| Research Article | ||
Open Vet. J.. 2025; 15(2): 931-938 Open Veterinary Journal, (2025), Vol. 15(2): 931-938 Research Article Mitigating Cadmium Toxicity: Role of Caffeine in the Reduction of Oxidative StressAhmed Aljazzar*Department of Pathology, College of Veterinary Medicine, King Faisal University, Al Hofuf, Saudi Arabia *Corresponding Author: Ahmed Aljazzar. Department of Pathology, College of Veterinary Medicine, King Faisal University, Al Hofuf, Saudi Arabia. Email: ajazzar [at] kfu.edu.sa Submitted: 30/12/2024 Accepted: 07/02/2025 Published: 28/02/2025 © 2025 Open Veterinary Journal
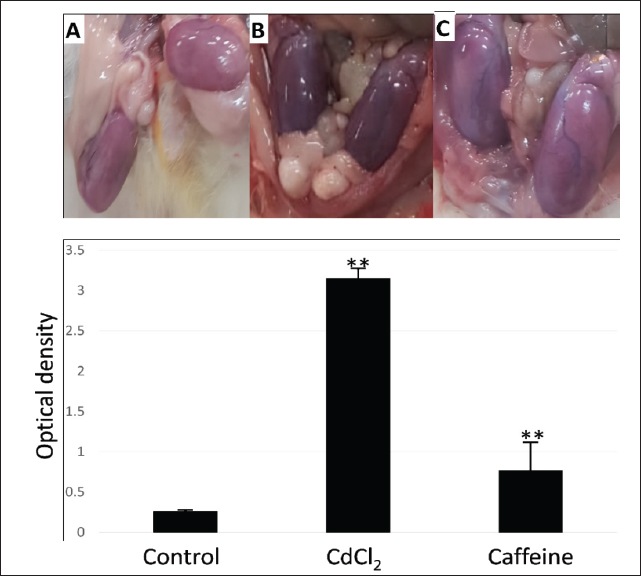
AbstractBackground: Cadmium (Cd) is a common environmental pollutant that causes severe health issues. Many studies have proposed antioxidant foods to combat Cd toxicity. One such antioxidant agent is caffeine, and hence, this study investigated its effects. Aim: To investigate the effect of caffeine as an antioxidant agent against Cd toxicity. Methods: Twelve male albino mice were divided into three groups of four: Group 1: Administered 1 ml of saline orally for 7 days and injected with 1 ml of saline subcutaneously (S/C) on the eighth day. Group 2: Patients were orally administered 1 ml of saline for 7 days and injected with 1-ml of cadmium chloride (CdCl2) S/C on the eighth day. Group 3: Administered 1 ml of caffeine orally for seven days and injected with CdCl2 S/C on the eighth day. Results: Results showed that caffeine can reduce Cd-induced testicular hemorrhage, lung injury, and kidney injury. Additionally, caffeine was found to significantly reduce the Cd-induced inflammatory response in lung-associated lymphoid tissue. Conclusion: These findings suggest that the daily intake of caffeine could protect against Cd toxicity. Keywords: Antioxidant, Cadmium, Caffeine, Heavy metal. IntroductionCadmium (Cd) is a prevalent environmental pollutant with significant adverse effects on human and animal health (Genchi et al., 2020). Unlike essential trace elements, Cd has no physiological role and is highly toxic even at low exposure levels (Sinicropi et al., 2010; Friberg et al., 2019; Genchi et al., 2020). It ranks seventh on the Agency for Toxic Substances and Disease Registry’s priority list, underscoring its potential danger (Cirmi et al., 2021). Human exposure to Cd commonly occurs through contaminated food, air, and water, primarily due to its use in products such as nickel-cadmium batteries, pigments, and electroplating materials to protect from corrosion (IARC, 1993; Karunakaran and Dhanalakshmi, 2009; Rafati Rahimzadeh et al., 2017; Hayat et al., 2019; Genchi et al., 2020). Cd is naturally found in zinc, copper, and lead sulfide ores. Due to its chemical properties, it is widely dispersed in the environment, leading to contamination of food, water, and air (WHO, 1993; Sinicropi et al., 2010). This widespread contamination results in Cd accumulation in various organs, particularly the kidneys, which are the primary targets of Cd toxicity (Brzóska et al., 2004; Babaknejad et al., 2016). Seafood such as crustaceans, oysters, and crabs, as well as offal such as livers and kidneys, contains high concentrations of Cd (Satarug, 2018; Genchi et al., 2020). The cumulative nature of Cd due to its long half-life and inefficient elimination mechanisms can lead to severe health issues, including nephrotoxicity, osteoporosis, cardiovascular problems, and reproductive issues (Satarug et al., 2017; Fatima et al., 2019; Kumar and Sharma, 2019; Cirmi et al., 2021). The toxic