. Biol. Macromol. 159, 804–822; doi:10.1016/j.ijbiomac.2020.05.104 Sprinkle, J.E., Schafer, D.W., Cuneo, S.P., Tolleson, D.R. and Enns, R.M. 2021. Effects of a long-acting trace mineral rumen bolus upon range cow productivity. Transl. Anim. Sci. 5, txaa232; doi:10.1093/tas/txaa232 Sridar, M. 2017. Scope of exogenous enzymes in enhancing ruminant productivity. J. Dairy, Vet. Anim. Res. 5, 67–72; doi:10.15406/jdvar.2017.05.00137 Suarjana, I.G.K., Pg, K.T. and Sudipa, P.H. 2021. Characteristics of rumen fluid, pH and number of microbia. J. Vet. Anim. Sci. 4, 6–10. Subbiah, V., Ebrahimi, F., Agar, O.T., Dunshea, F.R., Barrow, C.J. and Suleria, H.A.R. 2024. In vitro digestion and colonic fermentation of phenolic compounds and their antioxidant potential in Australian beach-cast seaweeds. Sci. Rep. 14, 1–14; doi:10.1038/s41598-024-54312-5 Tajima, T., Saiga, M., Yamamoto, H., Elbadawy, M., Abugomaa, A., Miura, R., Usui, T., Sasaki, K. and Shimoda, M. 2023. Oral pharmacokinetics of sulfadiazine and sulfamonomethoxine in female Holstein milking cows. J. Vet. Med. Sci. 85, 715–720; doi:10.1292/jvms.23-0110 Teixeira, S., Eblagon, K.M. and Figueiredo, J.L. 2021. Towards controlled degradation of poly(lactic) acid in technical applications. J. Carbon Res. 7, 1–43; doi:10.1007/978-0-8176-4829-9_1 Teixé-Roig, J., Oms-Oliu, G., Odriozola-Serrano, I. and Martín-Belloso, O. 2023. Emulsion-based delivery systems to enhance the functionality of bioactive compounds : towards the use of ingredients from natural, sustainable sources. Foods 12, 1502; doi:10.3390/foods12071502 Tolve, R., Tchuenbou-Magaia, F., Di Cairano, M., Caruso, M.C., Scarpa, T. and Galgano, F. 2021. Encapsulation of bioactive compounds for the formulation of functional animal feeds: The biofortification of derivate foods. Anim. Feed Sci. Technol. 279, 115036; doi:10.1016/j.anifeedsci.2021.115036 Tomic, I. and Cardot, J.-M. 2022. In vitro–in vivo correlations for modified release formulations. In Oral drug delivery for modified release formulations. Eds., Kostewicz, E.S., Vertzoni, M., Benson, H.A.E. and Roberts, M.S. Hoboken, NJ: Wiley, pp. 341–354; doi:10.1002/9781119772729.ch19 Tran, T.T.D. and Tran, P.H.L. 2019. Controlled release film forming systems in drug delivery: The potential for efficient drug delivery. Pharmaceutics 11, 1–16; doi:10.3390/pharmaceutics11060290 Unde, J.S., Ahirwar, K., Kumar, A., Ali Alshehri, S., Wahab, S., Kesharwani, P. and Shukla, R. 2024. Manoeuvring the innovative drug delivery systems for veterinary therapeutics: present day demand. Eur. Polym. J. 215, 113244; doi:10.1016/j.eurpolymj.2024.113244 Ungerfeld, E.M. 2020. Metabolic hydrogen flows in rumen fermentation: principles and possibilities of interventions. Front. Microbiol. 