effects of Cd are mediated through multiple pathways, primarily by inducing oxidative stress. Cd exposure increases the levels of reactive oxygen species (ROS), which in turn deplete antioxidants such as glutathione and cause protein cross-linking, lipid peroxidation, and inflammation (Matović et al., 2011; Ansari et al., 2017). Normally, the body mitigates this induced oxidative stress through enzymes such as superoxide dismutase and catalase (Ali Al-Gnami, 2014; Amamou et al., 2015). Emerging research suggests that polyphenols and antioxidants can mitigate Cd toxicity. Dietary intake of antioxidant-rich foods, such as grape seeds, ginger, tea, and berries, may have protective effects against Cd and toxicity from other heavy metals (Baiomy and Mansour, 2016; Brzóska et al., 2016). Caffeine, which is a constituent in many beverages and is commonly consumed, also has potential antioxidant protective effects (Ahluwalia and Herrick, 2015; Ősz et al., 2022). Hence, this study aimed to investigate the protective effects of caffeine against Cd-induced toxicity in various organs and contribute to the development of dietary strategies to counteract heavy metal toxicity. Materials and MethodsChemicalsCadmium chloride (CdCl2), tris-HCl, MgCl2, and CaCl2 were obtained from Sigma-Aldrich (St. Louis, MO). AnimalsTwelve apparently healthy male albino mice weighing 175–200 g were kept in plastic cages and were allowed free access to clean food and water ad-libitum. They were acclimatized for two weeks. The mice were randomly divided into three groups of four animals each. Group 1: This group was administered 1 ml of saline orally for 7 days, and on the eighth day, the mice were injected with 1 ml of saline subcutaneously (S/C) (control). Group 2: This group was administered 1 ml of saline orally for 7 days, and on the eighth day, the mice were injected with 1 ml CdCl2 S/C at a dose of 7 mg/kg.b.wt. (Positive control) (Niewenhuis and Prozialeck, 1987). Group 3: This group was administered 1 ml of caffeine extracted from green tea (30 g/100 ml boiling water) orally for 7 days, and on the eighth day, the mice were injected with CdCl2 S/C. Twenty-four hours later, the mice were euthanized and the organs were removed. The procedures used in this study were approved by the Institutional Animal Care and Use Committee and the Research Ethics Committee of King Faisal University; ethical approval number (ETHICS2416). HistopathologyThe lung and kidney tissues were harvested and immersion-fixed in 4% formalin for 72 hours. Fixed tissues were trimmed, embedded in paraffin wax, and sectioned at 5 μm before being stained with hematoxylin and eosin. The sections were then analyzed using histopathological techniques and light microscopy. Gas chromatography–mass spectrometry (GC–MS) analysisThe crude ethanolic extracts analysis was conducted using a Shimadzu GCMS-QP2010 Plus (Japan). The mobile phase is Helium high purity (99.9999% pure) used at a flow rate of 1 ml/minute. The instrument is equipped with a capillary column DB-5MS (30-m length, 0.25-mm thickness, 0.25 m diameter). A 1 µl sample was injected into the splitless inlet in splitless mode at 260°C. The temperature programming of the GC–MS manalysis starts by setting an oven’s temperature to 70°C for 7 minutes. This increased by 7°C/minute up to 280°C, which was then maintained for 10 minutes. The temperature of the interface was 200°C, and the temperature of the ion source was 250°C. The range of scan mode (50–550 amu) used for data acquisition. The relative amount of each constituent was calculated by measuring the corresponding peak area and represented as a percentage of the sum of the areas of all the peaks (El Sherif et al., 2022). Spectrophotometry of hemorrhagingTo estimate the level of CdCl2-induced hemorrhaging, testes from all different mice groups were homogenized in 10 ml of 5 mM (tris-HCl buffer, pH, 7.0) containing 1 mM MgCl2 and 100 mM CaCl2. The homogenate was then centrifuged at 5,000 rpm for 30 minutes. A 1-ml aliquot of the supernatant was diluted in 5 ml of buffer. The absorbance of hemoglobin as evidence of hemorrhaging was detected at 414 nm using a Spectronic 21 spectrophotometer (Busch and Lomb) (Niewenhuis and Prozialeck, 1987). ImageJ image analysisThe bronchus-associated lymphoid tissue (BALT) and cellular viability were evaluated using ImageJ software. For BALT, the areas of all BALT in a section of all the different treatment groups were measured using the ImageJ area analysis tool. For determining cellular viability, all intact nuclei in sections from different groups were counted using a nuclei-counting plugin and threshold tool. For both BALT and cellular viability, the means of the different groups were compared using Student’s t-tests. Statistical analysisAll statistical analyses were performed using the PSPP statistical package. Student’s t-test was used to compare two groups. Measurements are presented as ± SEM. ResultsCaffeine protects testes against Cd-induced damageTo investigate whether caffeine has any protective effect against Cd toxicity, we first performed an extraction process using green tea and GC–MS. This extraction process revealed only one component of caffeine. This might be due to the fact that this extraction method only retains the ethanol-soluble component. The extraction retention time was 15.835 min and yielded 3.25 x 100.000 (Fig. 1). As expected, testicles from mice that received 1 ml of CdCl2 S/C showed severe testicular hemorrhage (Fig. 2B) and significant increases in the optical density of hemoglobin (Fig. 2). However, the dietary intake of extracted caffeine for 7 days prior to CdCl2 treatment significantly reduced the observed testicular hemorrhaging and optical density of hemoglobin compared with the control group. This suggests that caffeine has a protective effect against Cd-induced toxicity. Caffeine ameliorates Cd-induced damage in lungs and kidneysConsistent with previous studies, histopathological analysis of lung sections from CdCl2-treated mice showed bronchial endothelia sloughing, alveolar capillaries congestion, and alveolar septal thickening (Fig. 3C and D) (El-Ebiary et al., 2016; Rasheed and Abdullah, 2022; Akaras et al., 2023). However, the addition of caffeine to the diet of treated mice resulted in intact bronchial endothelia and reduced the severity of Cd-induced congestion (Fig. 3E and F). Furthermore, the evaluation of bronchus-associated lymphoid organs revealed a significant increase in BALT areas in CdCl2-treated mice compared with the control group (Fig. 4). On the other hand, mice that received caffeine had significantly smaller BALT areas, which suggests that there is immune stimulation after CdCl2 treatment and that this immune stimulation can be protected via a caffeine diet (Fig. 4).
Fig. 1. Green tea China gas chromatography—mass spectrometry: The above figure shows a profile of the methanolic extract of green tea China. It is typical for caffeine, with a retention time of 15.835 and a concentration of 3.25 x 100.000. No other extracts were detected.