11, 589; doi:10.3389/fmicb.2020.00589 Ungerfeld, E.M. and Pitta, D. 2024. Review: Biological consequences of the inhibition of rumen methanogenesis. Animal 2024, 101170; doi:10.1016/j.animal.2024.101170 Upadhaya, S.D. and Kim, I.H. 2020. Importance of micronutrients in bone health of monogastric animals and techniques to improve the bioavailability of micronutrient supplementsa review. Asian-Australasian J. Anim. Sci. 33, 1885–1895; doi:10.5713/ajas.19.0945 Ur Rehman, H., Nawaz, M.A., Pervez, S., Jamal, M., Attaullah, M., Aman, A. and Ul Qader, S.A. 2020. Encapsulation of pectinase within polyacrylamide gel: characterization of its catalytic properties for continuous industrial uses. Heliyon 6, e04578; doi:10.1016/j.heliyon.2020.e04578 Valenzuela, C., Lagos, G., Figueroa, J. and Tadich, T. 2016. Behavior of suckling pigs supplemented with an encapsulated iron oral formula. J. Vet. Behav. 13, 6–9; doi:10.1016/j.jveb.2016.03.002 van Herten, J. and Meijboom, F.L.B. 2018. 43. Veterinary responsibilities within the one health framework. Food Ethics 2019, 281–286; doi:10.3920/978-90-8686-869-8_43 Vidhamaly, V., Bellingham, K., Newton, P.N. and Caillet, C. 2022. The quality of veterinary medicines and their implications for One Health. BMJ Glob. Heal. 7, 1–13; doi:10.1136/bmjgh-2022-008564 Vítor, A.C.M., Francisco, A.E., Silva, J., Pinho, M., Huws, S.A., Santos-Silva, J., Bessa, R.J.B. and Alves, S.P. 2021. Freeze-dried Nannochloropsis oceanica biomass protects eicosapentaenoic acid (EPA) from metabolization in the rumen of lambs. Sci. Rep. 11, 1–16; doi:10.1038/s41598-021-01255-w Vittorazzi, P.C., Marques, J.A., Takiya, C.S., Chesini, R.G., Bugoni, M., da Silva, G.G., Nunes, A.T., Silva, T.B.P., Dias, M.S.S., Grigoletto, N.T.S., Cortinhas, C.S., Acedo, T.S. and Renno, F.P. 2021. Increasing doses of carbohydrases: effects on rumen fermentation, nutrient digestibility, and performance of mid-lactation cows. J. Dairy Sci. 104, 12508–12519; doi:10.3168/jds.2021-20514 Volmer, J.G., McRae, H. and Morrison, M. 2023. The evolving role of methanogenic archaea in mammalian microbiomes. Front. Microbiol. 14, 1–21; doi:10.3389/fmicb.2023.1268451 Wang, D., Jiang, Q., Dong, Z., Meng, T., Hu, F., Wang, J. and Yuan, H. 2023a. Nanocarriers transport across the gastrointestinal barriers: The contribution to oral bioavailability via blood circulation and lymphatic pathway. Adv. Drug Deliv. Rev. 203, 115130; doi:10.1016/j.addr.2023.115130 Wang, J., Deng, L., Chen, M., Che, Y., Li, L., Zhu, L., Chen, G. and Feng, T. 2024. Phytogenic feed additives as natural antibiotic alternatives in animal health and production: A review of the literature of the last decade. Anim. Nutr. 17, 244–264; doi:10.1016/j.aninu.2024.01.012 Wang, L., Li, Y., Zhang, Y. and Wang, L. 2020. The effects of different concentrate-to-forage ratio diets on rumen bacterial microbiota and the structures of holstein cows during the feeding cycle. Animals 10, 957; doi:10.3390/ani10060957 Wang, Y., Ahmad, I., Leung, T., Lin, J., Chen, W., Liu, F., Ng, A.M.C., Zhang, Y. and Djurišić, A.B. 2022. Encapsulation and stability testing of perovskite solar cells for real life applications. ACS Mater. Au 2, 215–236; doi:10.1021/acsmaterialsau.1c00045 Wang, Z., Li, Q., Lan, X., Shen, W., Wan, F., He, J., Tang, S. and Tan, Z. 2023b. Evaluation of stirring time through a rumen simulation technique: Influences on rumen fermentation and bacterial community. Front. Microbiol. 