Fig. 2. Caffeine mitigating cadmium (Cd)-induced testicular damage: (A) control, (B) cadmium chloride (CdCl2), and (C) caffeine and CdCl2. This figure shows how caffeine treatment relieved Cd-induced damage. (B) shows congested and hemorrhagic testicles. When caffeine is included in the diet, there is clear protection against Cd-induced damage (D). Moreover, analysis of kidney tissue from CdCl2-treated mice revealed vacuolation and degeneration of kidney tubules (Fig. 5B). Nonetheless, receiving caffeine as part of the diet resulted in a mild improvement in pathological damage (Fig. 5C). To further investigate kidney health, the number of nuclei in sections from all groups was counted to determine whether Cd affects the number of viable cells in tissue. Unexpectedly, no significant differences were found between all treated groups; the counts of nuclei were comparable (Fig. 5).
Fig. 3. Caffeine extract reduces cadmium chloride (CdCl2)-induced tissue damage: Mice treated with CdCl2 showed sloughed bronchial epithelial linings and the thickening/congestion of alveolar septa (C, D). Caffein administration protected the bronchial epithelial lining and prevented congestion of the alveolar capillaries. However, the thickness of the alveolar septa did not change (E, F).
Fig. 4. Caffeine extract mitigates cadmium chloride (CdCl2)-induced immune responses: Analysis of BALT area sizes from all groups showed an overt immune response in the CdCl2-treated group compared with the control. The addition of caffeine extract to the treatment regimen significantly reduced this immune reaction. DiscussionCd toxicity due to contaminated food and water is a global health hazard. As a protective measure against this toxicity, studies have demonstrated that the inclusion of foods and beverages with high antioxidant properties might protect against Cd-induced oxidative stress and tissue damage, which are the main reasons for its toxicity. Therefore, since caffeine is a constituent of most consumed beverages, such as tea and coffee, and has been demonstrated to have antioxidant properties, we investigated its potential protective properties against Cd toxicity. In this study, our data demonstrate that caffeine has protective properties against Cd-induced toxicity. Mice that consumed caffeine for 7 days before receiving 1 ml of CdCl2 showed clear resistance to Cd-induced damage. For instance, testes from these mice showed a significant reduction in the amount of visually detected hemorrhaging, which was evaluated by measuring the amount of Hg in the testicular tissue via spectrophotometry at an absorbance of 400 nm. Caffeine significantly reduced testicular hemorrhaging and the optical density of Hg in the testicles of mice treated with CdCl2. This suggests that caffeine can alleviate Cd-induced testicular damage. The antioxidant properties of caffeine, which help reduce oxidative stress and inflammation, likely play a crucial role in this protective effect (Schepici et al., 2020). Furthermore, lung tissue from Cd-treated mice showed pathological damage, as represented by bronchial endothelia sloughing, alveolar capillaries congestion, and alveolar septal thickening. However, these pathologies were mitigated if caffeine was included in the diet, showing a clear decrease in the level of alveolar capillary congestion and intact bronchial endothelium. This protective effect may be supported by caffeine’s ability to enhance antioxidant defenses and reduce oxidative stress in pulmonary tissues (Manca et al., 1994; Wang et al., 2023). Furthermore, caffeine’s anti-inflammatory properties may also contribute to its protective effects on lung tissue, as inflammation is a significant component of Cd-induced pulmonary damage (Genchi et al., 2020). Additionally, sections of lung tissue from Cd-treated mice showed obvious expansion of the BALT, suggesting that Cd stimulates an immune response. This immune response was mitigated by caffeine consumption, as mice that consumed caffeine exhibited significantly normal BALT sizes. This finding aligns with studies that demonstrated that Cd can stimulate immune responses (Blakley, 1985; Knoell and Wyatt, 2021; Wang et al., 2021). In kidney tissue, Cd-treated mice exhibited significant vacuolation and degeneration of kidney tubules. However, mice that received caffeine showed mild improvement in these pathological changes with no changes in the number of cells in the section. This indicates that caffeine can partially protect against Cd-induced nephrotoxicity. The lack of significant differences in the number of viable cells among the treated groups suggests that caffeine’s protective effect may primarily involve preserving cellular function rather than preventing cell death. Caffeine’s role in preserving kidney function could be attributed to its ability to reduce oxidative damage and improve mitochondrial function, which is critical in maintaining renal health (Satarug, 2018; Vieira et al., 2020; Ősz et al., 2022).
Fig. 5. Caffeine has mild protective effects for kidneys: Sections of kidneys from cadmium chloride-treated mice (B) show degenerated and vacuolated kidney tubules (arrowhead) compared with the control group (A). Caffein treatment resulted in mild protective effects on kidney parenchyma (C). The analysis of kidney nuclei numbers in all groups revealed no significant differences (D). The primary mechanism through which caffeine exerts its protective effects against Cd toxicity is likely related to its antioxidant properties. Cd exposure induces oxidative stress by ROS and depletes antioxidant defenses, such as glutathione. Caffeine, which is an effective antioxidant, can counteract this oxidative stress, reducing the damage caused by ROS (Lacorte et al., 2013; Ikram et al., 2020). Additionally, caffeine’s role in enhancing the activity of endogenous antioxidant enzymes, such as superoxide dismutase and catalase, further supports its protective effects (Ősz et al., 2022). Additionally, caffeine may modulate the expression of microRNAs (miRNAs) involved in oxidative stress and inflammation. Cd exposure is known to dysregulate miRNA expression, contributing to tissue damage and disease progression (Nguyen and Kim, 2023). By influencing miRNA expression, caffeine may help restore normal cellular function and mitigate Cd-induced toxicity. For instance, certain miRNAs affected by Cd exposure are involved in pathways related to cell proliferation, apoptosis, and immune response, and modulating these miRNAs could significantly impact the cellular response to Cd (Khan et al., 2019; Ikram et al., 2020; Labib and Galal, 2021; Nguyen and Kim, 2023). The findings of this study suggest that incorporating caffeine-rich foods and beverages into the diet could provide a dietary method for counteracting Cd toxicity. Given the widespread use of caffeine in beverages such as tea and coffee, this approach can be easily implemented. Moreover, the potential of caffeine as a protective agent against other heavy metals and environmental toxins warrants further investigation (Ősz et al., 2022). Future research should focus on further elucidating the molecular mechanisms underlying caffeine’s protective effects against Cd toxicity. Exploring the combined effects of caffeine with other dietary antioxidants could also reveal synergistic protective mechanisms that enhance resilience against Cd toxicity (Khan et al., 2019; Labib and Galal, 2021). AcknowledgmentsI would like to thank Professors Yehia Hussein and Ahmed Meligy for providing the samples for this research. Conflict of interestThere are no conflicts of interest to declare. FundingThis research was funded by the Deanship of Scientific Research at King Faisal University, Saudi Arabia. Grant no. 241807. Author’s contributionsAll the analysis in this research was performed by Dr Ahmed Aljazzar. Data availabilityAll data supporting the findings of this study are available in the manuscript. ReferencesAhluwalia, N. and Herrick, K., 2015. Caffeine intake from food and beverage sources and trends among children and adolescents in the United States: review of national quantitative studies from 1999 to 2011. Adv. Nutr. 6(1), 