14, 1–9; doi:10.3389/fmicb.2023.1103222 Wei, W., Zhen, Y., Wang, Y., Shahzad, K. and Wang, M. 2022. Advances of rumen functional bacteria and the application of micro-encapsulation fermentation technology in ruminants: a review. Fermentation 8, 564; doi:10.3390/fermentation8100564 Weimer, P.J. 2022. Degradation of cellulose and hemicellulose by ruminal microorganisms. Microorganisms 10, 345; doi:10.3390/microorganisms10122345 Windsor, P.A. 2022. Role of topical anaesthesia in pain management of farm animals, a changing paradigm. Animals 12, 459; doi:10.3390/ani12182459 Wu, Q., Xing, Z., Liao, J., Zhu, L., Zhang, R., Wang, S., Wang, C., Ma, Y. and Wang, Y. 2022. Effects of glutamine on rumen digestive enzymes and the barrier function of the ruminal epithelium in hu lambs fed a high-concentrate finishing diet. Animals 12, 1–11; doi:10.3390/ani12233418 Xu, Q., Qiao, Q., Gao, Y., Hou, J., Hu, M., Du, Y., Zhao, K. and Li, X. 2021. Gut microbiota and their role in health and metabolic disease of dairy cow. Front. Nutr. 8, 701511; doi:10.3389/fnut.2021.701511 Yang, Y. and Park, K. 2024. Effects of Centella asiatica extracts on rumen in vitro fermentation characteristics and digestibility. Animals 14, 1–8. Yanza, Y.R., Szumacher-Strabel, M., Jayanegara, A., Kasenta, A.M., Gao, M., Huang, H., Patra, A.K., Warzych, E. and Cieślak, A. 2021. The effects of dietary medium-chain fatty acids on ruminal methanogenesis and fermentation in vitro and in vivo: a meta-analysis. J. Anim. Physiol. Anim. Nutr. (Berl). 105, 874–889; doi:10.1111/jpn.13367 Yilmaz Atay, H. 2019. Antibacterial activity of chitosan-based systems BT. In Functional chitosan: drug delivery and biomedical applications. Eds., Sougata, J. and Subrata, J. Singapore: Springer Singapore, pp: 457–489; doi:10.1007/978-981-15-0263-7_15 Youssef, F.S., El-Banna, H.A., Elzorba, H.Y. and Galal, A.M. 2019. Application of some nanoparticles in the field of veterinary medicine. Int. J. Vet. Sci. Med. 7, 78–93; doi:10.1080/23144599.2019.1691379 Yu, Z., Yan, M. and Somasundaram, S. 2024. Rumen protozoa and viruses: The predators within and their functions—A mini-review. JDS Commun. 5, 236–240; doi:10.3168/jdsc.2023-0433 Zaaba, N.F. and Jaafar, M. 2020. A review on degradation mechanisms of polylactic acid: hydrolytic, photodegradative, microbial, and enzymatic degradation. Polym. Eng. Sci. 60, 2061–2075; doi:10.1002/pen.25511 Zabot, G.L., Schaefer Rodrigues, F., Polano Ody, L., Vinícius Tres, M., Herrera, E., Palacin, H., Córdova-Ramos, J.S., Best, I. and Olivera-Montenegro, L. 2022. Encapsulation of bioactive compounds for food and agricultural applications. Polymers (Basel). 14, 194; doi:10.3390/polym14194194 Zhang, T., Luo, Y., Wang, M., Chen, F., Liu, J., Meng, K. and Zhao, H. 2020. Double-layered microcapsules significantly improve the long-term effectiveness of essential oil. Polymers 12, 651; doi:10.3390/POLYM12081651 Zhang, Z., Gao, X., Dong, W., Huang, B., Wang, Y., Zhu, M. and Wang, C. 2022. Plant cell wall breakdown by hindgut microorganisms: can we get scientific insights from rumen microorganisms? J. Equine Vet. Sci. 115, 