102–11. Akaras, N., Ileriturk, M., Gur, C., Kucukler, S., Oz, M. and Kandemir, F.M. 2023. The protective effects of chrysin on cadmium-induced pulmonary toxicity; a multi-biomarker approach. Environm. Sci. Pollut. Res. 30(38), 89479–94. Ali Al-Gnami, S.A. 2014. Effect of polyphenols which extracted from green tea in reduce toxic effects of cadmium sulfate in rat, s livers. IOSR J. Pharm. Biol. Sci. 9(2), 53–8. Amamou, F., Nemmiche, S., Meziane, R. kaouthar, Didi, A., Yazit, S.M. and Chabane-Sari, D. 2015. Protective effect of olive oil and colocynth oil against cadmium-induced oxidative stress in the liver of Wistar rats. Food Chem. Toxicol. 78, 177–84. Ansari, M.A., Raish, M., Ahmad, A., Alkharfy, K.M., Ahmad, S.F., Attia, S.M., Alsaad, A.M.S. and Bakheet, S.A. 2017. Sinapic acid ameliorate cadmium-induced nephrotoxicity: in vivo possible involvement of oxidative stress, apoptosis, and inflammation via NF-κB downregulation. Environ.Toxicol. Pharmacol. 51, 100–7. Babaknejad, N., Moshtaghie, A.A., Nayeri, H., Hani, M. and Bahrami, S. 2016. Protective role of zinc and magnesium against cadmium nephrotoxicity in male wistar rats. Biol. Trace Elem. Res. 174(1), 112–20. Baiomy, A.A. and Mansour, A.A. 2016. Genetic and histopathological responses to cadmium toxicity in rabbit’s kidney and liver: protection by ginger (Zingiber officinale). Biol. Trace Elem. Res. 170(2), 320–9. Blakley, B.R. 1985. The effect of cadmium chloride on the immune response in mice. Can. J. Comp. Med. 49(1), 104–8. Brzóska, M.M., Borowska, S. and Tomczyk, M. 2016. Antioxidants as a potential preventive and therapeutic strategy for cadmium. Curr. Drug Targets. 17(12), 1350–84. Brzóska, M.M., Kamiński, M., Dziki, M. and Moniuszko-Jakoniuk, J. 2004. Changes in the structure and function of the kidney of rats chronically exposed to cadmium. II. Histoenzymatic studies. Arch. Toxicol. 78(4), 226–31. Cirmi, S., Maugeri, A., Micali, A., Marini, H.R., Puzzolo, D., Santoro, G., Freni, J., Squadrito, F., Irrera, N., Pallio, G., Navarra, M. and Minutoli, L. 2021. Cadmium-induced kidney injury in mice is counteracted by a flavonoid-rich extract of bergamot juice, alone or in association with curcumin and resveratrol, via the enhancement of different defense mechanisms. Biomedicines 9(12), 1797. El-Ebiary, A.A., El-Ghaiesh, S., Hantash, E. and Alomar, S. 2016. Mitigation of cadmium-induced lung injury by Nigella sativa oil. Envirn. Sci. Pollut. Res. 23(24), 25356–63. El Sherif, F., Alkuwayti, M.A. and Khattab, S. 2022. Foliar spraying of salicylic acid enhances growth, yield, and curcuminoid biosynthesis gene expression as well as curcuminoid accumulation in Curcuma longa. Horticulturae 8(5), 417. Fatima, G., Raza, A.M., Hadi, N., Nigam, N. and Mahdi, A.A. 2019. Cadmium in human diseases: it’s more than just a mere metal. Indian J. Clin. Biochem. 34(4), 371–8. Friberg, L., Kjellström, T., Elinder, C.