104027; doi:10.1016/j.jevs.2022.104027 Zhao, C., Zhu, Y., Kong, B., Huang, Y., Yan, D., Tan, H. and Shang, L. 2020. Dual-core prebiotic microcapsule encapsulating probiotics for metabolic syndrome. ACS Appl. Mater. Interfaces 12, 42586–42594; doi:10.1021/acsami.0c13518 Zhou, J., Xue, Benchu, Hu, A., Yue, S., Wu, M., Hong, Q., Wu, Y., Wang, Z., Wang, L., Peng, Q., Xue. and Bai, 2022. Effect of dietary peNDF levels on digestibility and rumen fermentation, and microbial community in growing goats. Front. Microbiol. 13, 950587; doi:10.3389/fmicb.2022.950587 Zhou, Y., Guo, L., Dai, G., Li, B., Bai, Y., Wang, W., Chen, S. and Zhang, J. 2024. An overview of polymeric nanoplatforms to deliver veterinary antimicrobials. Nanomaterials 14, 1–21; doi:10.3390/nano14040341 Zhu, J., Ren, A., Jiao, J., Shen, W., Yang, L., Zhou, C. and Tan, Z. 2022. Effects of non-protein nitrogen sources on in vitro rumen fermentation characteristics and microbial diversity. Front. Anim. Sci. 3, 1–10; doi:10.3389/fanim.2022.891898 Zou, X., Liu, G., Meng, F., Hong, L., Li, Y., Lian, Z., Yang, Z., Luo, C. and Liu, D. 2020. Exploring the rumen and cecum microbial community from fetus to adulthood in goat. Animal 10, 639; doi:10.3390/ani10091639 Zuhud, R. 2020. Food security challenges and opportunities in Indonesia post COVID-19. In Advances in food security and sustainability. Ed., Cohen, M.J. Washington, DC: Elsevier Inc., pp: 119–62. | ||
| How to Cite this Article |
| Pubmed Style Yedi Herdiana. Polymeric rumen-stable delivery systems for delivering nutricines. Open Vet. J.. 2025; 15(2): 565-593. doi:10.5455/OVJ.2025.v15.i2.7 Web Style Yedi Herdiana. Polymeric rumen-stable delivery systems for delivering nutricines. https://www.openveterinaryjournal.com/?mno=228425 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.7 AMA (American Medical Association) Style Yedi Herdiana. Polymeric rumen-stable delivery systems for delivering nutricines. Open Vet. J.. 2025; 15(2): 565-593. doi:10.5455/OVJ.2025.v15.i2.7 Vancouver/ICMJE Style Yedi Herdiana. Polymeric rumen-stable delivery systems for delivering nutricines. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 565-593. doi:10.5455/OVJ.2025.v15.i2.7 Harvard Style Yedi Herdiana (2025) Polymeric rumen-stable delivery systems for delivering nutricines. Open Vet. J., 15 (2), 565-593. doi:10.5455/OVJ.2025.v15.i2.7 Turabian Style Yedi Herdiana. 2025. Polymeric rumen-stable delivery systems for delivering nutricines. Open Veterinary Journal, 15 (2), 565-593. doi:10.5455/OVJ.2025.v15.i2.7 Chicago Style Yedi Herdiana. "Polymeric rumen-stable delivery systems for delivering nutricines." Open Veterinary Journal 15 (2025), 565-593. doi:10.5455/OVJ.2025.v15.i2.7 MLA (The Modern Language Association) Style Yedi Herdiana. "Polymeric rumen-stable delivery systems for delivering nutricines." Open Veterinary Journal 15.2 (2025), 565-593. Print. doi:10.5455/OVJ.2025.v15.i2.7 APA (American Psychological Association) Style Yedi Herdiana (2025) Polymeric rumen-stable delivery systems for delivering nutricines. Open Veterinary Journal, 15 (2), 565-593. doi:10.5455/OVJ.2025.v15.i2.7 |