-G. and Nordberg, G.F. 2019. Cadmium and health: a toxicological and epidemiological appraisal. Boca Raton, FL: CRC Press. Genchi, G., Sinicropi, M. S., Lauria, G., Carocci, A. and Catalano, A. 2020. The effects of toxicity. Inter. J. Environ. Res. Public Health, 17(11), 3782. https://doi.org/10.3390/ijerph17113782 Hayat, M.T., Nauman, M., Nazir, N., Ali, S. and Bangash, N. 2019. Environmental hazards of cadmium: past, present, and future. In Cadmium toxicity and tolerance in plants: from physiology to remediation. Eds., Hasanuzzaman, M., Vara Prasad, M.N. and Fujita, M. Cambridge, MA: Academic Press, pp: 163–83. IARC. 1993. Beryllium, cadmium, mercury, and exposures in the glass manufacturing industry. IARC Monogr. Eval. Carcinog. Risks Hum. 58, 1–415. Available via http://www.ncbi.nlm.nih.gov/pubmed/8022054 Ikram, M., Park, T.J., Ali, T. and Kim, M.O. 2020. Antioxidant and neuroprotective effects of caffeine against Alzheimer’s and Parkinson’s disease: insight into the role of Nrf-2 and A2AR signaling. Antioxidants 9(9), 902. Karunakaran, C. and Dhanalakshmi, R. 2009. Selectivity in photocatalysis by particulate semiconductors. Central Europ. J. Chem. 7(1), 134–7. Khan, A., Ikram, M., Muhammad, T., Park, J. and Kim, M.O. 2019. Caffeine modulates cadmium-induced oxidative stress, neuroinflammation, and cognitive impairments by regulating Nrf-2/HO-1 in vivo and in vitro. J. Clin. Med. 8(5), 680. Knoell, D.L. and Wyatt, T.A. 2021. The adverse impact of cadmium on immune function and lung host defense. Semin. Cell Dev. Biol. 115, 70–6; doi:10.1016/j.semcdb.2020.10.007. Kumar, S. and Sharma, A. 2019. Cadmium toxicity: effects on human reproduction and fertility. Rev. Environ. Health 34(4), 327–38. Labib, H. and Galal, A. 2021. Caffeine versus antioxidant combination (Antox) and their role in modifying cadmium-induced testicular injury in adult male albino rats. Andrologia 53(2), e13948. Lacorte, L.M., Seiva, F.R.F., Rinaldi, J.C., Delella, F.K., Moroz, A., Sarobo, C., Godinho, A.F., Fávaro, W.J., Fernandes, A.A.H. and Felisbino, S.L. 2013. Caffeine reduces cadmium accumulation in the organism and enhances the levels of antioxidant protein expression in the epididymis. Reprod. Toxicol. 35(1), 137–43. Manca, D., Ricard, A.C., Van Tra, H. and Chevalier, G. 1994. Relation between lipid peroxidation and inflammation in the pulmonary toxicity of cadmium. Arch. Toxicol. 68(6), 364–9. Matović, V., Buha, A., Bulat, Z. and Dukić-ĆOsić, D. 2011. Cadmium toxicity revisited: focus on oxidative stress induction and interactions with zinc and magnesium. Arh. Hig. Rada. Toksikol. 62(1), 65–76. Nguyen, H.D. and Kim, M.S. 2023. Interactions between cadmium, lead, mercury, and arsenic and depression: a molecular mechanism involved. J. Affective Disorders 327, 315–29. Niewenhuis, R.J. and Prozialeck, W.C. 1987. Calmodulin inhibitors protect against cadmium-induced testicular damage in mice. Biol. Reprod. 37(1), 127–33. Ősz, B.-E., Jîtcă, G., Ștefănescu, R.-E., Pușcaș, A., Tero-Vescan, A. and Vari, C.-E. 2022. Caffeine and its antioxidant properties—it is all about dose and source. Inter. J. Mol. Sci. 23(21), 13074. Rafati Rahimzadeh, M., Rahimzadeh, R., Kazemi, M. and Moghadamnia, S. 2017. Cadmium toxicity and treatment: an update. Caspian J. Intern. Med. 8(3), 135–45. Rasheed, K.N. and Abdullah, A.M.K. 2022. Microscopic vision for a therapeutic effect of carnitine and arginine against cadmium toxicity in the histological structure of rat lung. Inter. J. Drug Deliv. Technol. 12(3), 1120–3. Satarug, S. 2018. Dietary cadmium intake and its effects on kidneys. Toxics 6(1), 15; doi:10.3390/toxics6010015. Satarug, S., Vesey, D.A. and Gobe, G.C. 2017. Health risk assessment of dietary cadmium intake: do current guidelines indicate how much is safe? Environ. Health Perspect. 125(3), 284–8. Schepici, G., Silvestro, S., Bramanti, P. and Mazzon, E. 2020. Caffeine: an overview of its beneficial effects in experimental models and clinical trials of Parkinson’s disease. Inter. J. Mol. Sci. 21(13), 4766. Sinicropi, M.S., Amantea, D., Caruso, A. and Saturnino, C. 2010. Chemical and biological properties of toxic metals and use of chelating agents for the pharmacological treatment of metal poisoning. Arch. Toxicol. 84(7), 501–20. Vieira, A.J.S.C., Gaspar, E.M. and Santos, P.M.P. 2020. Mechanisms of potential antioxidant activity of caffeine. Rad. Phys. Chem. 174, 108968. Wang, W.J., Peng, K., Lu, X., Zhu, Y.Y., Li, Z., Qian, Q.H., Yao, Y.X., Fu, L., Wang, Y., Huang, Y.C., Zhao, H., Wang, H., Xu, D.X. and Tan, Z.X. 2023. Long-term cadmium exposure induces chronic obstructive pulmonary disease-like lung lesions in a mouse model. Sci. Total Environ. 879, 163073. Wang, Z., Sun, Y., Yao, W., Ba, Q. and Wang, H. 2021. Effects of cadmium exposure on the immune system and immunoregulation. Front. Immunol. 12, 695484. WHO. 1993. Book reviews. Ann. Occup. Hyg. 37(3), 341; doi:10.1093/annhyg/37.3.341. | ||
| How to Cite this Article |
| Pubmed Style Ahmed Aljazzar. Mitigating cadmium toxicity: Role of caffeine in the reduction of oxidative stress. Open Vet. J.. 2025; 15(2): 931-938. doi:10.5455/OVJ.2025.v15.i2.42 Web Style Ahmed Aljazzar. Mitigating cadmium toxicity: Role of caffeine in the reduction of oxidative stress. https://www.openveterinaryjournal.com/?mno=240411 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i2.42 AMA (American Medical Association) Style Ahmed Aljazzar. Mitigating cadmium toxicity: Role of caffeine in the reduction of oxidative stress. Open Vet. J.. 2025; 15(2): 931-938. doi:10.5455/OVJ.2025.v15.i2.42 Vancouver/ICMJE Style Ahmed Aljazzar. Mitigating cadmium toxicity: Role of caffeine in the reduction of oxidative stress. Open Vet. J.. (2025), [cited January 25, 2026]; 15(2): 931-938. doi:10.5455/OVJ.2025.v15.i2.42 Harvard Style Ahmed Aljazzar (2025) Mitigating cadmium toxicity: Role of caffeine in the reduction of oxidative stress. Open Vet. J., 15 (2), 931-938. doi:10.5455/OVJ.2025.v15.i2.42 Turabian Style Ahmed Aljazzar. 2025. Mitigating cadmium toxicity: Role of caffeine in the reduction of oxidative stress. Open Veterinary Journal, 15 (2), 931-938. doi:10.5455/OVJ.2025.v15.i2.42 Chicago Style Ahmed Aljazzar. "Mitigating cadmium toxicity: Role of caffeine in the reduction of oxidative stress." Open Veterinary Journal 15 (2025), 931-938. doi:10.5455/OVJ.2025.v15.i2.42 MLA (The Modern Language Association) Style Ahmed Aljazzar. "Mitigating cadmium toxicity: Role of caffeine in the reduction of oxidative stress." Open Veterinary Journal 15.2 (2025), 931-938. Print. doi:10.5455/OVJ.2025.v15.i2.42 APA (American Psychological Association) Style Ahmed Aljazzar (2025) Mitigating cadmium toxicity: Role of caffeine in the reduction of oxidative stress. Open Veterinary Journal, 15 (2), 931-938. doi:10.5455/OVJ.2025.v15.i2.42 |